Abstract
Background
Consumption of refined carbohydrates has risen in recent years alongside chronic diseases such as type 2 diabetes mellitus, dyslipidemia, obesity, and non-alcoholic fatty liver disease (NAFLD). Fructose is a monosaccharide made widely available in industrialized products, capable of inducing excessive weight gain and liver steatosis in animal models, while omega-3 fatty acids, present in foods such as fatty fish and fish oil, have shown to inhibit genes related to lipogenesis and decrease cardiovascular risk. Therefore, our objective was to evaluate the impact of a high-fructose diet on weight gain, biochemical and oxidative stress parameters, and liver histology and investigate fish oil’s potential protective role. Thirty male Wistar rats were divided into 3 groups: regular chow diet (CT), regular chow diet plus 20% fructose in drinking water (Fr), and a diet containing 10% fish oil plus 20% fructose in drinking water (FOFr). After 12 weeks, tissues of interest were collected for biochemical and histological analyses.
Results
Although fructose consumption did not lead to increased hepatic fat, it caused a significant increase in weight gain, white adipose tissue, and serum triglycerides in the Fr group, while fish oil promoted normalized serum triglycerides and even reduced adiposity in the FOFr group. Additionally, the inclusion of fish oil in the FOFr diet led to increased liver lipid peroxidation in the form of increased hepatic MDA.
Conclusions
It is concluded that fish oil can prevent important metabolic alterations caused by fructose consumption, but its dosage must be taken into account to prevent oxidative stress and potential liver damage.
Similar content being viewed by others
Background
The alarming increase in obesity prevalence around the world is a public health concern [1]. This pathological expansion of adipose tissue is accompanied by an inflammatory state and many comorbidities, such as type 2 diabetes (T2D), dyslipidemia, cardiovascular diseases (CVD), and non-alcoholic fatty liver disease (NAFLD) [2]. The latter is currently considered the most prevalent liver disease in the world, characterized by a higher than 5% fat deposition in the liver of individuals with no history of excessive alcohol intake. It is believed NAFLD progression occurs initially through increased lipid accumulation in the liver (the first “hit”), followed by oxidative stress and an increase in lipid peroxidation (the second “hit”) [3] leading to its inflammatory form, non-alcoholic steatohepatitis (NASH), and possibly more serious liver diseases such as cirrhosis and hepatocellular carcinoma [4].
Oxidative stress is a condition in which unstable molecules known as reactive oxygen species (ROS) overwhelm antioxidant defenses, leading to potential damage of cellular constituents such as proteins, lipids, and DNA. ROS are endogenously produced by the mitochondrial respiratory chain [5], as such, an increase in presence and oxidation of glucose and free fatty acids (FFA) can lead to increased generation of electron donors, resulting in ROS overproduction [6]. Furthermore, oxidative stress has been recognized as a contributor to the development of hypertension, inflammation, insulin resistance, and other chronic diseases [7].
A sedentary lifestyle and excessive energy intake, favored by the consumption of simple carbohydrates and energy-dense foods are major causes of obesity and NAFLD [8]. More specifically, fructose is a monosaccharide naturally present in fruits but made excessively available in industrialized products, and its role in the development of obesity and related comorbidities has been highly studied. After its intestinal absorption, fructose is rapidly transported into the liver via hepatic GLUT2 and is able to bypass important rate-limiting steps of glycolysis, therefore leading to an increase in triose phosphate intermediates at a faster rate than glucose, which can then be utilized in different pathways, including lipogenesis [9]. Additionally, fructose is able to interact with transcription factors such as SREBP1c and ChREBP, which are responsible for the expression of enzymes related to fatty acid synthesis and glycolysis [10, 11].
Fatty acids, another component of human diets, have been shown to interact with transcription factors and modulate lipogenesis [12]. Fish oil, a compost rich in marine omega-3 (n-3) polyunsaturated fatty acids (PUFAs), is known to have anti-inflammatory properties, as well as being able to reduce hepatic triglycerides (TG), fatty acid synthesis, and adipose cell size [3, 13]. On the other hand, excessive incorporation of n-3 fatty acids into cell membranes raises its peroxidizability index due to an increase in sites susceptible to lipid peroxidation, which can result in membrane glycolipid, phospholipid, and cholesterol peroxidation [14, 15].
Therefore, considering the important increase in the consumption of industrialized products and in the prevalence of obesity and its related comorbidities, the aim of this study was to evaluate the effects of a marine n-3 fatty acid-rich diet in fructose-fed rats in terms of liver, biochemical, and oxidative stress parameters.
Methods
Animals and diet
All procedures were approved by the Ribeirão Preto Medical School’s Ethics Committee of Animal Experimentation (protocol number 185/2017). Thirty male Wistar rats were obtained from the Central Animal House of Ribeirão Preto Campus, University of São Paulo, Brazil, and kept in a 12-h light/12-h dark cycle, at an average temperature of 22 °C. Animals weighed an average of 200 g upon arrival and were randomly assigned to 3 groups (n = 10). The control group (CT) was fed a regular chow diet (Nuvilab Cr-1®, Nuvital Nutrientes S/A, Table 1) and tap water. The fructose (Fr) group was fed a regular chow diet and water containing 20% fructose. The fish oil-fructose group (FOFr) also received water containing 20% fructose but had its diet altered to contain 10% fish oil. Fish oil was acquired from Campestre Industry and Trade of Vegetable Oils (São Bernardo do Campo, Brazil).
Animals were weighed 3 times a week while also receiving fresh chow and water. Diet and water intake were also recorded every 2 days. After 12 weeks, animals were euthanized through decapitation following a 12-h fast. Blood was immediately collected and then centrifuged at 4 °C, and serum was stored at − 80 °C. Liver tissue was removed, weighed, and sampled for histological, oxidative stress, and total fat analyses and later stored at − 80 °C. Retroperitoneal and epididymal fat tissues were also collected and weighed.
Hepatic histology
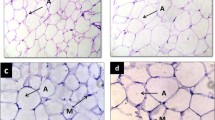
For histological analysis, hepatic tissues were dissected, fixed in 10% neutral buffered formalin, and embedded in paraffin according to standard procedures. Tissue sections of 5 μm thickness were stained with hematoxylin and eosin (H&E). Histological images were captured using the Scanscope (Olympus BX61VS).
Biochemical analyses
Fish oil’s fatty acids were determined using gas chromatography (Shimadzu, Europe, Duisburg, Germany), fitted with an SP2560 column (100 m, 0.25 mm, 0.20 μm). Helium was used as the carrier gas (1.6 ml/min flux), and a mix of methyl esters fatty acids were utilized as a standard (Supelco 37 Component FAME Mix).
Total fat was quantified through Bligh and Dyer’s [16] method utilizing 500 mg samples of hepatic tissue. Lipid peroxidation was assessed through the quantification of serum and hepatic malondialdehyde (MDA) following Gérard-Monnier et al.’s proposed method [17]. Reduced glutathione (GSH) was determined though Sedlak and Lindsay’s [18] method. Hepatic α-tocopherol concentration was measured according to the method proposed by Arnaud et al. [19] utilizing a high-performance liquid chromatograph (Shimadzu Europe, Duisburg, Germany) equipped with a C-18 column (Shimpack CLC-ODS 4.6 × 25 cm) and a 4 mm × 1 cm precolumn. The mobile phase was made by acetonitrile/dichloromethane/methanol (70/20/10) solution. Transaminases alanine aminotransferase (ALT) and aspartate aminotransferase (AST), total serum cholesterol, and serum TG were determined through commercial kits (Labtest Diagnóstica S.A., Brazil). Free fatty acids (FFAs) were determined through a quantitative colorimetric assay (BioAssay Systems, Hayward, CA, USA).
Statistical analysis
Data was subjected to ANOVA variance analysis with Tukey’s post hoc test (GraphPad Software, San Diego, CA, USA), with a significance level set at p < 0.05. Data is presented as means ± standard deviation.
Results
Table 2 shows the fatty acid composition of the fish oil utilized in the experiment, presenting a higher amount of n-3 fatty acids compared to n-6 fatty acids, and a significant amount of EPA and DHA, therefore demonstrating the FOFr diet indeed had an increased marine n-3 fatty acid content.
Table 3 shows consumption data regarding food, fluid (and therefore, fructose), and calorie intake. Food intake was similarly reduced in groups consuming fructose (Fr and FOFr). While fluid intake remained the same between CT and Fr, it was reduced in the FOFr group, leading to a subtle, albeit significant reduction in fructose intake compared to the Fr group. Curiously, total calorie intake was the same between experimental groups.
Figure 1 shows the initial and final body weight. Initially, body weight was the same between experimental groups. After 12 weeks, animals from the Fr showed significantly increased body weight, and FOFr had a similar weight to CT.
Figure 2 presents data regarding body composition. Retroperitoneal fat was significantly increased in the Fr group, and similar between CT and FOFr, while epididymal fat was reduced only in the FOFr group. When combined, total body fat was significantly lower in the FOFr group and higher in the Fr group. Additionally, liver weight corrected according to animal weight did not differ between animals.
Body composition. a Retroperitoneal fat, b epididymal fat, c sum of adipose tissues, and d liver weight corrected by animal weight. Values are mean ± SD. Asterisk indicates a significant difference when compared to CT and FOFr. Double asterisk indicates a significant difference when compared to CT and Fr (P < 0.05)
As for biochemical parameters (Fig. 3), serum TG was found to be elevated in the Fr group and normalized in the FOFr group. Additionally, total serum cholesterol was lower in the group consuming fish oil. No significant differences were seen in total hepatic fat, free fatty acids, or in the hepatic enzymes AST and ALT.
Biochemical parameters. a Serum triglycerides, b serum total cholesterol, c total hepatic fat, d free fatty acids, and e serum ALT and f AST. Values are mean ± SD. Asterisk indicates a significant difference when compared to CT and FOFr. Double asterisk indicates a significant difference when compared to CT and Fr (P < 0.05)
Figure 4 shows data regarding oxidative stress parameters. While serum MDA and GSH were not significantly different between groups, there was an important increase in hepatic MDA in the FOFr group. Hepatic GSH was also found to be significantly increased, but only in the Fr group. Additionally, hepatic α-tocopherol was found to be significantly reduced in FOFr animals. Finally, Fig. 5 displays H&E staining of liver samples, showing no significant difference between groups.
Discussion
In the present study, fructose consumption in water led to lower diet intake, causing caloric intake to be very similar between experimental groups. Indeed, previous research has shown that animals are capable of altering their food intake to maintain similar energy intake when consuming fructose in water [20, 21], although not always being sufficient to compensate for the additional calories from fructose [22, 23]. Equal energy ingestion may suggest that differences in weight gain and biochemical parameters seen in this study were due to fructose and fish oil’s significant effects on lipid metabolism and not due to a more energy-dense diet. Also, water intake remained the same between CT and Fr groups, and decreased slightly in the FOFr group, leading to marginally smaller fructose intake. In similar studies, animals receiving water in fructose tend to have increased water intake, which in turn leads to a higher daily fructose intake [20, 21]. It is unclear why water intake did not increase in our experiment; however, that may have protected animals from developing more serious metabolic complications, such as liver steatosis. Decreased fructose intake in the FOFr group presents a limitation of our study; however, since this decrease was subtle, fish oil can still be partially implicated in the positive changes seen in FOFr animals, such as normalized serum TGs and reduced adiposity. It may be the case that diets that are rich in lipids, such as the FOFr, lead to higher satiety and therefore reduced water intake, as seen in experiments offering high-fat diets to animals [24].
Fructose ingestion led to increased weight gain, mainly in terms of retroperitoneal fat, while fish oil prevented these changes and further reduced serum cholesterol and epididymal fat. Although fat deposition stems mainly from positive energy balance, fructose is known to have an important impact on lipid synthesis. Since fructose bypasses rate-limiting steps of glycolysis, it is rapidly metabolized in the liver into substrates that can be used for acetyl-CoA production and eventually for lipogenesis. Fructose-derived substrates also activate ChREBP, which further increases metabolic pathways such as glycolysis and fructolysis. Additionally, by inducing hyperinsulinemia [25], fructose promotes SREBP1-c activation [26], leading to increased expression of lipogenic enzymes such as fatty acid synthase and acetyl-CoA carboxylase [27]. Since intrahepatic lipids increase when their production surpasses hepatic lipid output [9], perhaps fructose ingested by rats in our experiment did not promote lipid accumulation in the liver but led to increased TG synthesis and its storage in fat tissue. In contrast, fish oil and marine n-3 fatty acids have been shown to reduce fatty acid synthesis by suppressing SREBP-1c expression and preventing LXRα activation [28, 29] and increase lipolysis by upregulating hepatic PPAR-α [30], therefore hindering TG synthesis and fat storage. In a similar experiment, the addition of fish oil in the animal diet failed to attenuate an increase in serum TGs due to fructose consumption 30 days after fructose ingestion in water was initiated [21]. It would seem therefore that fish oil is more efficient at preventing the development of elevated serum TGs rather than treating it, since in the aforementioned experiment fish oil ingestion started alongside fructose addition in water.
Although fructose is implicated in hepatic lipogenesis, no liver alterations were seen in our experiment, such as liver steatosis, fibrosis, increased liver weight, or increased hepatic enzymes. Indeed, multiple protocols are employed to study NAFLD in the literature [31,32,33,34,35], differing in terms of fructose concentration, experiment length, and animal model, making the comparison between heterogeneous data a difficult task. In similar experiments that utilized Wistar rats receiving fructose in drinking water, increased hepatic lipid accumulation was seen after 8 weeks of fructose consumption [35], and liver injury consisting of hepatocyte degeneration and hyperemia were seen after 24 weeks [34]. Even though water intake data was not available in these previous studies, it can be hypothesized that the extent to which fructose can lead to hepatic injury and lipid accumulation depends heavily on the experiment’s duration and water intake, which directly impacts fructose intake. As discussed previously, since water intake was not increased in the Fr group, as opposed to what is seen in similar experiments [20, 21], fructose intake was limited. Other methods that circumvent this limitation can be used to induce NAFLD and obesity in animals, such as including fructose in the actual diet alongside saturated fat [36,37,38], leading to an increased fructose intake and a stronger stimulus to provoke metabolic alterations, given the effects of saturated fats on the onset of inflammation and weight gain in animal models [39, 40].
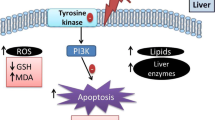
Fish oil also led to increased hepatic MDA and reduced hepatic α-tocopherol in the FOFr group, suggesting there was a significant increase in lipid peroxidation in the liver and that hepatic α-tocopherol, a fat-soluble antioxidant which acts as a hydrogen donor, was significantly depleted. It has been described that the susceptibility for free radical damage increases alongside the number of double bonds in fatty acids present in tissues [15]. As such, the incorporation of a significant amount of n-3 fatty acids such as EPA and DHA in the liver, which are available in fish oil and abundant in double bonds, can potentially lead to increased lipid peroxidation, and thus pose a threat to overall hepatic health. Oxidative stress is now recognized as a significant part in the development of many metabolic conditions such as chronic inflammation and hypertension [7], and in the liver, it is directly implicated in alcoholic hepatic diseases [41]. Therefore, these findings raise concerns regarding the safety of fish oil when ingested at such high dosages.
Experiments in which a fructose-rich diet containing 10% fish oil was given to rats also saw an increase in lipid peroxidation in the form of increased liver and serum TBARS, leading to significant DNA damage [42]. Later, the same group showed that a 5% dosage of fish oil could still ameliorate metabolic alterations of a fructose-enriched diet without increasing lipid peroxidation [43]. It would seem, therefore, that as the diet’s fish oil content rises, so does hepatic lipid peroxidation, but at a given proportion, fish oil retains its therapeutic properties without favoring oxidative stress. Curiously, hepatic GSH was increased in the Fr group, a result that is not seen in other experiments [42, 43]. This change may have protected these animals from oxidative stress, given that MDA levels were normal in the Fr group. The ratio of reduced glutathione to oxidized glutathione (GSH/GSSG) is an effective method to assess the redox state of glutathione in tissues, which unfortunately was not present in our experiment. It would be important to utilize this analysis in future experiments, possibly shedding more light on this specific alteration found in animals consuming fructose.
Conclusions
Obesity and its comorbidities affect an ever-growing number of patients around the world. As such, it is fundamental to study its causes and different therapeutic options. In our experiment, we found that fructose ingestion through water led to increased weight gain and increased adiposity and serum triglycerides, without causing liver injury or steatosis. The inclusion of fish oil in the diet prevented such alterations and further decreased serum cholesterol and fat tissue. However, at such a dosage, it also led to increased hepatic lipid peroxidation. Considering fish oil is a widely available pharmaceutical agent, the present study raises concerns regarding its unadvised use by patients. Therefore, we propose that fish oil and its PUFAs may be suitable agents to prevent the metabolic effects of fructose ingestion, but an adequate dosage must be tested in further studies and clinical trials with the aim of avoiding deleterious changes while still retaining a therapeutic effect.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- NAFLD:
-
Non-alcoholic fatty liver disease
- MDA:
-
Malondialdehyde
- GSH:
-
Reduced glutathione
- ROS:
-
Reactive oxygen species
- FFA:
-
Free fatty acids
- SREBP1c:
-
Sterol regulatory element-binding protein 1c
- ChREBP:
-
Carbohydrate-responsive element-binding protein
- PUFA:
-
Polyunsaturated fatty acid
- ALT:
-
Transaminases alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- LXRα:
-
Liver X receptor alpha
- PPAR-α:
-
Peroxisome proliferator-activated receptor alpha
- EPA:
-
Eicosapentaenoic acid
- DHA:
-
Docosahexaenoic acid
- TBARS:
-
Thiobarbituric acid reactive substances
References
Fruh SM (2017) Obesity: risk factors, complications, and strategies for sustainable long-term weight management. J Am Assoc Nurse Pract 29:S3–S14. https://doi.org/10.1002/2327-6924.12510
Cox AJ, West NP, Cripps AW (2015) Obesity, inflammation, and the gut microbiota. Lancet Diabetes Endocrinol 3:207–215. https://doi.org/10.1016/S2213-8587(14)70134-2
Castro GS, Calder PC (2018) Non-alcoholic fatty liver disease and its treatment with n-3 polyunsaturated fatty acids. Clin Nutr 37:37–55. https://doi.org/10.1016/j.clnu.2017.01.006
Milić S, Lulić D, Štimac D (2014) Non-alcoholic fatty liver disease and obesity: Biochemical, metabolic and clinical presentations. World J Gastroenterol 20:9330–9337. https://doi.org/10.3748/wjg.v20.i28.9330
Han D, Williams E, Cadenas E (2001) Mitochondrial respiratory chain-dependent generation of superoxide anion and its release into the intermembrane space. Biochem J 353:411. https://doi.org/10.1042/0264-6021:3530411
Brownlee M (2001) Banting Lecture 2004, the pathobiology of diabetic complications a unifying mechanism. Diabetes 51:7–18. https://doi.org/10.2337/diabetes.51.1.7
Matsuda M, Shimomura I (2013) Increased oxidative stress in obesity: implications for metabolic syndrome, diabetes, hypertension, dyslipidemia, atherosclerosis, and cancer. Obes Res Clin Pract 7:1–12. https://doi.org/10.1016/j.orcp.2013.05.004
Tappy L, Le KA (2010) Metabolic effects of fructose and the worldwide increase in obesity. Physiol Rev 90:23–46. https://doi.org/10.1152/physrev.00019.2009
Ter Horst KW, Serlie MJ (2017) Fructose consumption, lipogenesis, and non-alcoholic fatty liver disease. Nutrients 9:1–20. https://doi.org/10.3390/nu9090981
Iizuka K, Bruick RK, Liang G et al (2004) From the cover: deficiency of carbohydrate response element-binding protein (ChREBP) reduces lipogenesis as well as glycolysis. Proc Natl Acad Sci 101:7281–7286. https://doi.org/10.1073/pnas.0401516101
Aragno M, Tomasinelli CE, Vercellinatto I et al (2009) SREBP-1c in nonalcoholic fatty liver disease induced by Western-type high-fat diet plus fructose in rats. Free Radic Biol Med 47:1067–1074. https://doi.org/10.1016/j.freeradbiomed.2009.07.016
Jump DB, Botolin D, Wang Y et al (2006) Fatty acids and gene transcription. Scand J Food Nutr 50:5–12. https://doi.org/10.1080/17482970601069318
Lombardo YB, Chicco AG (2006) Effects of dietary polyunsaturated n-3 fatty acids on dyslipidemia and insulin resistance in rodents and humans. A review. J Nutr Biochem 17:1–13. https://doi.org/10.1016/j.jnutbio.2005.08.002
Ayala A, Muñoz MF, Argüelles S (2014) Lipid peroxidation: production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid Med Cell Longev 2014:1–31. https://doi.org/10.1155/2014/360438
Pamplona R, Portero-Otín M, Riba D et al (1998) Mitochondrial membrane peroxidizability index is inversely related to maximum life span in mammals. J Lipid Res 39:1989–1994
Bligh E, Dyer W (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:912–917
Gérard-Monnier D, Erdelmeier I, Régnard K et al (1998) Reactions of 1-methyl-2-phenylindole with malondialdehyde and 4-hydroxyalkenals. Analytical applications to a colorimetric assay of lipid peroxidation. Chem Res Toxicol 11:1176–1183. https://doi.org/10.1021/tx9701790
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205. https://doi.org/10.1016/0003-2697(68)90092-4
Arnaud J, Fortis I, Blachier S, et al (1991) Simultaneous determination of retinol, α-tocopherol and β-carotene in serum by isocratic high-performance liquid chromatography. J Chromatogr B Biomed Sci Appl 572:103–116. https://doi.org/10.1016/0378-4347(91)80476-S
Mamikutty N, Thent ZC, Sapri SR et al (2014) The establishment of metabolic syndrome model by induction of fructose drinking water in male wistar rats. Biomed Res Int 2014:1–8. https://doi.org/10.1155/2014/263897
Sulis PM, Motta K, Barbosa AM et al (2017) Impact of fish oil supplementation and interruption of fructose ingestion on glucose and lipid homeostasis of rats drinking different concentrations of fructose. Biomed Res Int:2017. https://doi.org/10.1155/2017/4378328
Baena M, Sangüesa G, Hutter N et al (2014) Fructose supplementation impairs rat liver autophagy through mTORC activation without inducing endoplasmic reticulum stress. Biochim Biophys Acta - Mol Cell Biol Lipids 1851:107–116. https://doi.org/10.1016/j.bbalip.2014.11.003
Vilà L, Roglans N, Perna V et al (2011) Liver AMP/ATP ratio and fructokinase expression are related to gender differences in AMPK activity and glucose intolerance in rats ingesting liquid fructose. J Nutr Biochem 22:741–751. https://doi.org/10.1016/j.jnutbio.2010.06.005
Poudyal H, Campbell F, Brown L (2010) Olive leaf extract attenuates cardiac, hepatic, and metabolic changes in high carbohydrate–, high fat–fed rats. J Nutr 140:946–953. https://doi.org/10.3945/jn.109.117812
Iizuka K, Horikawa Y (2008) ChREBP: a glucose-activated transcription factor involved in the development of metabolic syndrome. Endocr J 55:617–624. https://doi.org/10.1507/endocrj.K07E-110
Miyazaki M, Dobrzyn A, Man WC et al (2004) Stearoyl-CoA desaturase 1 gene expression is necessary for fructose-mediated induction of lipogenic gene expression by sterol regulatory element-binding protein-1c-dependent and independent mechanisms. J Biol Chem 279:25164–25171. https://doi.org/10.1074/jbc.M402781200
Ferré P, Foufelle F (2007) SREBP-1c transcription factor and lipid homeostasis: Clinical perspective. Horm Res 68:72–82. https://doi.org/10.1159/000100426
Howell G, Deng X, Yellaturu C et al (2009) N-3 polyunsaturated fatty acids suppress insulin-induced SREBP-1c transcription via reduced trans-activating capacity of LXRα. Biochim Biophys Acta - Mol Cell Biol Lipids 1791:1190–1196. https://doi.org/10.1016/j.bbalip.2009.08.008
Sekiya M, Yahagi N, Matsuzaka T et al (2003) Polyunsaturated fatty acids ameliorate hepatic steatosis in obese mice by SREBP-1 suppression. Hepatology 38:1529–1539. https://doi.org/10.1016/j.hep.2003.09.028
Deckelbaum RJ, Worgall TS, Seo T (2006) N-3 fatty acids and gene expression. Am J Clin Nutr 83:1520–1525. https://doi.org/10.2307/3976938
Abd El-Haleim EA, Bahgat AK, Saleh S (2016) Resveratrol and fenofibrate ameliorate fructose-induced nonalcoholic steatohepatitis by modulation of genes expression. World J Gastroenterol 22:2931–2948. https://doi.org/10.3748/wjg.v22.i10.2931
Chaturvedi A, Ravishankar B, Rao N, Mudugaru R (2016) Evaluation of metabolic syndrome signs in two different animal models. Pharmacol Toxicol Biomed Reports 2:41–44. https://doi.org/10.5530/ptb.2016.2.5
Moura RF, Ribeiro C, de Oliveira JA et al (2008) Metabolic syndrome signs in Wistar rats submitted to different high-fructose ingestion protocols. Br J Nutr 101:1178–1184. https://doi.org/10.1017/S0007114508066774
Pektas MB, Yücel G, Koca HB et al (2017) Dietary fructose-induced hepatic injury in male and female rats: influence of resveratrol. Drug Res (Stuttg) 67:103–110. https://doi.org/10.1055/s-0042-118386
Mamikutty N, Thent ZC (2015) Haji Suhaimi F (2015) Fructose-drinking water induced nonalcoholic fatty liver disease and ultrastructural alteration of hepatocyte mitochondria in Male wistar rat. Biomed Res Int. https://doi.org/10.1155/2015/895961
Lee JS, Jun DW, Kim EK et al (2015) Histologic and metabolic derangement in high-fat, high-fructose, and combination diet animal models. Sci World J 2015. https://doi.org/10.1155/2015/306326
Fakhoury-Sayegh N, Trak-Smayra V, Khazzaka A et al (2015) Characteristics of nonalcoholic fatty liver disease induced in wistar rats following four different diets. Nutr Res Pract 9:350–357. https://doi.org/10.4162/nrp.2015.9.4.350
Jegatheesan P, Beutheu S, Freese K et al (2016) Preventive effects of citrulline on Western diet-induced non-alcoholic fatty liver disease in rats. Br J Nutr 116:191–203. https://doi.org/10.1017/S0007114516001793
Maric T, Woodside B, Luheshi GN (2014) The effects of dietary saturated fat on basal hypothalamic neuroinflammation in rats. Brain Behav Immun 36:35–45. https://doi.org/10.1016/j.bbi.2013.09.011
Rocha-Rodrigues S, Rodríguez A, Gonçalves IO et al (2017) Impact of physical exercise on visceral adipose tissue fatty acid profile and inflammation in response to a high-fat diet regimen. Int J Biochem Cell Biol 87:114–124. https://doi.org/10.1016/j.biocel.2017.04.008
Cichoz-Lach H, Michalak A (2014) Oxidative stress as a crucial factor in liver diseases. World J Gastroenterol 20:8082–8091. https://doi.org/10.3748/wjg.v20.i25.8082
Castro GS, Santos RA, Portari GV et al (2012) Omega-3 improves glucose tolerance but increases lipid peroxidation and DNA damage in hepatocytes of fructose-fed rats. Appl Physiol Nutr Metab 37:233–240. https://doi.org/10.1139/h11-154
Castro GS, Deminice R, Simões-Ambrosio LMC et al (2015) Dietary docosahexaenoic acid and eicosapentaenoic acid influence liver triacylglycerol and insulin resistance in rats fed a high-fructose diet. Mar Drugs 13:1864–1881. https://doi.org/10.3390/md13041864
Acknowledgements
None.
Funding
GPE was supported by a research scholarship provided by the National Council for Scientific and Technological Development (CNPq, process number 157244/2017-2). The University of Sao Paulo was responsible for all of the project’s costs.
Author information
Authors and Affiliations
Contributions
GPE was responsible for manuscript elaboration and for conducting the project and worked on all of its stages. CSM and HPV-S assisted in animal handling and in discussions regarding the results. PPO and HH assisted in animal handling and biochemical assays. FSM and MKO assisted in histological procedures and result interpretation. AAJ is the main investigator responsible for the laboratory and supervised the project. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All animal procedures were submitted and approved by the Ribeirão Preto Medical School’s Ethics Committee of Animal Experimentation (protocol number 185/2017).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Esteves, G.P., Manca, C.S., Veida-Silva, H.P. et al. A fish oil-rich diet leads to lower adiposity and serum triglycerides but increases liver lipid peroxidation in fructose-fed rats. Egypt Liver Journal 10, 35 (2020). https://doi.org/10.1186/s43066-020-00045-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43066-020-00045-9