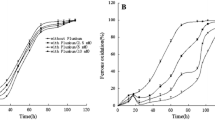
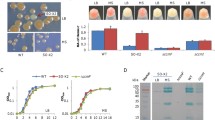
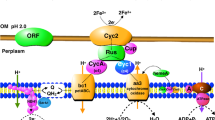
Abstract
Nitrosomonas europaea, as an ammonia-oxidizing bacterium, has a high Fe requirement and has 90 genes dedicated to Fe acquisition. Under Fe-limiting conditions (0.2 μM Fe), N. europaea was able to assimilate up to 70% of the available Fe in the medium even though it is unable to produce siderophores. Addition of exogenous siderophores to Fe-limited medium increased growth (final cell mass). Fe-limited cells had lower heme and cellular Fe contents, reduced membrane layers, and lower NH3- and NH2OH-dependent O2 consumption activities than Fe-replete cells. Fe acquisition-related proteins, such as a number of TonB-dependent Fe-siderophore receptors for ferrichrome and enterobactin and diffusion protein OmpC, were expressed to higher levels under Fe limitation, providing biochemical evidence for adaptation of N. europaea to Fe-limited conditions.


Similar content being viewed by others
Abbreviations
- OM:
-
Outer membrane
- AMO:
-
Ammonia monooxygenase
- HAO:
-
Hydroxylamine oxidoreductase
- HPLC/MS/MS:
-
High performance liquid chromatography tandem mass spectrometry
References
Andrews SC, Robinson AK, Rodriguez-Quinones F (2003) Bacterial iron homeostasis. FEMS Microbiol Rev 27:215–237
Arciero DM, Hooper AB (1993) Hydroxylamine oxidoreductase from Nitrosomonas europaea is a multimer of an octa-heme subunit. J Biol Chem 268:14645–14654
Arp DJ, Sayavedra-Soto LA, Hommes NG (2002) Molecular biology and biochemistry of ammonia oxidation by Nitrosomonas europaea. Arch Microbiol 178:250–255
Berry EA, Trumpower BL (1987) Simultaneous determination of hemes a, b, and c from pyridine hemochrome spectra. Anal Biochem 161:1–15
Braun V, Killmann H (1999) Bacterial solutions to the iron-supply problem. Trends Biochem Sci 24:104–109
Braun V, Mahren S, Ogierman M (2003) Regulation of the FecI-type ECF sigma factor by transmembrane signalling. Curr Opin Microbiol 6:173–180
Calugay RJ, Miyashita H, Okamura Y, Matsunaga T (2003) Siderophore production by the magnetic bacterium Magnetospirillum magneticum AMB-1. FEMS Microbiol Lett 218:371–375
Carter P (1971) Spectrophotometric determination of serum iron at the submicrogram level with a new reagent (ferrozine). Anal Biochem 40:450–458
Chain P et al (2003) Complete genome sequence of the ammonia-oxidizing bacterium and obligate chemolithoautotroph Nitrosomonas europaea. J Bacteriol 185:2759–2773
Champomier-Verges MC, Stintzi A, Meyer JM (1996) Acquisition of iron by the non-siderophore-producing Pseudomonas fragi. Microbiology 142(Pt 5):1191–1199
Chart H, Buck M, Stevenson P, Griffiths E (1986) Iron regulated outer membrane proteins of Escherichia coli: variations in expression due to the chelator used to restrict the availability of iron. J Gen Microbiol 132:1373–1378
Clarke TE, Tari LW, Vogel HJ (2001) Structural biology of bacterial iron uptake systems. Curr Top Med Chem 1:7–30
Ensign SA, Hyman MR, Arp DJ (1993) In vitro activation of ammonia monooxygenase from Nitrosomonas europaea by copper. J Bacteriol 175:1971–1980
Escolar L, Perez-Martin J, de Lorenzo V (1999) Opening the iron box: transcriptional metalloregulation by the Fur protein. J Bacteriol 181:6223–6229
Faraldo-Gomez JD, Sansom MS (2003) Acquisition of siderophores in gram-negative bacteria. Nat Rev Mol Cell Biol 4:105–116
Frederick CB, Szaniszlo PJ, Vickrey PE, Bentley MD, Shive W (1981) Production and isolation of siderophores from the soil fungus Epicoccum purpurascens. Biochemistry 20:2432–2436
Hageman RH, Hucklesby DP (1971) Nitrate reductase in higher plants. Meth Enzymol 23:491–503
Hassett RF, Romeo AM, Kosman DJ (1998) Regulation of high affinity iron uptake in the yeast Saccharomyces cerevisiae. Role of dioxygen and Fe. J Biol Chem 273:7628–7636
Herbik A, Bolling C, Buckhout TJ (2002) The involvement of a multicopper oxidase in iron uptake by the green algae Chlamydomonas reinhardtii. Plant Physiol 130:2039–2048
Hersman L, Lloyd T, Sposito G (1995) Siderophore-promoted dissolution of hematite. Geochim Cosmochim Acta 59:3327–3330
Hofte M, Buysens S, Koedam N, Cornelis P (1993) Zinc affects siderophore-mediated high affinity iron uptake systems in the rhizosphere Pseudomonas aeruginosa 7NSK2. Biometals 6:85–91
Hooper AB (1969) Biochemical basis of obligate autotrophy in Nitrosomonas europaea. J Bacteriol 97:776–779
Hooper AB, Erickson RH, Terry KR (1972) Electron transport systems of Nitrosomonas: isolation of a membrane-envelope fraction. J Bacteriol 110:430–438
Houk RS (1994) Elemental and isotopic analysis by inductively coupled plasma mass spectrometry. Acc Chem Res 27:333–339
Huston WM, Jennings MP, McEwan AG (2002) The multicopper oxidase of Pseudomonas aeruginosa is a ferroxidase with a central role in iron acquisition. Mol Microbiol 45:1741–1750
Hyman MR, Arp DJ (1993) An electrophoretic study of the thermal-dependent and reductant-dependent aggregation of the 27 kDa component of ammonia monooxygenase from Nitrosomonas europaea. Electrophoresis 14:619–627
Kammler M, Schon C, Hantke K (1993) Characterization of the ferrous iron uptake system of Escherichia coli. J Bacteriol 175:6212–6219
Keyer K, Gort AS, Imlay JA (1995) Superoxide and the production of oxidative DNA damage. J Bacteriol 177:6782–6790
Loper JE, Henkels MD (1999) Utilization of heterologous siderophores enhances levels of iron available to Pseudomonas putida in the rhizosphere. Appl Environ Microbiol 65:5357–5363
Mahren S, Braun V (2003) The FecI extracytoplasmic-function sigma factor of Escherichia coli interacts with the beta’ subunit of RNA polymerase. J Bacteriol 185:1796–1802
Matzanke BF (1991) Structures, coordination chemistry and functions of microbial iron chelates. In: Winkelmann G (eds) Handbook of microbial iron chelates. CRC Press Inc., Boca Raton, pp 15–64
Meyer JM (2000) Pyoverdines: pigments, siderophores and potential taxonomic markers of fluorescent Pseudomonas species. Arch Microbiol 174:135–142
Mossialos D et al (2000) Quinolobactin, a new siderophore of Pseudomonas fluorescens ATCC 17400, the production of which is repressed by the cognate pyoverdine. Appl Environ Microbiol 66:487–492
Murray RG, Watson SW (1965) Structure of Nitrosocystis oceanus and comparison with Nitrosomonas and Nitrobacter. J Bacteriol 89:1594–1609
Neilands JB (1981) Iron absorption and transport in microorganisms. Annu Rev Nutr 1:27–46
Neilands JB (1995) Siderophores: structure and function of microbial iron transport compounds. J Biol Chem 270:26723–26726
Outten CE, O’Halloran TV (2001) Femtomolar sensitivity of metalloregulatory proteins controlling zinc homeostasis. Science 292:2488–2492
Page WJ, Huyer M (1984) Derepression of the Azotobacter vinelandii siderophore system, using iron-containing minerals to limit iron repletion. J Bacteriol 158:496–502
Page WJ, von Tigerstrom M (1982) Iron- and molybdenum-repressible outer membrane proteins in competent Azotobacter vinelandii. J Bacteriol 151:237–242
Payne SM (1994) Detection, isolation, and characterization of siderophores. Methods Enzymol 235:329–344
Poole K, Neshat S, Heinrichs D (1991) Pyoverdine-mediated iron transport in Pseudomonas aeruginosa: involvement of a high-molecular-mass outer membrane protein. FEMS Microbiol Lett 62:1–5
Powell PE, Szaniszlo PJ, Reid CP (1983) Confirmation of occurrence of hydroxamate siderophores in soil by a novel Escherichia coli bioassay. Appl Environ Microbiol 46:1080–1083
Quatrini R, Jedlicki E, Holmes DS (2005) Genomic insights into the iron uptake mechanisms of the biomining microorganism Acidithiobacillus ferrooxidans. J Ind Microbiol Biotechnol 32:606–614
Ravel J, Cornelis P (2003) Genomics of pyoverdine-mediated iron uptake in pseudomonads. Trends Microbiol 11:195–200
Schalk IJ, Yue WW, Buchanan SK (2004) Recognition of iron-free siderophores by TonB-dependent iron transporters. Mol Microbiol 54:14–22
Schmidt I, Look C, Bock E, Jetten MS (2004) Ammonium and hydroxylamine uptake and accumulation in Nitrosomonas. Microbiology 150:1405–1412
Schwyn B, Neilands JB (1987) Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160:47–56
Shiemke AK, Arp DJ, Sayavedra-Soto LA (2004) Inhibition of membrane-bound methane monooxygenase and ammonia monooxygenase by diphenyliodonium: implications for electron transfer. J Bacteriol 186:928–937
Spizzo T, Byersdorfer C, Duesterhoeft S, Eide D (1997) The yeast FET5 gene encodes a FET3-related multicopper oxidase implicated in iron transport. Mol Gen Genet 256:547–556
Stearman R, Yuan DS, Yamaguchi-Iwai Y, Klausner RD, Dancis A (1996) A permease-oxidase complex involved in high-affinity iron uptake in yeast. Science 271:1552–1557
Stein LY, Arp DJ (1998) Ammonia limitation results in a loss of ammonia-oxidizing activity in Nitrosomonas europaea. Appl Environ Microbiol 64:1514–1521
Thompson H, Tersteegen A, Thauer RK, Hedderich R (1998) Two malate dehydrogenases in Methanobacterium thermoautotrophicum. Arch Microbiol 170:38–42
Tindale AE, Mehrotra M, Ottem D, Page WJ (2000) Dual regulation of catecholate siderophore biosynthesis in Azotobacter vinelandii by iron and oxidative stress. Microbiology 146(Pt 7):1617–1626
Upadhyay AK, Petasis DT, Arciero DM, Hooper AB, Hendrich MP (2003) Spectroscopic characterization and assignment of reduction potentials in the tetraheme cytochrome C554 from Nitrosomonas europaea. J Am Chem Soc 125:1738–1747
Velayudhan J et al (2000) Iron acquisition and virulence in Helicobacter pylori: a major role for FeoB, a high-affinity ferrous iron transporter. Mol Microbiol 37:274–286
Verderber E, Lucast LJ, Van Dehy JA, Cozart P, Etter JB, Best EA (1997) Role of the hemA gene product and delta-aminolevulinic acid in regulation of Escherichia coli heme synthesis. J Bacteriol 179:4583–4590
Visca P, Leoni L, Wilson MJ, Lamont IL (2002) Iron transport and regulation, cell signalling and genomics: lessons from Escherichia coli and Pseudomonas. Mol Microbiol 45:1177–1190
Whittaker M, Bergmann D, Arciero D, Hooper AB (2000) Electron transfer during the oxidation of ammonia by the chemolithotrophic bacterium Nitrosomonas europaea. Biochim Biophys Acta 1459:346–355
Wiebe MG (2002) Siderophore production by Fusarium venenatum A3/5. Biochem Soc Trans 30:696–698
Winkelmann G (1991) Specificity of iron transport in bacteria and fungi. In: Winkelmann G (eds) CRC handbook of microbial iron chelates. CRC Press, Boca Raton, p 366
Yang CC, Leong J (1982) Production of deferriferrioxamines B and E from a ferroverdin-producing Streptomyces species. J Bacteriol 149:381–383
Acknowledgments
We thank Dr. B. Dubbels for his insightful discussions, B. Arbogast (Central Laboratory, OSU) for help with the HPLC/MS/MS analysis, and M. Nesson for help with electron microscopy (Electron Microscope Facility, Department of Botany and Plant Pathology, OSU). We appreciate the help of A. Ungerer (College of Oceanic and Atmospheric Sciences, OSU) for Fe determination by ICP-MS. This research was supported by grant DE-FG03-01ER63149 to DJA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wei, X., Vajrala, N., Hauser, L. et al. Iron nutrition and physiological responses to iron stress in Nitrosomonas europaea . Arch Microbiol 186, 107–118 (2006). https://doi.org/10.1007/s00203-006-0126-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-006-0126-4