Abstract
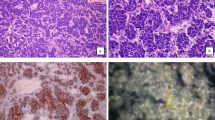
Gliosarcoma is a variant of glioblastoma and is characterized by distinct glial and sarcomatous components. Typically, there is no macroscopic boundary between the components and special stains are often required to distinguish the glial and sarcomatous elements. Some studies suggest similar genetic alterations in both components pointing to a common origin. We present an extreme case of gliosarcoma arising as a discrete fibrous nodule adjacent to a typical glioblastoma. A 65 year-old woman presented with progressive weakness, seizures and right-sided hemiparesis. CT scan demonstrated an irregular enhancing left frontal lobe mass and an adjacent discrete nodule with different imaging characteristics. The unique nature of this macroscopically biphasic neoplasm allowed us to compare the molecular characteristics of glial and sarcomatous elements which were strikingly similar except for small losses and gains in Chr 3. Studies are under way to determine the significance of chromosome 3 alterations in gliosarcomas.



Similar content being viewed by others
References
Feigin IH, Gross SW (1955) Sarcoma arising in glioblastoma of the brain. Am J Pathol 31:633–653
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114:97–109. doi:10.1007/s00401-007-0243-4
Lutterbach J, Guttenberger R, Pagenstecher A (2001) Gliosarcoma: a clinical study. Radiother Oncol 61:57–64
Galanis E, Buckner JC, Dinapoli RP, Scheithauer BW, Jenkins RB, Wang CH, O’Fallon JR, Farr G Jr (1998) Clinical outcome of gliosarcoma compared with glioblastoma multiforme: North Central Cancer Treatment Group results. J Neurosurg 89:425–430. doi:10.3171/jns.1998.89.3.0425
Monzon FA, Alvarez K, Gatalica Z, Bridge JA, Nelson M, Kim HJ, Hagenkord JM (2009) Detection of chromosomal aberrations in renal tumors: a comparative study of conventional cytogenetics and virtual karyotyping with single-nucleotide polymorphism microarrays. Arch Pathol Lab Med 133:1917–1922
Miller CR, Perry A (2007) Glioblastoma. Arch Pathol Lab Med 131:397–406
Meis JM, Ho KL, Nelson JS (1990) Gliosarcoma: a histologic and immunohistochemical reaffirmation. Mod Pathol 3:19–24
Jones H, Steart PV, Weller RO (1991) Spindle-cell glioblastoma or gliosarcoma? Neuropathol Appl Neurobiol 17:177–187
Actor B, Cobbers JM, Buschges R, Wolter M, Knobbe CB, Lichter P, Reifenberger G, Weber RG (2002) Comprehensive analysis of genomic alterations in gliosarcoma and its two tissue components. Genes Chromosomes Cancer 34:416–427. doi:10.1002/gcc.10087
Reis RM, Konu-Lebleblicioglu D, Lopes JM, Kleihues P, Ohgaki H (2000) Genetic profile of gliosarcomas. Am J Pathol 156:425–432
McComb RD, Jones TR, Pizzo SV, Bigner DD (1982) Localization of factor VIII/von Willebrand factor and glial fibrillary acidic protein in the hemangioblastoma: implications for stromal cell histogenesis. Acta Neuropathol 56:207–213
Kochi N, Budka H (1987) Contribution of histiocytic cells to sarcomatous development of the gliosarcoma. An immunohistochemical study. Acta Neuropathol 73:124–130
Grant JW, Steart PV, Aguzzi A, Jones DB, Gallagher PJ (1989) Gliosarcoma: an immunohistochemical study. Acta Neuropathol 79:305–309
Han SJ, Yang I, Tihan T, Chang SM, Parsa AT (2010) Secondary gliosarcoma: a review of clinical features and pathological diagnosis. J Neurosurg 112:26–32. doi:10.3171/2009.3.JNS081081
Biernat W, Aguzzi A, Sure U, Grant JW, Kleihues P, Hegi ME (1995) Identical mutations of the p53 tumor suppressor gene in the gliomatous and the sarcomatous components of gliosarcomas suggest a common origin from glial cells. J Neuropathol Exp Neurol 54:651–656
Boerman RH, Anderl K, Herath J, Borell T, Johnson N, Schaeffer-Klein J, Kirchhof A, Raap AK, Scheithauer BW, Jenkins RB (1996) The glial and mesenchymal elements of gliosarcomas share similar genetic alterations. J Neuropathol Exp Neurol 55:973–981
Creighton CJ, Chang JC, Rosen JM (2010) Epithelial-mesenchymal transition (EMT) in tumor-initiating cells and its clinical implications in breast cancer. J Mammary Gland Biol Neoplasia 15:253–260. doi:10.1007/s10911-010-9173-1
Taube JH, Herschkowitz JI, Komurov K, Zhou AY, Gupta S, Yang J, Hartwell K, Onder TT, Gupta PB, Evans KW, Hollier BG, Ram PT, Lander ES, Rosen JM, Weinberg RA, Mani SA (2010) Core epithelial-to-mesenchymal transition interactome gene-expression signature is associated with claudin-low and metaplastic breast cancer subtypes. Proc Natl Acad Sci USA 107:15449–15454. doi:10.1073/pnas.1004900107
Vincent T, Neve EP, Johnson JR, Kukalev A, Rojo F, Albanell J, Pietras K, Virtanen I, Philipson L, Leopold PL, Crystal RG, de Herreros AG, Moustakas A, Pettersson RF, Fuxe J (2009) A SNAIL1-SMAD3/4 transcriptional repressor complex promotes TGF-beta mediated epithelial-mesenchymal transition. Nat Cell Biol 11:943–950. doi:10.1038/ncb1905
Batlle E, Sancho E, Franci C, Dominguez D, Monfar M, Baulida J, Garcia De Herreros A (2000) The transcription factor snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nat Cell Biol 2:84–89. doi:10.1038/35000034
Nieto MA (2002) The snail superfamily of zinc-finger transcription factors. Nat Rev Mol Cell Biol 3:155–166. doi:10.1038/nrm757
Martinez-Estrada OM, Culleres A, Soriano FX, Peinado H, Bolos V, Martinez FO, Reina M, Cano A, Fabre M, Vilaro S (2006) The transcription factors Slug and Snail act as repressors of Claudin-1 expression in epithelial cells. Biochem J 394:449–457. doi:10.1042/BJ20050591
Ioannou M, Papagelopoulos PJ, Papanastassiou I, Iakovidou I, Kottakis S, Demertzis N (2008) Detection of somatostatin receptors in human osteosarcoma. World J Surg Oncol 6:99. doi:10.1186/1477-7819-6-99
Ferrari S, Dondi M, Fanti S, Zoboli S, Giacomini S, Mercuri M, Bacci G (2003) Somatostatin receptor (SSTR) scintigraphy in patients with osteosarcoma. Cancer Biother Radiopharm 18:847–851. doi:10.1089/108497803770418391
Ellis P, Fagan BM, Magness ST, Hutton S, Taranova O, Hayashi S, McMahon A, Rao M, Pevny L (2004) SOX2, a persistent marker for multipotential neural stem cells derived from embryonic stem cells, the embryo or the adult. Dev Neurosci 26:148–165. doi:10.1159/000082134
Han SJ, Yang I, Tihan T, Chang SM, Parsa AT (2010) Secondary gliosarcoma: a review of clinical features and pathological diagnosis. J Neurosurg 112:26–32. doi:10.3171/2009.3.JNS081081
Han SJ, Yang I, Ahn BJ, Otero JJ, Tihan T, McDermott MW, Berger MS, Prados MD, Parsa AT (2010) Clinical characteristics and outcomes for a modern series of primary gliosarcoma patients. Cancer 116:1358–1366. doi:10.1002/cncr.24857
Acknowledgment
We are deeply grateful to Dr. Shera Kash, PhD from the Molecular Pathology and Clinical Genomics Division of the Department of Pathology and Creighton Medical Labs for her expert contribution and the analysis of the 250 K Affymetrix NspI single nucleotide polymorphism (SNP) arrays.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jimenez, C., Powers, M., Parsa, A.T. et al. Sarcoma arising as a distinct nodule within glioblastoma: a morphological and molecular perspective on gliosarcoma. J Neurooncol 105, 317–323 (2011). https://doi.org/10.1007/s11060-011-0593-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-011-0593-6