Abstract
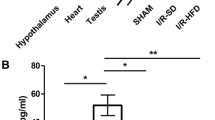
Nesfatin-1 is an anorexic nucleobindin-2 (NUCB2)-derived hypothalamic peptide. It controls feeding behavior, water intake, and glucose homeostasis. If intracerebrally administered, it induces hypertension, thus suggesting a role in central cardiovascular control. However, it is not known whether it is able to directly control heart performance. We aimed to verify the hypothesis that, as in the case of other hypothalamic satiety peptides, Nesfatin-1 acts as a peripheral cardiac modulator. By western blotting and QT-PCR, we identified the presence of both Nesfatin-1 protein and NUCB2 mRNA in rat cardiac extracts. On isolated and Langendorff-perfused rat heart preparations, we found that exogenous Nesfatin-1 depresses contractility and relaxation without affecting coronary motility. These effects did not involve Nitric oxide, but recruited the particulate guanylate cyclase (pGC) known as natriuretic peptide receptor A (NPR-A), protein kinase G (PKG) and extracellular signal-regulated kinases1/2 (ERK1/2). Co-immunoprecipitation and bioinformatic analyses supported an interaction between Nesfatin-1 and NPR-A. Lastly, we preliminarily observed, through post-conditioning experiments, that Nesfatin-1 protects against ischemia/reperfusion (I/R) injury by reducing infarct size, lactate dehydrogenase release, and postischemic contracture. This protection involves multiple prosurvival kinases such as PKCε, ERK1/2, signal transducer and activator of transcription 3, and mitochondrial KATP channels. It also ameliorates contractility recovery. Our data indicate that: (1) the heart expresses Nesfatin-1, (2) Nesfatin-1 directly affects myocardial performance, possibly involving pGC-linked NPR-A, the pGC/PKG pathway, and ERK1/2, (3) the peptide protects the heart against I/R injury. Results pave the way to include Nesfatin-1 in the neuroendocrine modulators of the cardiac function, also encouraging the clarification of its clinical potential in the presence of nutrition-dependent physio-pathologic cardiovascular diseases.








Similar content being viewed by others
Abbreviations
- 5-HD:
-
5-Hydroxydecanoate
- ARC:
-
Arcuate nuclei
- CP:
-
Coronary pressure
- eNOS:
-
Endothelial nitric oxide synthase
- ERK1/2:
-
Extracellular signal-regulated kinases1/2
- I/R:
-
Ischemia/reperfusion injury
- −(LVdP/dt)max :
-
Maximal rate of left ventricular pressure decline of LVP
- +(LVdP/dt)max :
-
Maximal values of the first derivative of LVP
- LVP:
-
Left ventricular pressure
- NO:
-
Nitric oxide
- NPR-A:
-
Natriuretic peptide receptor A
- NPY/AgRP:
-
Neuropeptide Y/agouti-related peptide
- NUCB2:
-
Nucleobindin-2
- pGC:
-
Particulate guanylate cyclase
- POMC/CART:
-
Pro-opiomelanocortin/cocaine and amphetamine responsive transcript
- PostC:
-
Post-conditioning protocol
- PVN:
-
Paraventricular nuclei
- SNO:
-
S-nitrosylation
- SON:
-
Supraoptic nuclei
- STAT3:
-
Signal transducer and activator of transcription 3
References
Angelone T, Quintieri AM, Amodio N, Cerra MC (2011) Endocrine orchestration of cardiovascular, gastrointestinal and hypothalamic control. Curr Med Chem 18(32):4976–4986
Vona-Davis LC, McFadden DW (2007) NPY family of hormones: clinical relevance and potential use in gastrointestinal disease. Curr Top Med Chem 7(17):1710–1720
Pedrazzini T, Seydoux J, Künstner P, Aubert JF, Grouzmann E, Beermann F, Brunner HR (1998) Cardiovascular response, feeding behavior and locomotor activity in mice lacking the NPY Y1 receptor. Nat Med 4(6):722–726
Oh-I S, Shimizu H, Satoh T, Okada S, Adachi S, Inoue K, Eguchi H, Yamamoto M, Imaki T, Hashimoto K, Tsuchiya T, Monden T, Horiguchi K, Yamada M, Mori M (2006) Identification of nesfatin-1 as a satiety molecule in the hypothalamus. Nature 443(7112):709–712
Elmquist JK, Coppari R, Balthasar N, Ichinose M, Lowell BB (2005) Identifying hypothalamic pathways controlling food intake, body weight, and glucose homeostasis. J Comp Neurol 493(1):63–71
Stengel A, Goebel M, Yakubov I, Wang L, Witcher D, Coskun T, Taché Y, Sachs G, Lambrecht NW (2009) Identification and characterization of nesfatin-1 immunoreactivity in endocrine cell types of the rat gastric oxyntic mucosa. Endocrinology 150(1):232–238
Su Y, Zhang J, Tang Y, Bi F, Liu JN (2010) The novel function of nesfatin-1: anti-hyperglycemia. Biochem Biophys Res Commun 391(1):1039–1042
Price TO, Samson WK, Niehoff ML, Banks WA (2007) Permeability of the blood-brain barrier to a novel satiety molecule nesfatin-1. Peptides 28(12):2372–2381
Brailoiu GC, Dun SL, Brailoiu E, Inan S, Yang J, Chang JK, Dun NJ (2007) Nesfatin-1: distribution and interaction with a G protein-coupled receptor in the rat brain. Endocrinology 148(10):5088–5094
Pałasz A, Krzystanek M, Worthington J, Czajkowska B, Kostro K, Wiaderkiewicz R, Bajor G (2012) Nesfatin-1, a unique regulatory neuropeptide of the brain. Neuropeptides http://dx.doi.org/10.1016/j.npep.2011.12.002
Maejima Y, Sedbazar U, Suyama S, Kohno D, Onaka T, Takano E, Yoshida N, Koike M, Uchiyama Y, Fujiwara K, Yashiro T, Horvath TL, Dietrich MO, Tanaka S, Dezaki K, Oh-I S, Hashimoto K, Shimizu H, Nakata M, Mori M, Yada T (2009) Nesfatin-1-regulated oxytocinergic signaling in the paraventricular nucleus causes anorexia through a leptin-independent melanocortin pathway. Cell Metab 10(5):355–365
Mimee A, Smith PM, Ferguson AV (2012) Nesfatin-1 influences the excitability of neurons in the nucleus of the solitary tract and regulates cardiovascular function. Am J Physiol Regul Integr Comp Physiol. doi:10.1152/ajpregu.00266.2011
Yosten GL, Samson WK (2009) Nesfatin-1 exerts cardiovascular actions in brain: possible interaction with the central melanocortin system. Am J Physiol Regul Integr Comp Physiol 297(2):R330–R336
Cerra MC, De Iuri L, Angelone T, Corti A, Tota B (2006) Recombinant N-terminal fragments of chromogranin-A modulate cardiac function of the Langendorff-perfused rat heart. Basic Res Cardiol 101(1):43–52
Shimizu H, Oh-I S, Hashimoto K, Nakata M, Yamamoto S, Yoshida N, Eguchi H, Kato I, Inoue K, Satoh T, Okada S, Yamada M, Yada T, Mori M (2009) Peripheral administration of nesfatin-1 reduces food intake in mice: the leptin-independent mechanism. Endocrinology 150(2):662–671
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(–C(T)) method. Methods 25:402–408
Cerra MC, Angelone T, Parisella ML, Pellegrino D, Tota B (2009) Nitrite modulates contractility of teleost (Anguilla anguilla and Chionodraco hamatus, i.e. the Antarctic hemoglobinless icefish) and frog (Rana esculenta) hearts. Biochim Biophys Acta 1787(7):849–855
Angelone T, Goumon Y, Cerra MC, Metz-Boutigue MH, Aunis D, Tota B (2006) The emerging cardioinhibitory role of the hippocampal cholinergic neurostimulating peptide. J Pharmacol Exp Ther 318(1):336–344
Pagliaro P, Mancardi D, Rastaldo R, Penna C, Gattullo D, Miranda KM, Feelisch M, Wink DA, Kass DA, Paolocci N (2003) Nitroxyl affords thiol-sensitive myocardial protective effects akin to early preconditioning. Free Radic Biol Med 34(1):33–43
Penna C, Mancardi D, Rastaldo R, Losano G, Pagliaro P (2007) Intermittent activation of bradykinin B2 receptors and mitochondrial KATP channels trigger cardiac postconditioning through redox signaling. Cardiovasc Res 75(1):168–177
Penna C, Rastaldo R, Mancardi D, Raimondo S, Cappello S, Gattullo D, Losano G, Pagliaro P (2006) Post-conditioning induced cardioprotection requires signaling through a redox-sensitive mechanism, mitochondrial ATP-sensitive K+ channel and protein kinase C activation. Basic Res Cardiol 101(2):180–189
Parat M, McNicoll N, Wilkes B, Fournier A, De Léan A (2008) Role of extracellular domain dimerization in agonist-induced activation of natriuretic peptide receptor A. Mol Pharmacol 73(2):431–440
Springer J, Azer J, Hua R, Robbins C, Adamczyk A, McBoyle S, Bissell MB, Rose RA (2012) The natriuretic peptides BNP and CNP increase heart rate and electrical conduction by stimulating ionic currents in the sinoatrial node and atrial myocardium following activation of guanylyl cyclase-linked natriuretic peptide receptors. J Mol Cell Cardiol http://dx.doi.org/10.1016/j.yjmcc.2012.01.018
Yamawaki H, Takahashi M, Mukohda M, Morita T, Okada M, Hara Y (2012) A novel adipocytokine, nesfatin-1 modulates peripheral arterial contractility and blood pressure in rats. Biochem Biophys Res Commun 418(4):676–681
Angelone T, Filice E, Quintieri AM, Imbrogno S, Amodio N, Pasqua T, Pellegrino D, Mulè F, Cerra MC (2010) Receptor identification and physiological characterisation of glucagon-like peptide-2 in the rat heart. Nutr Metab Cardiovasc Dis http://dx.doi.org/10.1016/j.numecd.2010.07.014
Yosten GL, Redlinger L, Samson WK (2012) Evidence for a role of endogenous nesfatin-1 in the control of water drinking. J Neuroendocrinol. doi:10.1111/j.1365-2826.2012.02304.x
Mohan H, Unniappan S (2012) Ontogenic pattern of nucleobindin-2/nesfatin-1 expression in the gastroenteropancreatic tissues and serum of Sprague Dawley rats. Regul Pept 175(1–3):61–69
Opie LH (2004) Heart physiology. From cell to circulation. 4th edn. Lippincott, Philadelphia
Sperelakis N, Kurachi Y, Terzic A, Cohen MV (2001) Heart physiology and pathophysiology, 4th edn. Academic, San Diego
Piascik MT, Perez DM (2001) Alpha1-adrenergic receptors: new insights and directions. J Pharmacol Exp Ther 298(2):403–410
Abi-Gerges N, Fischmeister R, Méry PF (2001) G protein-mediated inhibitory effect of a nitric oxide donor on the L-type Ca2+ current in rat ventricular myocytes. J Physiol 531(Pt 1):117–130
Lin X, Hänze J, Heese F, Sodmann R, Rudolf E (1995) Lang gene expression of natriuretic peptide receptors in myocardial cells. Circ Res 77:750–758
Potter LR, Abbey-Hosch S, Dickey DM (2006) Natriuretic peptides, their receptors, and cyclic guanosine monophosphate-dependent signaling functions. Endocr Rev 27(1):47–72
Dube′ PE, Forse CL, Bahrami J, Brubaker PL (2006) The essential role of insulin-like growth factor-1 in the intestinal tropic effects of glucagon-like peptide-2 in mice. Gastroenterology 131:589–605
Clerk A, Sugden PH (2004) Signaling through the extracellular signal regulated kinase 1/2 cascade in cardiac myocytes. Biochem Cell Biol 82(6):603–609
Darling CE, Jiang R, Maynard M, Whittaker P, Vinten-Johansen J, Przyklenk K (2005) Postconditioning via stuttering reperfusion limits myocardial infarct size in rabbit hearts: role of ERK1/2. Am J Physiol Heart Circ Physiol 289(4):H1618–H1626
Cappello S, Angelone T, Tota B, Pagliaro P, Penna C, Rastaldo R, Corti A, Losano G, Cerra MC (2007) Human recombinant chromogranin A-derived vasostatin-1 mimics preconditioning via an adenosine/nitric oxide signaling mechanism. Am J Physiol Heart Circ Physiol 293(1):H719–H727
Penna C, Alloatti G, Gallo MP, Cerra MC, Levi R, Tullio F, Bassino E, Dolgetta S, Mahata SK, Tota B, Pagliaro P (2010) Catestatin improves post-ischemic left ventricular function and decreases ischemia/reperfusion injury in heart. Cell Mol Neurobiol 30(8):1171–1179
Penna C, Abbadessa G, Mancardi D, Tullio F, Piccione F, Spaccamiglio A, Racca S, Pagliaro P (2008) Synergistic effects against post-ischemic cardiac dysfunction by sub-chronic nandrolone pretreatment and postconditioning: role of beta2-adrenoceptor. J Physiol Pharmacol 59(4):645–659
Pagliaro P, Moro F, Tullio F, Perrelli MG, Penna C (2011) Cardioprotective pathways during reperfusion: focus on redox signaling and other modalities of cell signaling. Antioxid Redox Signal 14(5):833–850
Hausenloy DJ, Lecour S, Yellon DM (2011) Reperfusion injury salvage kinase and survivor activating factor enhancement prosurvival signaling pathways in ischemic postconditioning: two sides of the same coin. Antioxid Redox Signal 14(5):893–907
Nishikimi T, Maeda N, Matsuoka H (2006) The role of natriuretic peptides in cardioprotection. Cardiovasc Res 9(2):318–328
Bolli R, Dawn B, Xuan YT (2003) Role of the JAK-STAT pathway in protection against myocardial ischemia/reperfusion injury. Trends Cardiovasc Med 13:72–79
Fuglesteg BN, Suleman N, Tiron C, Kanhema T, Lacerda L, Andreasen TV, Sack MN, Jonassen AK, Mjøs OD, Opie LH, Lecour S (2008) Signal transducer and activator of transcription 3 is involved in the cardioprotective signalling pathway activated by insulin therapy at reperfusion. Basic Res Cardiol 103:444–453
Mascareno E, El-Shafei M, Maulik N, Sato M, Guo Y, Das DK, Siddiqui MA (2001) JAK/STAT signaling is associated with cardiac dysfunction during ischemia and reperfusion. Circulation 104:325–329
Boengler K, Hilfiker-Kleiner D, Heusch G, Schulz R (2010) Inhibition of permeability transition pore opening by mitochondrial STAT3 and its role in myocardial ischemia/reperfusion. Basic Res Cardiol 105:771–785
Xuan YT, Guo Y, Han H, Zhu Y, Bolli R (2001) An essential role of the JAK-STAT pathway in ischemic preconditioning. Proc Natl Acad Sci USA 98:9050–9055
Hausenloy DJ, Yellon DM (2004) New directions for protecting the heart against ischaemia-reperfusion injury: targeting the reperfusion injury salvage kinase (RISK)-pathway. Cardiovasc Res 61:448–460
Penna C, Abbadessa G, Mancardi D, Spaccamiglio A, Racca S, Pagliaro P (2007) Nandrolone-pretreatment enhances cardiac beta(2)-adrenoceptor expression and reverses heart contractile down-regulation in the post-stress period of acute-stressed rats. J Steroid Biochem Mol Biol 107(1–2):106–113
Acknowledgments
This research was supported by grants from Ministero dell’Università e Ricerca Scientifica e Tecnologica (ex 60 % T.A. and M.C.C.), “Progetto Giovani Ricercatori 2010.” (E.F.) and National Institute of Cardiovascular Research (INRC).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
T. Angelone and E. Filice contributed equally to the work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Angelone, T., Filice, E., Pasqua, T. et al. Nesfatin-1 as a novel cardiac peptide: identification, functional characterization, and protection against ischemia/reperfusion injury. Cell. Mol. Life Sci. 70, 495–509 (2013). https://doi.org/10.1007/s00018-012-1138-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-012-1138-7