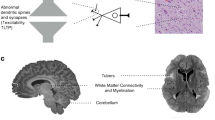
Summary
Until recently, the neuropsychiatric phenotype of tuberous sclerosis complex (TSC) was presumed to be caused by the structural brain abnormalities and/or seizures seen in the disorder. However, advances in the molecular biology of the disorder have shown that TSC is a mammalian target of rapamycin (mTOR) overactivation syndrome, and that direct molecular pathways exist between gene mutation and cognitive/neurodevelopmental phenotype. Molecularly-targeted treatments using mTOR inhibitors (such as rapamycin) are showing great promise for the physical and neurological phenotype of TSC. Pre-clinical and early-phase clinical studies of the cognitive and neurodevelopmental features of TSC suggest that some of the neuropsychiatric phenotypes might also be reversible, even in adults with the disorder. TSC, fragile X, neurofibromatosis type 1, and disorders associated with phosphatase and tensin homo (PTEN) mutations, all signal through the mTOR signaling pathway, with the TSC1-TSC2 protein complex as a molecular switchboard at its center. Together, these disorders represent as much as 14% of autism spectrum disorders (ASD). Therefore, we suggest that this signaling pathway is a key to the underlying pathophysiology of a significant subset of individuals with ASD. The study of molecularly targeted treatments in TSC and related disorders, therefore, may be of scientific and clinical value not only to those with TSC, but to a larger population that may have a neuropsychiatric phenotype attributable to mTOR overactivation or dysregulation.
Article PDF
Similar content being viewed by others
References
Crino PB, Nathanson KL, Henske EP. The tuberous sclerosis complex. N Engl J Med 2006;355: 1345–1356.
de Vries PJ, Howe CJ. The tuberous sclerosis complex proteins—a GRIPP on cognition and neurodevelopment. Trends Mol Med 2007;13: 319–326.
Jones AC. Molecular genetic and phenotypic analysis reveals differences between TSC1 and TSC2 associated familial and sporadic tuberous sclerosis. Hum Mol Genet 1997;6: 2155–2161.
Dabora SL. Mutational analysis in a cohort of 224 tuberous sclerosis patients indicates increased severity of TSC2, compared with TSC1, disease in multiple organs. Am J Hum Genet 2001;68: 64–80.
Roach ES, Gomez MR, Northrup H. Tuberous sclerosis consensus conference: revised diagnostic criteria. J Child Neurol 1998;13: 624–628.
Ridler K. Neuroanatomical correlates of memory deficits in tuberous sclerosis complex. Cereb Cortex 2007;17: 261–271.
Ridler K. Widespread anatomical abnormalities of grey and white matter structure in tuberous sclerosis. Psychol Med 2001;31: 1437–1446.
Franz DN, de Vries PJ, Crino PB. Giant cell astrocytomas in tuberous sclerosis complex. Arch Dis Child 2009;94: 75–76.
Franz DN, Leonard J, Tudor C, et al. Rapamycin causes regression of astrocytomas in tuberous sclerosis complex. Ann Neurol 2006;59: 490–498.
Wong M. Mammalian target of rapamycin (mTOR) inhibition as a potential antiepileptogenic therapy: from tuberous sclerosis to common acquired epilepsies. Epilepsia 2010;51: 27–36.
Ehninger D, de Vries PJ, Silva AJ. From mTOR to cognition: molecular and cellular mechanisms of cognitive impairment in tuberous sclerosis. J Intellect Disabil Res 2009;53: 838–851.
Prather P, de Vries PJ. Behavioral and cognitive aspects of tuberous sclerosis complex. J Child Neurol 2004;19: 666–674.
Joinson C. Learning disability and epilepsy in an epidemiological sample of individuals with tuberous sclerosis complex. Psychol Med 2003;33: 335–344.
de Vries PJ, Prather P. The tuberous sclerosis complex. N Engl J Med 2007;356: 92.
Bolton PF, Park RJ, Higgins NP, Griffiths PD, Pickles A. Neuro-epileptic determinants of autism spectrum disorders in tuberous sclerosis complex. Brain 2002;125: 1247–1255.
Kelleher RJ, Bear MF. The autistic neuron: troubled translation? Cell 2008;135: 401–406.
de Vries PJ, Hunt A, Bolton PF. The psychopathologies of children and adolescents with tuberous sclerosis complex (TSC): a postal survey of UK families. Eur Child Adolesc Psychiatry 2006;16: 16–24.
Lewis JC, Thomas HV, Murphy KC, Sampson JR. Genotype and psychological phenotype in tuberous sclerosis. J Med Genet 2004;41: 203–207.
Smalley SL, Burger F, Smith M. Phenotypic variation of tuberous sclerosis in a single extended kindred. J Med Genet 1994;31: 761–765.
de Vries PJ, Gardiner J, Bolton PF. Neuropsychological attention deficits in tuberous sclerosis complex (TSC). Am J Med Genet Part A 2009;149A: 387–395.
McCartney DL, Bullmore ET, Suckling J, et al. Lateralised spatial attentional bias and white matter tract connectivity in tuberous sclerosis complex (TSC). J Intell Disabil Res 2009;53: 836.
Sahakian BJ, Owen AM. Computerized assessment in neuropsychiatry using CANTAB: discussion paper. J Royal Soc Med 1992: 85: 399–402.
Kwiatkowski DJ, Manning BD. Tuberous sclerosis: a GAP at the crossroads of multiple signalling pathways. Hum Mol Genet 2005;14: R251-R258.
Sampson JR. Therapeutic targeting of mTOR in tuberous sclerosis. Biochem Soc Trans 2009;37: 259–264.
Jacinto ER, Loewith A, Schmidt S, et al. Mammalian TOR complex 2 controls the actin cytoskeleton and is rapamycin insensitive. Nature Cell Biol 2004;6: 1122–1128.
Gwinn DM, Shackelford DB, Egan DF, et al. AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol Cell 2008;30: 214–226.
Serfontein J, Nisbet RER, Howe CJ, de Vries PJ. Evolution of the TSC1/TSC2-TOR signalling pathway. Sci Signal 2010;3: ra49.
O’Callaghan FJK, Harris T, Joinson C, et al. The relation of infantile spasms, tubers, and intelligence in tuberous sclerosis complex. Arch Dis Child 2004;89: 530–533.
Raznahan A, Higgins NP, Griffiths PD, Humphrey A, Yates JRW, Bolton PF. Biological markers of intellectual disability in tuberous sclerosis. Psychol Med 2007;37: 1293–1304.
Jozwiak S, Domanska-Pakiela D, Kotulska K, et al. New indications for antiepileptic drugs application in infants with tuberous sclerosis complex — prevention of epilepsy. Pharmacol Reports 2009;61: 580–581.
Costa-Mattioli M, Sossin WS, Klann E, Sonenberg N. Translational control of long-lasting synaptic plasticity and memory. Neuron 2009;61: 10–26.
Goorden SMI, van Woerden GM, van der Weerd L, Cheadle JP, Elgersma Y. Cognitive deficits in Tsc1 +/− mice in the absence of cerebral lesions and seizures. Ann Neurol 2007;62: 648–655.
Ehninger D, Han S, Shilyansky C, et al. Reversal of learning deficits in a Tsc2 +/− mouse model of tuberous sclerosis. Nat Med 2008;14: 843–848.
Mozzaffari M, Hoogeveen-Westerveld M, Kwiatkowski D, et al. Identification of a region required for TSC1 stability by functional analysis of TSC1 missense mutations found in individuals with tuberous sclerosis complex. BMC Med Genet 2009;10: 88.
Nellist M, Sancak O, Goedbloed MA, et al. Distinct effects of single amino-acid changes to tuberin on the function of the tuberinhamartin complex. Eur J Hum Genet 2005;13: 59–68.
Persico AM, Bourgeron T. Searching for ways out of the autism maze: genetic, epigenetic and environmental clues. Trends Neurosci 2006;29: 349–358.
de Vries PJ. Genetics and neuropsychiatric disorders: genomewide, yet narrow. Nat Med 2009;15: 850–851.
Wang LW, Berry-Kravis E, Hagerman RJ. Fragile X: leading the way for targeted treatments in autism. Neurotherapeutics 2010;7: 264–274.
Kwon CH, Luikart BW, Powell CM, et al. PTEN regulates neuronal arborisation and social interaction in mice. Neuron 2006;50: 377–388.
Johannessen CM, Reczek EE, James MF, Brems H, Legius E, Cichowski K. The NF1 tumor suppressor critically regulates TSC2 and mTOR. Proc Natl Acad Sci USA 2005;102: 8573–8578.
Kenerson H, Dundon TA, Yeung RS. Effects of rapamycin in the Eker rat model of tuberous sclerosis complex. Pediatr Res 2005;57: 67–75.
Lee L, Sudentas P, Donohue B, et al. Efficacy of a rapamycin analog (CCI-779) and IFN-γ in tuberous sclerosis mouse models. Genes Chromosomes Cancer 2005;42: 213–227.
Bissler JJ, McCormack FX, Young LR, et al. Sirolimus for angiomyolipoma in tuberous sclerosis complex or lymphangioleiomyomatosis. N Engl J Med 2008;358: 140–151.
Davies DM, Johnson SR, Tattersfield AE, et al. Sirolimus therapy in tuberous sclerosis or sporadic lymphangioleiomyomatosis. N Engl J Med 2008;358: 200–203.
Hofbauer GFL, Marcollo-Pini A, Corsenca A, et al. The mTOR inhibitor rapamycin significantly improves facial angiofibroma lesions in a patient with tuberous sclerosis. B J Dermatol 2008;159: 473–475.
Rauktys A, Lee N, Lee L, Dabora SL. Topical rapamycin inhibits tuberous sclerosis tumor growth in a nude mouse model. BMC Dermatol 2008;8: 1.
Krueger DA, Care MM, Agricola K, et al. Safety and efficacy of RAD001 (everolimus) for the treatment of subependymal giant cell astrocytoma in pediatric and adult patients with TSC. Presented at the TSC International Research Conference, University of Sussex; September 11–13, 2008; Brighton, UK.
Zeng L-H, Xu L, Gutmann DH, Wong M. Rapamycin prevents epilepsy in a mouse model of tuberous sclerosis complex. Ann Neurol 2008;63: 444–453.
Meikle L, Pillizzi K, Egnor A, et al. Response of a neuronal model of tuberous sclerosis to mammalian target of rapamycin (mTOR) inhibitors: effects on mTORC1 and Akt signalling lead to improved survival and function. J Neurosci 2008;28: 5422–5432.
Krueger DA, Holland-Bouley K, Byers AW, et al. Effect of RAD001 (everolimus) on epilepsy and neurocognition in patients with TSC. Presented at the TSC International Research Conference, University of Sussex; September 11–13, 2008; Brighton, UK.
Chen X, Garelick MG, Wang HB, Athos J, Storm DR. PI3 kinase signalling is required for retrieval and extinction of contextual memory. Nat Neurosci 2005;8: 925–931.
Dash PK, Orsi SA, Moore AN. Spatial memory formation and memory-enhancing effect of glucose involves activation of the tuberous sclerosis complex-mammalian target of rapamycin pathway. J Neurosci 2006;26: 8048–8056.
de Vries PJ. The Neuroscience of TSC — are we at risk of getting lost between the bench and the bedside? Presented at the International TSC Research Conference: From DNA to Human Therapies; September 23–26, 2009; Chicago.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Vries, P.J. Targeted treatments for cognitive and neurodevelopmental disorders in tuberous sclerosis complex. Neurotherapeutics 7, 275–282 (2010). https://doi.org/10.1016/j.nurt.2010.05.001
Issue Date:
DOI: https://doi.org/10.1016/j.nurt.2010.05.001