Abstract
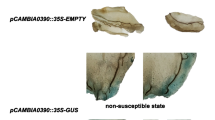
Very short exposures of embryogenic calli of Vitis vinifera cv. Superior Seedless grape plants to diluted cultures of Agrobacterium resulted in plant tissue necrosis and subsequent cell death. Antibiotics used for Agrobacterium elimination or as plant selectable markers were not responsible for this necrotic response. Rather, cell death seemed to be oxygen-dependent and correlated with elevated levels of peroxides. Therefore, we studied the effects on necrosis of various combinations of antioxidants during and after grape-Agrobacterium cocultivation. The combination of polyvinylpolypyrrolidone and dithiothreitol was found to improve plant viability. Tissue necrosis was completely inhibited by these antioxidants while Agrobacterium virulence was not effected. These treatments enabled the recovery of stable transgenic grape plants resistant to hygromycin.
Similar content being viewed by others
References
Barbault, T.J., Skene, K.G., Cain, P.A. and Scott, N.S. 1990. Genetic transformation of grapevine cells. J. Exp. Bot. 41: 1045–1049.
Berres, R., Otten, L., Tinland, B., Malgarini-Clog, E. and Walter, B. 1992. Transformation of Vitis tissue by different strains of Agrobacterium tumefaciens containing the T-6B gene. Plant Cell Rep. 11: 192–195.
Guellec, V., David, C., Branchard, M. and Tempe, J. 1990. Agrobacterium rhizogenes mediated transformation of grapevine (Vitis vinifera L.). Plant Cell Tissue and Organ Culture. 20: 211–215.
Mullins, M.G., Tang, F.C.A. and Facciotti, D. 1990. Agrobacterium-mediated genetic transformation of grapevines: transgenic plants of Vitis rupestris Scheele and buds of Vitis vinifera L. Bio/Technology 8: 1041–1045.
Le Gall, O., Torregrosa, L., Danglot, Y., Candresse, T. and Bouquet, A. 1994. Agrabacterium-mediated genetic transformation of grapevine somatic embryos and regeneration of transgenic plants expressing the coat protein of grapevine chrome mosaic nepovirus (GCMV). Plant Sci. 102: 161–170.
Walter, B. 1994. Aspects botanique et genetique de l'application des nouvelles techniques de biologie moleculaire d'informatique et de biotechnologies (transfert de genes) à la vigne,in The 74th General Assembly of the International Vine Office (O.I.V), Paris, France. Moët-Hennessy (ed.). pp. 1–43.
Martinelli, L. and Mandoline, G. 1994. Genetic transformation and regeneration of transgenic plants in grapevine (Vitis rupestris S.). Theor. Appl. Genet. 88: 621–628.
Gray, D.J. and Meredith, C.P. 1992. The grape, in Biotechnology in Agriculture, No. 8. Biotechnology of Perennial Fruit Crops. Hammerschlagm, F. and Litz, R.E. (eds.). CAB International, Wallingford. pp. 229–264.
Kikkert, J.R., Hebert-Soule, D., Wallace, P. and Reisch, B.I. 1995. Transgenic plantlets of ‘Chencellor’ grapevine (Vitis sp.) from biolistic transformation of embryo-genie cell suspension. Plant Cell Rep. In press
Colby, S.M., Juncosa, A.M. and Meredith, C.P. 1991. Cellular differences in Agrobacterium susceptibility and regeneration capacity restrict the development of transgenic grapevines. J. Amer. Soc. Hort. Sci. 116: 356–361.
Perl, A., Saad, S., Sahar, N. and Holland, D. 1995. Establishment of long term embryogenic cultures of seedless Vitis vinifera cultivars—a synergistic effect of auxins and the role of abscisic acid. Plant Sci. 104: 193–200.
Stamp, J.A. and Meredith, C. 1988. Proliferative somatic embryogenesis from zygotic embryos of grapevine. J. Amer. Soc. Hort. Sci. 113: 941–945.
Stamp, J.A. and Meredith, C.P. 1988. Somatic embryogenesis from leaves and anthers of grapevine. Scientia. Hortic. 35: 235–250.
Ream, W. 1989. Agrobacterium tumefaciens and interkingdom genetic exchange. Annual Review of Phytopathology 27: 583–618.
Yanofsky, M., Lowe, B., Montoya, A., Rubin, R., Krul, W., Gordon, M., and Nester, E. 1985. Molecular and genetic analysis of factors controlling host range in Agrobacterium tumefaciens . Mol. Gene. Gen. 201: 237–246.
Pu, X.A. and Goodman, R.N. 1992. Induction of necrogenesis by Agrobacterium tumefaciens on grape explants. Physiol. and Mol. Plant Pathol. 41: 241–254.
Klement, Z. and Goodman, R.N. 1967. The hypersensitive reaction to infection by bacterial plant pathogens. Annal. Review of Phytopathology 5: 17–44.
Thomashow, M.F., Panagopoulos, C.G., Gordon, M.P. and Nester, E.W. 1980. Host range of Agrobacterium tumefaciens is determined by the Ti-plasmid. Nature 283: 794–796.
Robertson, T. 1983. The Organic Consistuent of Higher Plants. 5th ed. Cordus, North Amherst, MA.
Mayer, A.M. and Harel, E. 1979. Polyphenol oxidases in plants. Phytochemistry 18: 193–215.
Vaugh, K.C. and Duke, S.O. 1984. Function of polyphenol oxidase in higher plants. Physiol. Plant. 60: 106–112.
Lagrimini, L.M. 1992. Wound-induced deposition of polyphenols in transgenic plants over expressing peroxidase. Plant Physiol. 96: 26–31.
Hahlobrock, K. and Scheel, D. 1989. Physiology and molecular biology of phenyl-propanoid metabolism. Ann. Rev. Plant. Physiol. Plant Mol. Biol. 40: 347–369.
Preece, J.E. and Compton, M.E. 1991. Problems with explant exudation in micro-propagation, in Biotechnology in Agriculture and Forestry, High Tech and Micropropagation, Vol. 17, Bajaj, Y.P.S. (ed.). Springer-Verlag, Berlin, Heidelberg. pp. 168–189.
Mathews, H., Litz, R.E., Wilde, H.D., Merkle, S.A. and Wetzstein, H.Y. 1992. Stable integration and expression of β-glucuronidase and NPTII genes in mango somatic embryos. In Vitro Cell Dev. Biol. 28P: 172–178.
Mathews, H., Litz, R.E., Wilde, H.D. and Wetzstein, H.Y. 1993. Genetic transformation of mango. Acta Horticulture 341: 93–97.
Murashige, T. and Skoog, F. 1962. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15: 473–497.
Perl, A., Shaul, O. and Galili, G. 1993. Bacterial dihydrodipicolinate synthase and desensitized aspartate kinase: Two novel selectable markers for plant transformation relying on overproduction of lysine and threonine. Bio/Technology 11: 715–718.
Becker, D., Kemper, E., Schell, J. and Masterson, R. 1992. New plant binary with selectable markers proximal to the left T-DNA border. Plant Mol. Biol. 20: 1195–1197.
Vancanneyt, G., Schmidt, R., O'Connor-Sanchez, A., Willmitzer, L. and Rocha-Sosa, M. 1990. Construction of an intron-containing marker gene: Splicing of the intron in transgenic plants and its use in Agrobacterium-mediated plant transformation. Mol. Gen. Genet. 220: 245–250.
Uoyd, G., McCown, B. 1981. Commercially feasible micrpropagation of Mountain laurel, Kalmia latifolia, by the use of shoot tip culture. Int. Plant Prop. Soc. Proc. 30: 421–427.
Fhance, B. and Maehly, A.C. 1955. Methods in Enzymology, II, Collowick, S.R and Kaplan, N.O. (eds.). Academic Press, New York. pp. 764–775.
Steenkamp, J., Wiid, I., Lourens, A. and Van Helden, P. 1994. Improved method for DNA extraction from Vitis vinifera . Am. J. Enol. Vitic. 45: 102–106.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Perl, A., Lotan, O., Abu-Abied, M. et al. Establishment of an Agrobacterium-mediated transformation system for grape (Vitis vinifera L.): The role of antioxidants during grape–Agrobacterium interactions. Nat Biotechnol 14, 624–628 (1996). https://doi.org/10.1038/nbt0596-624
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0596-624
- Springer Nature America, Inc.
This article is cited by
-
Establishment of an efficient callus transient transformation system for Vitis vinifera cv. ‘Chardonnay’
Protoplasma (2024)
-
Development of efficient embryo-derived regeneration system and optimization of genetic transformation in cumin (Cuminum cyminum L.)
Plant Cell, Tissue and Organ Culture (PCTOC) (2023)
-
Efficient silencing gene construct for resistance to multiple common bean (Phaseolus vulgaris L.) viruses
3 Biotech (2020)
-
Comparison of regeneration capacity and Agrobacterium-mediated cell transformation efficiency of different cultivars and rootstocks of Vitis spp. via organogenesis
Scientific Reports (2019)
-
Breeding next generation tree fruits: technical and legal challenges
Horticulture Research (2017)