Key Points
-
Rabies is a devastating infectious disease, known to mankind since the 23rd century B.C., that is distributed almost worldwide. Effective control mechanisms in the developed world overshadow the fact that rabies is still an enormous threat to human health in developing countries, with an annual death toll of more than 55,000 people. Therefore, rabies is considered a neglected infectious disease.
-
New methods to genetically manipulate the rabies virus genome fostered a new era in the study of the rabies virus life cycle, rabies pathogenicity and the development of vaccines and therapeutics.
-
Replication and transcription of rabies virus in vitro is now well understood; however, many other steps of the viral replication cycle, such as the entry of rabies virus into its target cells, remain poorly explored. Others, such as the intracellular transport of rabies virus, have only recently become targets of intense study. Also, the nature of the rabies virus receptor and its importance for the neurotropism of rabies virus are still matters of debate.
-
Apoptosis was previously suggested as a mechanism in rabies virus pathogenesis. However, recent research strongly suggests that pathogenic rabies virus probably prevents apoptosis rather than induce it.
-
Great progress has been made in understanding the interaction of rabies virus with the innate immune system. Rabies virus interferes with the host cell's ability to block early steps in viral replication through the type I interferon response by its multifunctional phosphoprotein.
-
Although considerable progress has been made in the past 10 years in elucidating the interaction of rabies virus with its host cell, we suggest that a standardized use of viral strains will simplify the combination and comparison of research results, thus facilitating further understanding of this important viral pathogen and the prevention and treatment of the devastating disease it causes.
Abstract
Rabies virus, the prototypical neurotropic virus, causes one of the most lethal zoonotic diseases. According to official estimates, over 55,000 people die of the disease annually, but this is probably a severe underestimation. A combination of virulence factors enables the virus to enter neurons at peripheral sites and travel through the spinal cord to the brain of the infected host, where it often induces aggression that facilitates the transfer of the virus to a new host. This Review summarizes the current knowledge of the replication cycle of rabies virus and virus– host cell interactions, both of which are fundamental elements in our quest to understand the life cycle of rabies virus and the pathogenesis of rabies.





Similar content being viewed by others
References
Knobel, D. L. et al. Re-evaluating the burden of rabies in Africa and Asia. Bull. World Health Organ. 83, 360–368 (2005). This study describes the effect of rabies in developing countries.
Baer, G. M. in Rabies (eds Jackson, A. C. & Wunner, W. H.) 1–22 (Elsevier, London, 2007).
Zinke, G. Neue Ansichten der Hundswuth, ihre Ursachen und Folgen, nebst einer sicheren Behandlungsart von tollen Tieren gebissenen Menschen. Jena Review 16, 212–218 (1804).
Galtier, V. Etudes sur la rage. Annales Medicine Veterinaire 28, 627–639 (1879).
Pasteur, L. Methode pour prevenir la rage apres morsure. Compte Rendue Academie Science 101, 765–773 (1885).
Takayama, N. Rabies: a preventable but incurable disease. J. Infect. Chemother. 14, 8–14 (2008).
Warrell, D. A. The clinical picture of rabies in man. Trans. R. Soc. Trop. Med. Hyg. 70, 188–195 (1976).
Dietzschold, B., Faber, M. & Schnell, M. J. New approaches to the prevention and eradication of rabies. Expert Rev. Vaccines 2, 399–406 (2003). This is an overview about current vaccine approaches to control rabies.
Cleaveland, S., Fevre, E. M., Kaare, M. & Coleman, P. G. Estimating human rabies mortality in the United Republic of Tanzania from dog bite injuries. Bull. World Health Organ. 80, 304–310 (2002).
Jackson, A. C. Rabies. Neurol. Clin. 26, 717–726 (2008).
Kelly, R. M. & Strick, P. L. Rabies as a transneuronal tracer of circuits in the central nervous system. J. Neurosci. Methods 103, 63–71 (2000).
Ugolini, G. Use of rabies virus as a transneuronal tracer of neuronal connections: implications for the understanding of rabies pathogenesis. Dev. Biol. (Basel) 131, 493–506 (2008).
Ugolini, G. Specificity of rabies virus as a transneuronal tracer of motor networks: transfer from hypoglossal motoneurons to connected second-order and higher order central nervous system cell groups. J. Comp. Neurol. 356, 457–480 (1995).
Rathelot, J. A. & Strick, P. L. Subdivisions of primary motor cortex based on cortico-motoneuronal cells. Proc. Natl Acad. Sci. USA 106, 918–923 (2009).
Schnell, M. J., Mebatsion, T. & Conzelmann, K. K. Infectious rabies viruses from cloned cDNA. EMBO J. 13, 4195–4203 (1994). This study describes the first infectious clone of a non-segmented negative-stranded RNA virus.
Nadin-Davis, S. A. & Fehlner-Gardiner, C. Lyssaviruses: current trends. Adv. Virus Res. 71, 207–250 (2008).
Hanlon, C. A. et al. Efficacy of rabies biologics against new lyssaviruses from Eurasia. Virus Res. 111, 44–54 (2005).
Conzelmann, K. K., Cox, J. H., Schneider, L. G. & Thiel, H. J. Molecular cloning and complete nucleotide sequence of the attenuated rabies virus SAD B19. Virology 175, 485–499 (1990).
Tordo, N., Poch, O., Ermine, A., Keith, G. & Rougeon, F. Walking along the rabies genome: is the large G-L intergenic region a remnant gene? Proc. Natl Acad. Sci. USA 83, 3914–3918 (1986). This is the first complete sequence and analysis for a rabies virus genome.
Gaudin, Y., Ruigrok, R. W., Tuffereau, C., Knossow, M. & Flamand, A. Rabies virus glycoprotein is a trimer. Virology 187, 627–632 (1992).
Mebatsion, T., Weiland, F. & Conzelmann, K. K. Matrix protein of rabies virus is responsible for the assembly and budding of bullet-shaped particles and interacts with the transmembrane spike glycoprotein G. J. Virol. 73, 242–250 (1999).
Preuss, M. A. et al. Intravenous inoculation of a bat-associated rabies virus causes lethal encephalopathy in mice through invasion of the brain via neurosecretory hypothalamic fibers. PLoS Pathog. 5, e1000485 (2009).
Etessami, R. et al. Spread and pathogenic characteristics of a G-deficient rabies virus recombinant: an in vitro and in vivo study. J. Gen. Virol. 81, 2147–2153 (2000).
Mebatsion, T., Konig, M. & Conzelmann, K. K. Budding of rabies virus particles in the absence of the spike glycoprotein. Cell 84, 941–951 (1996). This is a key paper describing the budding and assembly of rabies virus.
Lentz, T. L., Burrage, T. G., Smith, A. L., Crick, J. & Tignor, G. H. Is the acetylcholine receptor a rabies virus receptor? Science 215, 182–184 (1982).
Lafon, M. Rabies virus receptors. J. Neurovirol. 11, 82–87 (2005). This is an comprehensive review on rabies virus receptors.
Murphy, F. A. & Bauer, S. P. Early street rabies virus infection in striated muscle and later progression to the central nervous system. Intervirology 3, 256–268 (1974).
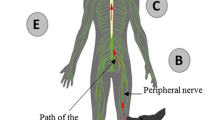
Watson, H. D., Tignor, G. H. & Smith, A. L. Entry of rabies virus into the peripheral nerves of mice. J. Gen. Virol. 56, 372–382 (1981).
Thoulouze, M. I. et al. The neural cell adhesion molecule is a receptor for rabies virus. J. Virol. 72, 7181–7190 (1998).
Tuffereau, C., Benejean, J., Blondel, D., Kieffer, B. & Flamand, A. Low-affinity nerve-growth factor receptor (P75NTR) can serve as a receptor for rabies virus. EMBO J. 17, 7250–7259 (1998).
Tuffereau, C. et al. The rabies virus glycoprotein receptor p75NTR is not essential for rabies virus infection. J. Virol. 81, 13622–13630 (2007).
Superti, F., Derer, M. & Tsiang, H. Mechanism of rabies virus entry into CER cells. J. Gen. Virol. 65, 781–789 (1984).
Conti, C., Superti, F. & Tsiang, H. Membrane carbohydrate requirement for rabies virus binding to chicken embryo related cells. Intervirology 26, 164–168 (1986).
Wunner, W. H., Reagan, K. J. & Koprowski, H. Characterization of saturable binding sites for rabies virus. J. Virol. 50, 691–697 (1984).
Dietzschold, B. et al. Differences in cell-to-cell spread of pathogenic and apathogenic rabies virus in vivo and in vitro. J. Virol. 56, 12–18 (1985).
Pulmanausahakul, R., Li, J., Schnell, M. J. & Dietzschold, B. The glycoprotein and the matrix protein of rabies virus affect pathogenicity by regulating viral replication and facilitating cell-to-cell spread. J. Virol. 82, 2330–2338 (2008).
McGettigan, J. P. et al. Rabies virus-based vectors expressing human immunodeficiency virus type 1 (HIV-1) envelope protein induce a strong, cross-reactive cytotoxic T-lymphocyte response against envelope proteins from different HIV-1 isolates. J. Virol. 75, 4430–4434 (2001).
Mebatsion, T. & Conzelmann, K. K. Specific infection of CD4+ target cells by recombinant rabies virus pseudotypes carrying the HIV-1 envelope spike protein. Proc. Natl Acad. Sci. USA 93, 11366–11370 (1996).
Roche, S., Bressanelli, S., Rey, F. A. & Gaudin, Y. Crystal structure of the low-pH form of the vesicular stomatitis virus glycoprotein G. Science 313, 187–191 (2006).
Roche, S., Albertini, A. A., Lepault, J., Bressanelli, S. & Gaudin, Y. Structures of vesicular stomatitis virus glycoprotein: membrane fusion revisited. Cell. Mol. Life Sci. 65, 1716–1728 (2008).
Malgaroli, A., Vallar, L. & Zimarino, V. Protein homeostasis in neurons and its pathological alterations. Curr. Opin. Neurobiol. 16, 270–274 (2006).
Jacob, Y., Badrane, H., Ceccaldi, P. E. & Tordo, N. Cytoplasmic dynein LC8 interacts with lyssavirus phosphoprotein. J. Virol. 74, 10217–10222 (2000).
Raux, H., Flamand, A. & Blondel, D. Interaction of the rabies virus P protein with the LC8 dynein light chain. J. Virol. 74, 10212–10216 (2000).
Mebatsion, T. Extensive attenuation of rabies virus by simultaneously modifying the dynein light chain binding site in the P protein and replacing Arg333 in the G protein. J. Virol. 75, 11496–11502 (2001).
Tan, G. S., Preuss, M. A., Williams, J. C. & Schnell, M. J. The dynein light chain 8 binding motif of rabies virus phosphoprotein promotes efficient viral transcription. Proc. Natl Acad. Sci. USA 104, 7229–7234 (2007). This research indicates that dynein LC8 doesn't have a role in transport but instead is involved in the transcription of rabies virus.
Klingen, Y., Conzelmann, K. K. & Finke, S. Double-labeled rabies virus: live tracking of enveloped virus transport. J. Virol. 82, 237–245 (2008).
Parveen, Z. et al. Cell-type-specific gene delivery into neuronal cells in vitro and in vivo. Virology 314, 74–83 (2003).
Martin-Rendon, E., Azzouz, M. & Mazarakis, N. D. Lentiviral vectors for the treatment of neurodegenerative diseases. Curr. Opin. Mol. Ther. 3, 476–481 (2001).
Mazarakis, N. D. et al. Rabies virus glycoprotein pseudotyping of lentiviral vectors enables retrograde axonal transport and access to the nervous system after peripheral delivery. Hum. Mol. Genet. 10, 2109–2121 (2001). This study suggests a major role of the glycoprotein in retrograde axonal transport.
Azzouz, M., Kingsman, S. M. & Mazarakis, N. D. Lentiviral vectors for treating and modeling human CNS disorders. J. Gene Med. 6, 951–962 (2004).
Faber, M. et al. Overexpression of the rabies virus glycoprotein results in enhancement of apoptosis and antiviral immune response. J. Virol. 76, 3374–3381 (2002).
Whelan, S. P., Barr, J. N. & Wertz, G. W. Transcription and replication of nonsegmented negative-strand RNA viruses. Curr. Top. Microbiol. Immunol. 283, 61–119 (2004).
Lahaye, X. et al. Functional characterization of Negri bodies (NBs) in rabies virus infected cells: evidence that NBs are sites of viral transcription and replication. J. Virol. 83, 7948–7958 (2009).
Menager, P. et al. Toll-like receptor 3 (TLR3) plays a major role in the formation of rabies virus Negri bodies. PLoS Pathog. 5, e1000315 (2009). This study shows that an important factor of the innate immune response (TLR3) is used for efficient viral production.
Conzelmann, K. K. & Schnell, M. Rescue of synthetic genomic RNA analogs of rabies virus by plasmid-encoded proteins. J. Virol. 68, 713–719 (1994).
Tordo, N. & Kouknetzoff, A. The rabies virus genome: an overview. Onderstepoort J. Vet. Res. 60, 263–269 (1993).
Albertini, A. A. et al. Crystal structure of the rabies virus nucleoprotein-RNA complex. Science 313, 360–363 (2006).
Iverson, L. E. & Rose, J. K. Localized attenuation and discontinuous synthesis during vesicular stomatitis virus transcription. Cell 23, 477–484 (1981).
Finke, S., Cox, J. H. & Conzelmann, K. K. Differential transcription attenuation of rabies virus genes by intergenic regions: generation of recombinant viruses overexpressing the polymerase gene. J. Virol. 74, 7261–7269 (2000). This is a detailed study of the intergenic region of rabies virus and their function for transcription and replication.
Rose, J. K. Complete intergenic and flanking gene sequences from the genome of vesicular stomatitis virus. Cell 19, 415–421 (1980).
Brzozka, K., Finke, S. & Conzelmann, K. K. Identification of the rabies virus α/β interferon antagonist: phosphoprotein P interferes with phosphorylation of interferon regulatory factor 3. J. Virol. 79, 7673–7681 (2005).
Yang, J., Koprowski, H., Dietzschold, B. & Fu, Z. F. Phosphorylation of rabies virus nucleoprotein regulates viral RNA transcription and replication by modulating leader RNA encapsidation. J. Virol. 73, 1661–1664 (1999).
Yang, J. et al. The specificity of rabies virus RNA encapsidation by nucleoprotein. Virology 242, 107–117 (1998).
Liu, P., Yang, J., Wu, X. & Fu, Z. F. Interactions amongst rabies virus nucleoprotein, phosphoprotein and genomic RNA in virus-infected and transfected cells. J. Gen. Virol. 85, 3725–3734 (2004).
McGettigan, J. P. et al. Functional human immunodeficiency virus type 1 (HIV-1) Gag-Pol or HIV-1 Gag-Pol and env expressed from a single rhabdovirus-based vaccine vector genome. J. Virol. 77, 10889–10899 (2003).
Wojczyk, B. S., Stwora-Wojczyk, M., Shakin-Eshleman, S., Wunner, W. H. & Spitalnik, S. L. The role of site-specific N-glycosylation in secretion of soluble forms of rabies virus glycoprotein. Glycobiology 8, 121–130 (1998).
Wu, X., Lei, X. & Fu, Z. F. Rabies virus nucleoprotein is phosphorylated by cellular casein kinase II. Biochem. Biophys. Res. Commun. 304, 333–338 (2003).
Toriumi, H. & Kawai, A. Association of rabies virus nominal phosphoprotein (P) with viral nucleocapsid (NC) is enhanced by phosphorylation of the viral nucleoprotein (N). Microbiol. Immunol. 48, 399–409 (2004).
Wu, X., Gong, X., Foley, H. D., Schnell, M. J. & Fu, Z. F. Both viral transcription and replication are reduced when the rabies virus nucleoprotein is not phosphorylated. J. Virol. 76, 4153–4161 (2002).
Carroll, A. R. & Wagner, R. R. Role of the membrane (M) protein in endogenous inhibition of in vitro transcription by vesicular stomatitis virus. J. Virol. 29, 134–142 (1979).
Wagner, R. R. in The Rhabdoviruses.(ed. Wagner, R. R.) 9–74 (Plenum, New York, 1987).
Finke, S., Mueller-Waldeck, R. & Conzelmann, K. K. Rabies virus matrix protein regulates the balance of virus transcription and replication. J. Gen. Virol. 84, 1613–1621 (2003).
Komarova, A. V. et al. Rabies virus matrix protein interplay with eIF3, new insights into rabies virus pathogenesis. Nucleic Acids Res. 35, 1522–1532 (2007).
Ganser-Pornillos, B. K., Yeager, M. & Sundquist, W. I. The structural biology of HIV assembly. Curr. Opin. Struct. Biol. 18, 203–217 (2008).
Schnell, M. J. et al. Recombinant rabies virus as potential live-viral vaccines for HIV-1. Proc. Natl Acad. Sci. USA 97, 3544–3549 (2000).
McKenna, P. M. et al. Immunogenicity study of glycoprotein-deficient rabies virus expressing simian/human immunodeficiency virus SHIV89.6P envelope in a rhesus macaque. J. Virol. 78, 13455–13459 (2004).
Schnell, M. J. et al. Requirement for a non-specific glycoprotein cytoplasmic domain sequence to drive efficient budding of vesicular stomatitis virus. EMBO J. 17, 1289–1296 (1998).
Graham, S. C. et al. Rhabdovirus matrix protein structures reveal a novel mode of self-association. PLoS Pathog. 4, e1000251 (2008).
Chen, B. J. & Lamb, R. A. Mechanisms for enveloped virus budding: can some viruses do without an ESCRT? Virology 372, 221–232 (2008). This is a comprehensive review that describes the mechanism for viral budding, including that of rhabdoviruses.
Wirblich, C. et al. PPEY motif within the rabies virus (RV) matrix protein is essential for efficient virion release and RV pathogenicity. J. Virol. 82, 9730–9738 (2008).
Harty, R. N., Paragas, J., Sudol, M. & Palese, P. A proline-rich motif within the matrix protein of vesicular stomatitis virus and rabies virus interacts with WW domains of cellular proteins: implications for viral budding. J. Virol. 73, 2921–2929 (1999). This is the first study showing that rhadboviruses recruit the cellular machinery for their budding process.
Irie, T., Licata, J. M., McGettigan, J. P., Schnell, M. J. & Harty, R. N. Budding of PPxY-containing rhabdoviruses is not dependent on host proteins TGS101 and VPS4A. J. Virol. 78, 2657–2665 (2004).
Fensterl, V. & Sen, G. C. Interferons and viral infections. Biofactors 35, 14–20 (2009).
Baloul, L. & Lafon, M. Apoptosis and rabies virus neuroinvasion. Biochimie 85, 777–788 (2003).
Baloul, L., Camelo, S. & Lafon, M. Up-regulation of Fas ligand (FasL) in the central nervous system: a mechanism of immune evasion by rabies virus. J. Neurovirol. 10, 372–382 (2004).
Johnson, N. et al. Lyssavirus infection activates interferon gene expression in the brain. J. Gen. Virol. 87, 2663–2667 (2006).
Johnson, N. et al. Inflammatory responses in the nervous system of mice infected with a street isolate of rabies virus. Dev. Biol. (Basel) 131, 65–72 (2008).
Wang, Z. W. et al. Attenuated rabies virus activates, while pathogenic rabies virus evades, the host innate immune responses in the central nervous system. J. Virol. 79, 12554–12565 (2005).
Hornung, V. et al. 5′-triphosphate RNA is the ligand for RIG-I. Science 314, 994–997 (2006).
Jackson, A. C., Rossiter, J. P. & Lafon, M. Expression of Toll-like receptor 3 in the human cerebellar cortex in rabies, herpes simplex encephalitis, and other neurological diseases. J. Neurovirol. 12, 229–234 (2006).
Faul, E. J., Wanjalla, C. N., McGettigan, J. P. & Schnell, M. J. Interferon-β expressed by a rabies virus-based HIV-1 vaccine vector serves as a molecular adjuvant and decreases pathogenicity. Virology 382, 226–238 (2008).
Vidy, A., Chelbi-Alix, M. & Blondel, D. Rabies virus P protein interacts with STAT1 and inhibits interferon signal transduction pathways. J. Virol. 79, 14411–14420 (2005).
Brzozka, K., Finke, S. & Conzelmann, K. K. Inhibition of interferon signaling by rabies virus phosphoprotein P: activation-dependent binding of STAT1 and STAT2. J. Virol. 80, 2675–2683 (2006).
Pasdeloup, D. et al. Nucleocytoplasmic shuttling of the rabies virus P protein requires a nuclear localization signal and a CRM1-dependent nuclear export signal. Virology 334, 284–293 (2005).
Vidy, A., El Bougrini, J., Chelbi-Alix, M. K. & Blondel, D. The nucleocytoplasmic rabies virus P protein counteracts interferon signaling by inhibiting both nuclear accumulation and DNA binding of STAT1. J. Virol. 81, 4255–4263 (2007).
Moseley, G. W. et al. Dual modes of rabies P-protein association with microtubules: a novel strategy to suppress the antiviral response. J. Cell Sci. 122, 3652–3662 (2009). This study describes a new pathway of how rabies virus can block IFN signalling.
Blondel, D. et al. Rabies virus P and small P products interact directly with PML and reorganize PML nuclear bodies. Oncogene 21, 7957–7970 (2002).
Bernardi, R. & Pandolfi, P. P. Structure, dynamics and functions of promyelocytic leukaemia nuclear bodies. Nature Rev. Mol. Cell Biol. 8, 1006–1016 (2007).
Chelbi-Alix, M. K., Vidy, A., El Bougrini, J. & Blondel, D. Rabies viral mechanisms to escape the IFN system: the viral protein P interferes with IRF-3, Stat1, and PML nuclear bodies. J. Interferon Cytokine Res. 26, 271–280 (2006).
Isamu, M., Yukihiro, N., Takashi, Y. & Yoshinobu, K. Virus-induced neuronal apoptosis as pathological and protective responses of the host. Rev. Med. Virol. 14, 209–216 (2004).
Jackson, A. C. & Rossiter, J. P. Apoptosis plays an important role in experimental rabies virus infection. J. Virol. 71, 5603–5607 (1997).
Jackson, A. C. & Park, H. Apoptotic cell death in experimental rabies in suckling mice. Acta Neuropathol. 95, 159–164 (1998).
Fu, Z. F. & Jackson, A. C. Neuronal dysfunction and death in rabies virus infection. J. Neurovirol. 11, 101–106 (2005).
Bijlenga, C. & Hernandez-Baumgarten, E. M. Adaptation, attenuation and plaque purification of a rabiesvirus isolate (V319) from a vampire bat (Desmodus rotundus). Cornell Vet. 70, 290–299 (1980).
Thoulouze, M. I., Lafage, M., Montano-Hirose, J. A. & Lafon, M. Rabies virus infects mouse and human lymphocytes and induces apoptosis. J. Virol. 71, 7372–7380 (1997).
Morimoto, K., Hooper, D. C., Spitsin, S., Koprowski, H. & Dietzschold, B. Pathogenicity of different rabies virus variants inversely correlates with apoptosis and rabies virus glycoprotein expression in infected primary neuron cultures. J. Virol. 73, 510–518 (1999).
Hemachudha, T. et al. Pathophysiology of human paralytic rabies. J. Neurovirol. 11, 93–100 (2005).
Dhingra, V., Li, X., Liu, Y. & Fu, Z. F. Proteomic profiling reveals that rabies virus infection results in differential expression of host proteins involved in ion homeostasis and synaptic physiology in the central nervous system. J. Neurovirol. 13, 107–117 (2007).
Sarmento, L., Tseggai, T., Dhingra, V. & Fu, Z. F. Rabies virus-induced apoptosis involves caspase-dependent and caspase-independent pathways. Virus Res. 121, 144–151 (2006).
Thoulouze, M. I. et al. High level of Bcl-2 counteracts apoptosis mediated by a live rabies virus vaccine strain and induces long-term infection. Virology 314, 549–561 (2003).
Ubol, S., Kasisith, J., Pitidhammabhorn, D. & Tepsumethanol, V. Screening of pro-apoptotic genes upregulated in an experimental street rabies virus-infected neonatal mouse brain. Microbiol. Immunol. 49, 423–431 (2005).
Pulmanausahakul, R. et al. Overexpression of cytochrome c by a recombinant rabies virus attenuates pathogenicity and enhances antiviral immunity. J. Virol. 75, 10800–10807 (2001).
Prehaud, C., Lay, S., Dietzschold, B. & Lafon, M. Glycoprotein of nonpathogenic rabies viruses is a key determinant of human cell apoptosis. J. Virol. 77, 10537–10547 (2003).
Thoulouze, M. I. et al. Apoptosis inversely correlates with rabies virus neurotropism. Ann. N.Y. Acad. Sci. 1010, 598–603 (2003).
Préhaud, C. et al. Attenuation of rabies virulence: takeover by the cytoplasmic domain of its envelope protein. Sci. Signal. (in the press).
Koprowski, H. et al. In vivo expression of inducible nitric oxide synthase in experimentally induced neurologic diseases. Proc. Natl Acad. Sci. USA 90, 3024–3027 (1993).
Roy, A., Phares, T. W., Koprowski, H. & Hooper, D. C. Failure to open the blood-brain barrier and deliver immune effectors to central nervous system tissues leads to the lethal outcome of silver-haired bat rabies virus infection. J. Virol. 81, 1110–1118 (2007).
Phares, T. W., Fabis, M. J., Brimer, C. M., Kean, R. B. & Hooper, D. C. A peroxynitrite-dependent pathway is responsible for blood-brain barrier permeability changes during a central nervous system inflammatory response: TNF-α is neither necessary nor sufficient. J. Immunol. 178, 7334–7343 (2007).
Ubol, S., Sukwattanapan, C. & Maneerat, Y. Inducible nitric oxide synthase inhibition delays death of rabies virus-infected mice. J. Med. Microbiol. 50, 238–242 (2001).
Roy, A. & Hooper, D. C. Lethal silver-haired bat rabies virus infection can be prevented by opening the blood-brain barrier. J. Virol. 81, 7993–7998 (2007).
Fabis, M. J., Phares, T. W., Kean, R. B., Koprowski, H. & Hooper, D. C. Blood-brain barrier changes and cell invasion differ between therapeutic immune clearance of neurotrophic virus and CNS autoimmunity. Proc. Natl Acad. Sci. USA 105, 15511–15516 (2008).
Rossiter, J. P., Hsu, L. & Jackson, A. C. Selective vulnerability of dorsal root ganglia neurons in experimental rabies after peripheral inoculation of CVS-11 in adult mice. Acta Neuropathol. 118, 249–259 (2009).
Wu, X. & Rupprecht, C. E. Glycoprotein gene relocation in rabies virus. Virus Res. 131, 95–99 (2008).
Ito, N., Takayama, M., Yamada, K., Sugiyama, M. & Minamoto, N. Rescue of rabies virus from cloned cDNA and identification of the pathogenicity-related gene: glycoprotein gene is associated with virulence for adult mice. J. Virol. 75, 9121–9128 (2001).
Faber, M. et al. Identification of viral genomic elements responsible for rabies virus neuroinvasiveness. Proc. Natl Acad. Sci USA 101, 16328–16332 (2004).
Jackson, A. C. in Rabies (eds Jackson, A. C. & Wunner, W. H.) 309–340 (Elsevier, London, 2007).
Hanlon, C. A., Niezgoda, M. & Rupprecht, C. E. in Rabies (eds Jackson, A. C. & Wunner, W. H.) 201–258 (Elsevier, London, 2007).
Manning, S. E. et al. Human rabies prevention — United States, 2008: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm. Rep. 57, 1–28 (2008).
Lodmell, D. L. & Ewalt, L. C. Post-exposure DNA vaccination protects mice against rabies virus. Vaccine 19, 2468–2473 (2001).
Lodmell, D. L., Parnell, M. J., Bailey, J. R., Ewalt, L. C. & Hanlon, C. A. One-time gene gun or intramuscular rabies DNA vaccination of non-human primates: comparison of neutralizing antibody responses and protection against rabies virus 1 year after vaccination. Vaccine 20, 838–844 (2001).
Lodmell, D. L., Parnell, M. J., Bailey, J. R., Ewalt, L. C. & Hanlon, C. A. Rabies DNA vaccination of non-human primates: post-exposure studies using gene gun methodology that accelerates induction of neutralizing antibody and enhances neutralizing antibody titers. Vaccine 20, 2221–2228 (2002).
Lodmell, D. L., Ray, N. B. & Ewalt, L. C. Gene gun particle-mediated vaccination with plasmid DNA confers protective immunity against rabies virus infection. Vaccine 16, 115–118 (1998).
Lodmell, D. L. et al. DNA immunization protects nonhuman primates against rabies virus. Nature Med. 4, 949–952 (1998).
Lodmell, D. L., Ray, N. B., Ulrich, J. T. & Ewalt, L. C. DNA vaccination of mice against rabies virus: effects of the route of vaccination and the adjuvant monophosphoryl lipid A (MPL). Vaccine 18, 1059–1066 (2000).
Cenna, J. et al. Immune modulating effect by a phosphoprotein-deleted rabies virus vaccine vector expressing two copies of the rabies virus glycoproteins. Vaccine (2008).
Ito, N. et al. Characterization of M gene-deficient rabies virus with advantages of effective immunization and safety as a vaccine strain. Microbiol. Immunol. 49, 971–979 (2005).
Lees, C. Y. et al. Induction of protective immunity by topic application of a recombinant adenovirus expressing rabies virus glycoprotein. Vet. Microbiol. 85, 295–303 (2002).
Li, W. H., Zhang, Y., Wang, S. H., Liu, L. & Yang, F. [Recombinant replication-defective adenovirus based rabies vaccine]. Chung Kuo i Hsueh Ko Hsueh Yuan Hsueh Pao Acta Academiae Medicinae Sinicae 25, 650–654 (2003).
Morimoto, K., Shoji, Y. & Inoue, K. Characterizatio of P gene-deficient rabies virus: propagation, pathogenicity and antigenicity. Virus Res. 111, 61–67 (2005).
Vos, A. et al. Immunogenicity of an E1-deleted recombinant human adenovirus against rabies by different routes of administration. J. Gen. Virol. 82, 2191–2197 (2001).
Dodet, B. et al. Fighting rabies in Africa: the Africa Rabies Expert Bureau (AfroREB). Vaccine 26, 6295–6298 (2008).
Lang, J., Feroldi, E. & Vien, N. C. Pre-exposure purified vero cell rabies vaccine and concomitant routine childhood vaccinations: 5-year post-vaccination follow-up study of an infant cohort in Vietnam. J. Trop. Pediatr. 55, 26–31 (2009).
Shanbag, P. et al. Protecting Indian schoolchildren against rabies: pre-exposure vaccination with purified chick embryo cell vaccine (PCECV) or purified verocell rabies vaccine (PVRV). Hum. Vaccin. 4, 365–369 (2008).
Acknowledgements
We thank Gene Tan for assistance in designing figure 2 and Paul Schiffmacher for assistance in designing figures 3–5. This work was supported in part by a Center for Neuroanatomy and Neurotropic Viruses grant to Peter Strick, Center for Neuroscience, University of Pittsburgh (USPHS-NCRR P40RR028604, subcontract to M.J.S.) and internal funds of Thomas Jefferson University, Medical College, Jefferson Vaccine Center, Philadelphia, USA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Matthias J. Schnell, James P. McGettigan, Christoph Wirblich and Amy Papaneri
The cell biology of rabies virus: using stealth to reach the brain. Nature Reviews Microbiology 30 Nov 2009, [doi:nrmicro2260].
J. P. McGettigan and M. J. Schnell have patents and/or patent applications for rabies virus-based vaccines and vectors, grants from companies as well as personal financial interests.
Related links
Glossary
- Trans-complemented
-
Complemented by expression of missing components in the host cell.
- Postsynaptic
-
Positioned after the synapse.
- Presynaptic
-
Positioned before the synapse.
- Aggresome
-
An inclusion body in which proteins accumulate when the cells degradation machinery is not functioning properly or is overwhelmed.
- Toll-like receptor
-
A receptor that recognizes viral, bacterial or fungal material and signals to induce an immune response.
- Internal ribosome entry site
-
RNA sequence that allows ribosomes to bind to an mRNA at a position other than the 5′ end.
Rights and permissions
About this article
Cite this article
Schnell, M., McGettigan, J., Wirblich, C. et al. The cell biology of rabies virus: using stealth to reach the brain. Nat Rev Microbiol 8, 51–61 (2010). https://doi.org/10.1038/nrmicro2260
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro2260
- Springer Nature Limited
This article is cited by
-
A plant cytorhabdovirus modulates locomotor activity of insect vectors to enhance virus transmission
Nature Communications (2023)
-
Nucleic acid drug vectors for diagnosis and treatment of brain diseases
Signal Transduction and Targeted Therapy (2023)
-
Serological surveillance for rabies and canine distemper in wild boar in Heilongjiang province, China
European Journal of Wildlife Research (2023)
-
Lighting Up Neural Circuits by Viral Tracing
Neuroscience Bulletin (2022)
-
Revealing the Cell Entry Dynamic Mechanism of Single Rabies Virus Particle
Chemical Research in Chinese Universities (2022)