Abstract
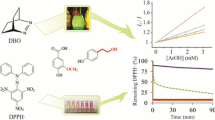
Antioxidant capacity of N-(1-naphthyl)valerohydroxamic acid (NVHA) and N-(1-naphthyl)phenylacetohydroxamic acid (NPAHA) has been evaluated by a novel approach employing the fluorescence microscopic single molecule observation method. This method allows direct observation of the changes in single DNA molecules. The DNA cleavage protection activity of the compounds was also assessed by the gel electrophoresis method. The applied methods confirmed that both compounds are capable of inhibiting the free radical mediated DNA damage. Free radical scavenging activity was assessed via the 2,2′-diphenyl-1-picrylhydrazyl free radical (DPPH) and lipid peroxidation inhibition methods. The effective concentration causing a 50 % inhibition of the DPPH concentration, EC50, was found to be 371.54 mM for NVHA and 365.95 mM for NPAHA. Its lipid peroxidation inhibition ability was calculated to be 40.91 % at 371.54 mM for NVHA and 41.14 % at 365.95 mM for NPAHA. These results show the antioxidant potential of the naphthyl hydroxamic acids.
Similar content being viewed by others
References
Alam, M. N., Bristi, N. J., & Rafiquzzaman, M. (2013). Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharmaceutical Journal, 21, 143–152. DOI: 10.1016/j.jsps.2012.05.002.
Ames, B. N., Shigena, M. K., & Hagen, T. M. (1993). Oxidants, antioxidants and the degenerative diseases of aging. The Proceedings of the National Academy of Sciences USA, 90, 7915–7922.
Aruoma, O. I. (1996). Assessment of potential prooxidant and antioxidant actions. Journal of the American Oil Chemists’ Society, 73, 1617–1625. DOI: 10.1007/bf02517962.
Aruoma, O. I., & Cuppett, S. L. (1997). Antioxidant methodology: In vivo and in vitro concepts (pp. 39–51). Champain, IL, USA: AOCS Press.
Autore, G., Caruso, A., Marzocco, S., Nicolaus, B., Palladino, C., Pinto, A., Popolo, A., Sinicropi, M. S., Tommonaro, G., & Saturnino, C. (2010). Acetamide derivatives with antioxidant activity and potential anti-inflammatory activity. Molecules, 15, 2028–2038. DOI: 10.3390/molecules15032028.
Behl, C., Skutella, T., Lezoualc’h, F., Post, A., Widmann, M., Newton, C. J., & Holsboer, F. (1997). Neuroprotection against oxidative stress by estrogens: Structure-activity relationship. Molecular Pharmacology, 51, 535–541. DOI: 10.1124/mol.51.4.535.
Broderick, C. E., & Cooke, P. H. (2009). Fruit composition, tissues and localization of antioxidants and capsaicinoids in Capsicum peppers by fluorescence microscopy. Acta Horticulturae, 841, 85–90.
Gupta, V. K., & Tandon, S. G. (1972). Hydroxamic acids. Preparation and properties of N-1-napthylhydroxamic acids. Journal of Chemical & Engineering Data, 17, 248–249. DOI: 10.1021/je60053a008.
Harman, D. (1995). Role of antioxidant nutrients in aging: Overview. Age, 18, 51–62. DOI: 10.1007/bf02432519.
Houseal, T. W., Bustamante, C., Stump, R. F., & Maestre, M. F. (1989). Real-time imaging of single DNA molecules with fluorescence microscopy. Biophysical Journal, 56, 507–516. DOI: 10.1016/s0006-3495(89)82697-9.
Hyogo, A., Kobayashi, T., del Saz, E. G., & Seguchi, H. (2010). Antioxidant effects of protocatechuic acid, ferulic acid and caffeic acid in human neutrophils using a fluorescent substance. International Journal of Morphology, 28, 911–920. DOI: 10.4067/s0717-95022010000300040.
Khare, D., Verma, B., & Pande, R. (2012). Antioxidant activity of N -phenylbenzohydroxamic acid. Asian Journal of Pharmaceutical and Clinical Research, 5, 121–124.
Končić, M., Rajić, Z., Petrić, N., & Zorc, B. (2009). Antioxidant activity of NSAID hydroxamic acids. Acta Pharmaceutica, 59, 235–242. DOI: 10.2478/v10007-009-0017-8.
Nakamura, K., Fushimi, K., Kouchi, H., Mihara, K., Miyazaki, M., Ohe, T., & Namba, M. (1998). Inhibitory effects of antioxidants on neonatal rat cardiac myocyte hypertrophy induced by tumor necrosis factor-α and angiotensin II. Circulation, 98, 794–799. DOI: 10.1161/01.cir.98.8.794.
Nie, S., & Zare, R. N. (1997). Optical detection of single molecules. Annuual Review on Biophysics and Biomolecular Structure, 26, 567–596. DOI: 10.1146/annurev.biophys.26.1.567.
Oana, H., Tsumoto, K., Yoshikawa, Y., & Yoshikawa, K. (2002). Folding transition of large DNA completely inhibits the action of a restriction endonuclease as revealed by single-chain observation. FEBS Letters, 530, 143–146. DOI: 10.1016/s0014-5793(02)03448-8.
Patre, S., Thakur, P., & Pande, R. (2011). Solubility and hydrophobic descriptors of N-arylhydroxamic acids. International Journal of Bioscience, Biochemistry and Bioinformatics, 1, 267–270. DOI: 10.7763/ijbbb.2011.v1.50.
Rajwade, R. P., Pande, R., Mishra, K. P., Kumar, A., & Pandey, B. N. (2008). Quantitative structure — activity relationship (QSAR) of N-arylsubstituted hydroxamic acids as inhibitors of human adenocarcinoma cells A431. Medicinal Chemistry, 4, 237–243. DOI: 10.2174/157340608784325106.
Rajwade, R. P., Pande, R., Mishra, K. P., Kumar, A., & Pandey, B. N. (2009). Hydroxamic acids analogous against breast cancer cells: 2D-QSAR and 3D-QSAR stud ies. QSAR & Combinatorial Sciences, 28, 1500–1508. DOI: 10.1002/qsar.200910023.
Ramadas, D., & Leela, S. (2011). Antioxidant effects of 28 kDa antioxidant protein from turmeric (Curcuma longa L). Asian Journal of Pharmaceutical and Clinical Research, 4, 112–118.
Rigler, R., Mets, Ü., Widengren, J., & Kask, P. (1993). Fluorescence correlation spectroscopy with high count rate and low background: Analysis of translational diffusion. European Biophysics Journal, 22, 169–175. DOI: 10.1007/bf00185777.
Sautin, Y. Y., Nakagawa, T., Zharikov, S., & Johnson, R. J. (2007). Adverse effects of the classic antioxidant uric acid in adipocytes: NADPH oxidase-mediated oxidative/nitrosative stress. AJP: Cell Physiology, 293, C584–C596. DOI: 10.1152/ajpcell.00600.2006.
Sato, Y. T., Hamada, T., Kubo, K., Yamada, A., Kishida, T., Mazda, O., & Yoshikawa, K. (2005). Folding transition into a loosely collapsed state in plasmid DNA as revealed by single-molecule observation. FEBS Letters, 579, 3095–3099. DOI: 10.1016/j.febslet.2005.04.072.
Sharma, U. S., & Kumar, A. (2011). In vitro antioxidant activity of Rubus ellipticus fruits. Journal of Advanced Pharmaceutical Technology & Research, 2, 47–50. DOI: 10.4103/2231-4040.79805.
Shirinzadeh, H., Eren, B., Gurer-Orhan, H., Suzen, S., & Özden, S. (2010). Novel indole-based analogs of melatonin: Synthesis and in vitro antioxidant activity studies. Molecules, 15, 2187–2202. DOI: 10.3390/molecules15042187.
Tiwari, V., & Pande, R. (2006). Molecular descriptors of Narylhydroxamic acids: A tool in drug design. Chemical Biology & Drug Design, 68, 225–228. DOI: 10.1111/j.1747-0285.2006.00433.x.
Valko, M., Rhodes, C. J., Moncol, J., Izakovic, M., & Mazur, M. (2006). Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chemico-Biological Interactions, 160, 1–40. DOI: 10.1016/j.cbi.2005.12.009.
Valko, M., Leibfritz, D., Moncol, J., Cronin, M. T. D., Mazur, M., & Telser, J. (2007). Free radicals and antioxidants in normal physiological functions and human disease. The International Journal of Biochemistry & Cell Biology, 39, 44–84. DOI: 10.1016/j.biocel.2006.07.001.
Wu, Y., Sun, F. F., Tong, D. M., & Taylor, B. M. (1996). Changes in membrane properties during energy depletioninduced cell injury studied with fluorescence microscopy. Biophysical Journal, 71, 91–100. DOI: 10.1016/s0006-3495(96)79243-3.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, P., Khare, D. & Pande, R. Evaluation of antioxidant activity and DNA cleavage protection effect of naphthyl hydroxamic acid derivatives through conventional and fluorescence microscopic methods. Chem. Pap. 68, 1298–1304 (2014). https://doi.org/10.2478/s11696-014-0576-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-014-0576-0