Abstract
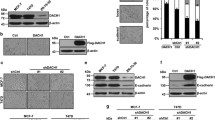
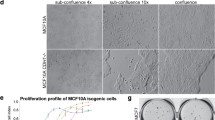
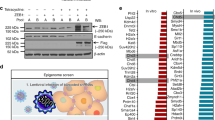
The loss of E-cadherin causes dysfunction of the cell–cell junction machinery, which is an initial step in epithelial-to-mesenchymal transition (EMT), facilitating cancer cell invasion and the formation of metastases. A set of transcriptional repressors of E-cadherin (CDH1) gene expression, including Snail1, Snail2 and Zeb2 mediate E-cadherin downregulation in breast cancer. However, the molecular mechanisms underlying the control of E-cadherin expression in breast cancer progression remain largely unknown. Here, by using global gene expression approaches, we uncover a novel function for Cdc42 GTPase-activating protein (CdGAP) in the regulation of expression of genes involved in EMT. We found that CdGAP used its proline-rich domain to form a functional complex with Zeb2 to mediate the repression of E-cadherin expression in ErbB2-transformed breast cancer cells. Conversely, knockdown of CdGAP expression led to a decrease of the transcriptional repressors Snail1 and Zeb2, and this correlated with an increase in E-cadherin levels, restoration of cell–cell junctions, and epithelial-like morphological changes. In vivo, loss of CdGAP in ErbB2-transformed breast cancer cells impaired tumor growth and suppressed metastasis to lungs. Finally, CdGAP was highly expressed in basal-type breast cancer cells, and its strong expression correlated with poor prognosis in breast cancer patients. Together, these data support a previously unknown nuclear function for CdGAP where it cooperates in a GAP-independent manner with transcriptional repressors to function as a critical modulator of breast cancer through repression of E-cadherin transcription. Targeting Zeb2–CdGAP interactions may represent novel therapeutic opportunities for breast cancer treatment.









Similar content being viewed by others
Accession codes
References
Gonzalez DM, Medici D . Signaling mechanisms of the epithelial-mesenchymal transition. Sci Signal 2014; 7: re8.
Wong TS, Gao W, Chan JY . Transcription regulation of E-cadherin by zinc finger E-box binding homeobox proteins in solid tumors. Biomed Res Int 2014; 2014: 921564.
King CR, Kraus MH, Aaronson SA . Amplification of a novel v-erbB-related gene in a human mammary carcinoma. Science 1985; 229: 974–976.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL . Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 1987; 235: 177–182.
Northey JJ, Chmielecki J, Ngan E, Russo C, Annis MG, Muller WJ et al. Signaling through ShcA is required for TGFβ and Neu/ErbB-2 induced breast cancer cell motility and invasion. Mol Cell Biol 2008; 28: 3162–3176.
Laurin M, Huber J, Pelletier A, Houalla T, Park M, Fukui Y et al. Rac-specific guanine nucleotide exchange factor DOCK1 is a critical regulator of HER2-mediated breast cancer metastasis. Proc Natl Acad Sci USA 2013; 110: 7434–7439.
Lamarche-Vane N, Hall A . CdGAP, a novel proline-rich GTPase-activating protein for Cdc42 and Rac. J Biol Chem 1998; 273: 29172–29177.
Tcherkezian J, Danek EI, Jenna S, Triki I, Lamarche-Vane N . Extracellular signal-regulated kinase 1 interacts with and phosphorylates CdGAP at an important regulatory site. Mol Cell Biol 2005; 25: 6314–6329.
Danek EI, Tcherkezian J, Meriane M, Triki I, Lamarche-Vane N . Glycogen synthase kinase-3 phosphorylates CdGAP at a consensus ERK1 regulatory site. J Biol Chem 2007; 282: 3624–3631.
Southgate L, Machado RD, Snape KM, Primeau M, Dafou D, Ruddy DM et al. Gain-of-function mutations of ARHGAP31, a Cdc42/Rac1 GTPase regulator, cause syndromic cutis aplasia and limb anomalies. Am J Hum Genet 2011; 88: 574–585.
Isrie M, Wuyts W, Van Esch H, Devriendt K . Isolated terminal limb reduction defects: extending the clinical spectrum of Adams-Oliver syndrome and ARHGAP31 mutations. Am J Med Genet A 2014; 164A: 1576–1579.
He Y, Northey JJ, Primeau M, Machado RD, Trembath R, Siegel PM et al. CdGAP is required for transforming growth factor β- and Neu/ErbB-2-induced breast cancer cell motility and invasion. Oncogene 2011; 30: 1032–1045.
Hajra KM, Chen DY, Fearon ER . The SLUG zinc-finger protein represses E-cadherin in breast cancer. Cancer Res 2002; 62: 1613–1618.
Elloul S, Elstrand MB, Nesland JM, Tropé CG, Kvalheim G, Goldberg I et al. Snail, Slug, and Smad-interacting protein 1 as novel parameters of disease aggressiveness in metastatic ovarian and breast carcinoma. Cancer 2005; 103: 1631–1643.
Wang CA, Drasin D, Pham C, Jedlicka P, Zaberezhnyy V, Guney M et al. Homeoprotein Six2 promotes breast cancer metastasis via transcriptional and epigenetic control of E-cadherin expression. Cancer Res 2014; 74: 7357–7370.
Long J, Zuo D, Park M . Pc2-mediated sumoylation of Smad-interacting protein 1 attenuates transcriptional repression of E-cadherin. J Biol Chem 2005; 280: 35477–35489.
Sánchez-Tilló E, Siles L, de Barrios O, Cuatrecasas M, Vaquero EC, Castells A et al. Expanding roles of ZEB factors in tumorigenesis and tumor progression. Am J Cancer Res 2011; 1: 897–912.
Holliday DL, Speirs V . Choosing the right cell line for breast cancer research. Breast Cancer Res 2011; 13: 215.
van de Vijver MJ, He YD, van't Veer LJ, Dai H, Hart AA, Voskuil DW et al. A gene-expression signature as a predictor of survival in breast cancer. N Engl J Med 2002; 347: 1999–2009.
Haibe-Kains B, Desmedt C, Loi S, Culhane AC, Bontempi G, Quackenbush J et al. A three-gene model to robustly identify breast cancer molecular subtypes. J Natl Cancer Inst 2012; 104: 311–325.
Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal 2013; 6: pl1.
Hu QP, Kuang JY, Yang QK, Bian XW, Yu SC . Beyond a tumor suppressor: soluble E-cadherin promotes the progression of cancer. Int J Cancer 2015; 138: 2804–2812.
Tcherkezian J, Lamarche-Vane N . Current knowledge of the large RhoGAP family of proteins. Biol Cell 2007; 99: 67–86.
Kim TY, Vigil D, Der CJ, Juliano RL . Role of DLC-1, a tumor suppressor protein with RhoGAP activity, in regulation of the cytoskeleton and cell motility. Cancer Metastasis Rev 2009; 28: 77–83.
Lahoz A, Hall A . DLC1: a significant GAP in the cancer genome. Genes Dev 2008; 22: 1724–1730.
Barras D, Widmann C . GAP-independent functions of DLC1 in metastasis. Cancer Metastasis Rev 2014; 33: 87–100.
Naji L, Pacholsky D, Aspenström P . ARHGAP30 is a Wrch-1-interacting protein involved in actin dynamics and cell adhesion. Biochem Biophys Res Commun 2011; 409: 96–102.
Wang J, Qian J, Hu Y, Kong X, Chen H, Shi Q et al. ArhGAP30 promotes p53 acetylation and function in colorectal cancer. Nat Commun 2014; 5: 4735.
Meester JA, Southgate L, Stittrich AB, Venselaar H, Beekmans SJ, den Hollander N et al. Heterozygous loss-of-function mutations in DLL4 causeAdams-Oliver syndrome. Am J Hum Genet 2015; 97: 475–482.
Southgate L, Sukalo M, Karountzos AS, Taylor EJ, Collinson CS, Ruddy D et al. Haploinsufficiency of the Notch1 receptor as a cause of Adams-Oliver syndrome with variable cardiac anomalies. Circ Cardiovasc Genet 2015; 8: 572–581.
Shaheen R, Aglan M, Keppler-Noreuil K, Faqeih E, Ansari S, Horton K et al. Mutations in EOGT confirm the genetic heterogeneity of autosomalrecessive Adams-Oliver syndrome. Am J Hum Genet 2013; 92: 598–604.
Hassed SJ, Wiley GB, Wang S, Lee JY, Li S, Xu W et al. RBPJ mutations identified in two families affected by Adams-Oliver syndrome. Am J Hum Genet 2012; 91: 391–395.
Wu ZQ, Rowe RG, Lim KC, Lin Y, Willis A, Tang Y et al. Snail1/Notch1 signalling axis controls embryonic vascular development. Nat Commun 2013; 5: 3998.
Caron C, DeGeer J, Fournier P, Duquette PM, Luangrath V, Ishii H et al. The Cdc42/Rac1 GTPase regulator CdGAP is critical for vascular development and VEGF-mediated angiogenesis. Sci Rep 2016; 6: 27485.
Koboldt DC, Fulton RS, McLellan MD, Schmidt H, Kalicki-Veizer J, McMichael JF et al. Comprehensive molecular portraits of human breast tumours. Nature 2012; 490: 61–70.
Metzger-Filho O, Sun Z, Viale G, Price KN, Crivellari D, Snyder RD et al. Patterns of recurrence and outcome according to breast cancer subtypes in lymph node-negative disease: results from international breast cancer study group trials VIII and IX. J Clin Oncol 2013; 31: 3083–3090.
Karimzadeh F, Primeau M, Mountassif D, Rouiller I, Lamarche-Vane N . A stretch of polybasic residues mediates Cdc42 GTPase-activating protein (CdGAP) binding to phosphatidylinositol 3,4,5-trisphosphate and regulates its GAP activity. J Biol Chem 2012; 287: 19610–19621.
Bolger AM, Lohse M, Usadel B . Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 2014; 30: 2114–2120.
Kim D, Pertea G, Trapnell C, Pimentel H, Kelley R, Salzberg SL . TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol 2013; 14: R36.
Trapnell C, Roberts A, Goff L, Pertea G, Kim D, Kelley DR, Pimentel H et al. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat Protoc 2012; 7: 562–578.
Tabariès S, Annis MG, Hsu BE, Tam CE, Savage P, Park M et al. Lyn modulates Claudin-2 expression and is a therapeutic target for breast cancer liver metastasis. Oncotarget 2015; 6: 9476–9487.
Parker RL, Huntsman DG, Lesack DW, Cupples JB, Grant DR, Akbari M et al. Assessment of interlaboratory variation in the immunohistochemical determination of estrogen receptor status using a breast cancer tissue microarray. Am J Clin Pathol 2002; 117: 723–728.
Makretsov N, Gilks CB, Coldman AJ, Hayes M, Huntsman D . Tissue microarray analysis of neuroendocrine differentiation and its prognostic significance in breast cancer. Hum Pathol 2003; 34: 1001–1008.
Liu CL, Prapong W, Natkunam Y, Alizadeh A, Montgomery K, Gilks CB et al. Software tools for high-throughput analysis and archiving of immunohistochemistry staining data obtained with tissue microarrays. Am J Pathol 2002; 161: 1557–1565.
Azim HA Jr, Michiels S, Bedard PL, Singhal SK, Criscitiello C, Ignatiadis M et al. Elucidating prognosis and biology of breast cancer arising in young women using gene expression profiling. Clin Cancer Res 2012; 18: 1341–1351.
Desmedt C, Piette F, Loi S, Wang Y, Lallemand F, Haibe-Kains B et al. Strong time dependence of the 76-gene prognostic signature for node-negative breast cancer patients in the TRANSBIG multicenter independent validation series. Clin Cancer Res 2007; 13: 3207–3214.
Acknowledgements
We thank Dr Patricia Tonin for helpful discussions and Dr John J Bergeron for critically reading this manuscript. We are grateful to Dr Morag Park (McGill University) for providing the Zeb2 and E-cadherin-luc constructs. We thank Dr Torsten Nielsen for providing the human breast cancer TMA and the CdGAP IHC staining through the Genetic Pathology Evaluation Center (GPEC), ZF Dong for IHC staining of primary tumors, Dr Min Fu for assistance with confocal microscopy at the imaging platform of the RI-MUHC, Martin Primeau and Vilayphone Luangrath for affinity purification of the CdGAP antibodies. NL-V was a recipient of a FRSQ chercheur-national and a William Dawson scholar. Canadian Institute for Health Research (CIHR) MOP-119544 to NL-V and MOP- 144425 to JFC. JFC is a recipient of a FRQS senior scholarship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
He, Y., Northey, J., Pelletier, A. et al. The Cdc42/Rac1 regulator CdGAP is a novel E-cadherin transcriptional co-repressor with Zeb2 in breast cancer. Oncogene 36, 3490–3503 (2017). https://doi.org/10.1038/onc.2016.492
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.492
- Springer Nature Limited
This article is cited by
-
CdGAP maintains podocyte function and modulates focal adhesions in a Src kinase-dependent manner
Scientific Reports (2022)
-
The emerging roles of srGAPs in cancer
Molecular Biology Reports (2022)
-
ARHGAP10 inhibits the epithelial–mesenchymal transition of non-small cell lung cancer by inactivating PI3K/Akt/GSK3β signaling pathway
Cancer Cell International (2021)
-
MEF2A transcriptionally upregulates the expression of ZEB2 and CTNNB1 in colorectal cancer to promote tumor progression
Oncogene (2021)
-
CdGAP promotes prostate cancer metastasis by regulating epithelial-to-mesenchymal transition, cell cycle progression, and apoptosis
Communications Biology (2021)