Abstract
Purpose of Review
Combined positron emission tomography and computer tomography with 2-deoxy-[fluorine-18]-flouro-D-glucose ([18F]FDG-PET/CT) is increasingly used in suspected infection and inflammation. Evidence is mounting within several areas. We believe [18F]FDG-PET/CT is a key modality in infection and inflammation and this overview outlines the diagnostic values in most common uses within this domain.
Recent Findings
[18F]FDG-PET/CT is considered helpful in establishing the underlying disease in 50–60% of FUO patients. In patients with complex blood stream infections, [18F]FDG-PET/CT changes treatment and reduces relapse rates and mortality—if scans are negative prognosis is favorable and it may be safe to withhold or de-escalate treatment strategy. In infectious endocarditis, [18F]FDG-PET/CT has an impact in prosthetic valve endocarditis and cardiovascular implantable electronic devices whereas its diagnostic use in NVE is limited. In spondylodiscitis, [18F]FDG-PET/CT and MRI have overall equally and complementary diagnostic performance with combined sensitivity and specificity of ~ 100%. In vascular graft infections, [18F]F DG-PET/CT is highly sensitive (> 90%) with a high negative predictive value, whereas false positive findings are challenging, especially early post-operative. Leucocyte scintigraphy combined with bone marrow scintigraphy has a better overall accuracy compared to [18F]FDG-PET/CT in suspected hip and knee prosthetic joint infections, but several practical issues favor [18F]FDG-PET/CT. Future developments of more specific tracers and novel scanner technology holds potential.
Summary
Evidence for [18F]FDG-PET/CT in infectious and inflammatory disease supports the use in fever of unknown origin, bloodstream infections, spondylodiscitis, infective endocarditis, vascular graft infections, and prosthetic joint infections. However, the literature is generally heterogeneous and several issues remain unclarified, e.g., patient selection and interpretation criteria. [18F]FDG-PET/CT has a definite role in infectious and inflammatory imaging, but firm evidence is still lacking on its precise place in the diagnostic pathways.


Similar content being viewed by others
Data Availability
No datasets were generated or analysed during the current study.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Palestro CJ. A brief history of nuclear medicine imaging of infection. J Infect Dis. 2023;228(Suppl 4):S237–40. https://doi.org/10.1093/infdis/jiac466.
Basu S, Hess S, Nielsen Braad PE, Olsen BB, Inglev S, Hoilund-Carlsen PF. The basic principles of FDG-PET/CT imaging. PET Clin. 2014;9(4):355–70. https://doi.org/10.1016/j.cpet.2014.07.006.
Gormsen LC, Hess S. Challenging but clinically useful: fluorodeoxyglucose PET/computed tomography in inflammatory and infectious diseases. PET Clin. 2020;15(2):xi–xii. https://doi.org/10.1016/j.cpet.2020.01.002.
Wright WF, Mulders-Manders CM, Auwaerter PG, Bleeker-Rovers CP. Fever of unknown origin (FUO) - a call for new research standards and updated clinical management. Am J Med. 2022;135(2):173–8. https://doi.org/10.1016/j.amjmed.2021.07.038.
Hess S. FDG-PET/CT in fever of unknown origin, bacteremia, and febrile neutropenia. PET Clin. 2020;15(2):175–85. https://doi.org/10.1016/j.cpet.2019.11.002.
Erdem H, Baymakova M, Alkan S, Letaief A, Yahia WB, Dayyab F, et al. Classical fever of unknown origin in 21 countries with different economic development: an international ID-IRI study. Eur J Clin Microbiol Infect Dis: Off Publ Eur Soc Clin Microbiol. 2023;42(4):387–98. https://doi.org/10.1007/s10096-023-04561-5.
Bharucha T, Rutherford A, Skeoch S, Alavi A, Brown M, Galloway J. Diagnostic yield of FDG-PET/CT in fever of unknown origin: a systematic review, meta-analysis, and Delphi exercise. Clin Radiol. 2017;72(9):764–71. https://doi.org/10.1016/j.crad.2017.04.014.
Yadav BK, Pannu AK, Kumar R, Rohilla M, Kumari S. Fever of unknown origin in older adults: a prospective observational study from North India. J Assoc Phys India. 2021;69(10):11–2.
Betrains A, Boeckxstaens L, Moreel L, Wright WF, Blockmans D, Van Laere K, et al. Higher diagnostic yield of 18F-FDG PET in inflammation of unknown origin compared to fever of unknown origin. Eur J Intern Med. 2023;110:71–6. https://doi.org/10.1016/j.ejim.2023.01.025.
• van Rijsewijk ND, Ffa IJ, Wouthuyzen-Bakker M, Glaudemans A. Molecular imaging of fever of unknown origin: an update. Sem Nucl Med. 2023;53(1):4–17. https://doi.org/10.1053/j.semnuclmed.2022.07.002. An excellent overview of the clinical value of molecular imaging in FUO based on a systematic literature review.
Wahl RL, Dilsizian V, Palestro CJ. At Last, (18)F-FDG for inflammation and infection! J Nucl Med: Off Publ Soc Nucl Med. 2021;62(8):1048–9. https://doi.org/10.2967/jnumed.121.262446.
Li Q, Tian R, Wang H, Li L, Wu T, Ren Y, et al. Quantifying the contribution of (18)F-FDG PET to the diagnostic assessment of pediatric patients with fever of unknown origin: a systematic review and meta-analysis. Pediatr Radiol. 2022;52(8):1500–11. https://doi.org/10.1007/s00247-022-05333-7.
Dong MJ, Zhao K, Liu ZF, Wang GL, Yang SY, Zhou GJ. A meta-analysis of the value of fluorodeoxyglucose-PET/PET-CT in the evaluation of fever of unknown origin. Eur J Radiol. 2011;80(3):834–44. https://doi.org/10.1016/j.ejrad.2010.11.018.
Gafter-Gvili A, Raibman S, Grossman A, Avni T, Paul M, Leibovici L, et al. [18F]FDG-PET/CT for the diagnosis of patients with fever of unknown origin. QJM : Monthly J Assoc Phys. 2015;108(4):289–98. https://doi.org/10.1093/qjmed/hcu193.
Palestro CJ, Brandon DC, Dibble EH, Keidar Z, Kwak JJ. FDG PET in evaluation of patients with fever of unknown origin: AJR expert panel narrative review. AJR Am J Roentgenol. 2023;221(2):151–62. https://doi.org/10.2214/ajr.22.28726.
Takeuchi M, Nihashi T, Gafter-Gvili A, Garcia-Gomez FJ, Andres E, Blockmans D, et al. Association of 18F-FDG PET or PET/CT results with spontaneous remission in classic fever of unknown origin: a systematic review and meta-analysis. Medicine. 2018;97(43):e12909. https://doi.org/10.1097/md.0000000000012909.
Keidar Z, Gurman-Balbir A, Gaitini D, Israel O. Fever of unknown origin: the role of 18F-FDG PET/CT. J Nucl Med. 2008;49(12):1980–5. https://doi.org/10.2967/jnumed.108.054692.
• Hess S. [18F]FDG-PET/CT in patients with bacteremia: clinical impact on patient management and outcome. Front Med. 2023;10. https://doi.org/10.3389/fmed.2023.1157692. A comprehensive overview of the role of [18F]FDG-PET/CT in bloodstream infections.
Brondserud MB, Pedersen C, Rosenvinge FS, Hoilund-Carlsen PF, Hess S. Clinical value of FDG-PET/CT in bacteremia of unknown origin with catalase-negative Gram-positive cocci or Staphylococcus aureus. Eur J Nucl Med Mol Imaging. 2019;46(6):1351–8. https://doi.org/10.1007/s00259-019-04289-5.
Tsai HY, Lee MH, Wan CH, Yang LY, Yen TC, Tseng JR. C-reactive protein levels can predict positive (18)F-FDG PET/CT findings that lead to management changes in patients with bacteremia. J Microbiol Immunol Infect = Wei Mian Yu Gan Ran Za Zhi. 2018;51(6):839–46. https://doi.org/10.1016/j.jmii.2018.08.003.
Vos FJ, Bleeker-Rovers CP, Sturm PD, Krabbe PF, van Dijk AP, Cuijpers ML, et al. 18F-FDG PET/CT for detection of metastatic infection in gram-positive bacteremia. J Nucl Med: Off Publ Soc Nucl Med. 2010;51(8):1234–40. https://doi.org/10.2967/jnumed.109.072371.
Berrevoets MAH, Kouijzer IJE, Aarntzen E, Janssen MJR, De Geus-Oei LF, Wertheim HFL, et al. (18)F-FDG PET/CT optimizes treatment in Staphylococcus aureus bacteremia and is associated with reduced mortality. J Nucl Med: Off Publ Soc Nucl Med. 2017;58(9):1504–10. https://doi.org/10.2967/jnumed.117.191981.
• Lapa C, Rischpler C, Bundschuh RA, Dierks A, Lang S, Wassilew G, et al. Value of [18F]FDG PET/CT in diagnosis and management of spondylodiscitis. Z Orthop Unfall. 2023;161(5):544–51. https://doi.org/10.1055/a-2075-8873. A good contemporary review on the value of [18F]FDG-PET/CT in spondylodiscitis.
Lazzeri E, Bozzao A, Cataldo MA, Petrosillo N, Manfrè L, Trampuz A, et al. Joint EANM/ESNR and ESCMID-endorsed consensus document for the diagnosis of spine infection (spondylodiscitis) in adults. Eur J Nucl Med Mol Imaging. 2019;46(12):2464–87. https://doi.org/10.1007/s00259-019-04393-6.
Paez D, Sathekge MM, Douis H, Giammarile F, Fatima S, Dhal A, et al. Comparison of MRI, [(18)F]FDG PET/CT, and (99m)Tc-UBI 29–41 scintigraphy for postoperative spondylodiscitis-a prospective multicenter study. Eur J Nucl Med Mol Imaging. 2021;48(6):1864–75. https://doi.org/10.1007/s00259-020-05109-x.
Smids C, Kouijzer IJ, Vos FJ, Sprong T, Hosman AJ, de Rooy JW, et al. A comparison of the diagnostic value of MRI and (18)F-FDG-PET/CT in suspected spondylodiscitis. Infection. 2017;45(1):41–9. https://doi.org/10.1007/s15010-016-0914-y.
Delgado V, Ajmone Marsan N, de Waha S, Bonaros N, Brida M, Burri H, et al. 2023 ESC guidelines for the management of endocarditis. Eur Heart J. 2023;44(39):3948–4042. https://doi.org/10.1093/eurheartj/ehad193.
Habib G, Lancellotti P, Antunes MJ, Bongiorni MG, Casalta JP, Del Zotti F, et al. ESC guidelines for the management of infective endocarditis: the task force for the management of infective endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J. 2015;36(44):3075–128. https://doi.org/10.1093/eurheartj/ehv319.
Yen R-F, Chen Y-C, Wu Y-W, Pan M-H, Chang S-C. Using 18-fluoro-2-deoxyglucose positron emission tomography in detecting infectious endocarditis/endoarteritis. Acad Radiol. 2004;11(3):316–21. https://doi.org/10.1016/s1076-6332(03)00715-3.
Wang TKM, Sánchez-Nadales A, Igbinomwanhia E, Cremer P, Griffin B, Xu B. Diagnosis of infective endocarditis by subtype using (18)F-fluorodeoxyglucose positron emission tomography/computed tomography: a contemporary meta-analysis. Circ Cardiovasc Imaging. 2020;13(6):e010600. https://doi.org/10.1161/circimaging.120.010600.
• Boczar KE, Lau L, Hejji N, Wiefels C. Infective endocarditis: the role of PET imaging in diagnosis and management. J Med Imaging Radiat Sci. 2024. https://doi.org/10.1016/j.jmir.2023.12.012. A very good contemporary review on the value of [18F]FDG-PET/CT in spondylodiscitis.
Hess S, Scholtens AM, Gormsen LC. Patient preparation and patient-related challenges with FDG-PET/CT in infectious and inflammatory disease. PET Clin. 2020;15(2):125–34. https://doi.org/10.1016/j.cpet.2019.11.001.
Chakfé N, Diener H, Lejay A, Assadian O, Berard X, Caillon J, et al. Editor’s Choice - European Society for Vascular Surgery (ESVS) 2020 clinical practice guidelines on the management of vascular graft and endograft infections. Eur J Vasc Endovasc Surg: Off J Eur Soc Vasc Surg. 2020;59(3):339–84. https://doi.org/10.1016/j.ejvs.2019.10.016.
Lauri C, Signore A, Glaudemans A, Treglia G, Gheysens O, Slart R, et al. Evidence-based guideline of the European Association of Nuclear Medicine (EANM) on imaging infection in vascular grafts. Eur J Nucl Med Mol Imaging. 2022;49(10):3430–51. https://doi.org/10.1007/s00259-022-05769-x.
Rojoa D, Kontopodis N, Antoniou SA, Ioannou CV, Antoniou GA. 18F-FDG PET in the diagnosis of vascular prosthetic graft infection: a diagnostic test accuracy meta-analysis. Eur J Vasc Endovasc Sur: Off J Eur Soc Vasc Surg. 2019;57(2):292–301. https://doi.org/10.1016/j.ejvs.2018.08.040.
Reinders Folmer EI, Von Meijenfeldt GCI, Van der Laan MJ, Glaudemans A, Slart R, Saleem BR, et al. Diagnostic imaging in vascular graft infection: a systematic review and meta-analysis. Eur J Vasc Endovasc Surg: Off J Eur Soc Vasc Surg. 2018;56(5):719–29. https://doi.org/10.1016/j.ejvs.2018.07.010.
Sunde SK, Beske T, Gerke O, Clausen LL, Hess S. FDG-PET/CT as a diagnostic tool in vascular graft infection: a systematic review and meta-analysis. Clin Trans Imaging. 2019;7(4):255–65. https://doi.org/10.1007/s40336-019-00336-1.
Kim SJ, Lee SW, Jeong SY, Pak K, Kim K. A systematic review and meta-analysis of (18)F-fluorodeoxyglucose positron emission tomography or positron emission tomography/computed tomography for detection of infected prosthetic vascular grafts. J Vasc Surg. 2019;70(1):307–13. https://doi.org/10.1016/j.jvs.2019.01.051.
ReindersFolmer EI, von Meijenfeldt GCI, Te Riet OokGenaamdScholten RS, van der Laan MJ, Glaudemans A, Slart R, et al. A systematic review and meta-analysis of (18)F-fluoro-d-deoxyglucose positron emission tomography interpretation methods in vascular graft and endograft infection. J Vasc Surg. 2022;72(6):2174–85. https://doi.org/10.1016/j.jvs.2020.05.065.
Mahmoodi Z, Salarzaei M, Sheikh M. Prosthetic vascular graft infection: a systematic review and meta-analysis on diagnostic accuracy of 18FDG PET/CT. Gen Thorac Cardiovasc Surg. 2022;70(3):219–29. https://doi.org/10.1007/s11748-021-01682-6.
•• Husmann L, Ledergerber B, Anagnostopoulos A, Stolzmann P, Sah BR, Burger IA, et al. The role of FDG PET/CT in therapy control of aortic graft infection. Eur J Nucl Med Mol Imaging. 2018;45(11):1987–97. https://doi.org/10.1007/s00259-018-4069-1. This prospective clinical study described the value of FDG PET/CT in therapy control of aortic graft infection.
Premkumar A, Kolin DA, Farley KX, Wilson JM, McLawhorn AS, Cross MB, et al. Projected economic burden of periprosthetic joint infection of the hip and knee in the United States. J Arthroplasty. 2021;36(5):1484-9.e3. https://doi.org/10.1016/j.arth.2020.12.005.
Signore A, Sconfienza LM, Borens O, Glaudemans A, Cassar-Pullicino V, Trampuz A, et al. Consensus document for the diagnosis of prosthetic joint infections: a joint paper by the EANM, EBJIS, and ESR (with ESCMID endorsement). Eur J Nucl Med Mol Imaging. 2019;46(4):971–88. https://doi.org/10.1007/s00259-019-4263-9.
Glaudemans AW, Prandini N, Girolamo MD, Argento G, Lauri C, Lazzeri E, et al. Hybrid imaging of musculoskeletal infections. Quart J Nucl Med Mol Imag: off Publ Ital Assoc Nucl Med (AIMN) Int Assoc Radiopharmacol (IAR) Sect So. 2018;62(1):3–13. https://doi.org/10.23736/s1824-4785.17.03045-x.
Sconfienza LM, Signore A, Cassar-Pullicino V, Cataldo MA, Gheysens O, Borens O, et al. Diagnosis of peripheral bone and prosthetic joint infections: overview on the consensus documents by the EANM, EBJIS, and ESR (with ESCMID endorsement). Eur Radiol. 2019;29(12):6425–38. https://doi.org/10.1007/s00330-019-06326-1.
Romanò CL, Petrosillo N, Argento G, Sconfienza LM, Treglia G, Alavi A et al. The role of imaging techniques to define a peri-prosthetic hip and knee joint infection: multidisciplinary consensus statements. J Clin Med 2020;9(8). https://doi.org/10.3390/jcm9082548.
Sollini M, Lauri C, Boni R, Lazzeri E, Erba PA, Signore A. Current status of molecular imaging in infections. Curr Pharm Des. 2018;24(7):754–71. https://doi.org/10.2174/1381612824666180110103348.
Loharkar S, Basu S. PET-computed tomography in bone and joint infections. PET Clin. 2023;18(1):49–69. https://doi.org/10.1016/j.cpet.2022.08.002.
Falstie-Jensen T, Daugaard H, Søballe K, Ovesen J, Arveschoug AK, Lange J. Labeled white blood cell/bone marrow single-photon emission computed tomography with computed tomography fails in diagnosing chronic periprosthetic shoulder joint infection. J Shoulder Elbow Surg. 2019;28(6):1040–8. https://doi.org/10.1016/j.jse.2018.10.024.
Falstie-Jensen T, Lange J, Daugaard H, Vendelbo MH, Sørensen AK, Zerahn B, et al. 18F FDG-PET/CT has poor diagnostic accuracy in diagnosing shoulder PJI. Eur J Nucl Med Mol Imaging. 2019;46(10):2013–22. https://doi.org/10.1007/s00259-019-04381-w.
Signore A. The need of shared diagnostic protocols. Quart J Nucl Med Mol Imag Off Publ Ital Assoc Nucl Med (AIMN) Int Assoc Radiopharmacol (IAR) Sect So. 2014;58(1):1.
Singh SB, Bhandari S, Siwakoti S, Bhatta R, Raynor WY, Werner TJ et al. Is imaging bacteria with PET a realistic option or an illusion? Diagnostics (Basel, Switzerland). 2023;13(7). https://doi.org/10.3390/diagnostics13071231.
Signore A, Artiko V, Conserva M, Ferro-Flores G, Welling MM, Jain SK et al. Imaging bacteria with radiolabelled probes: is it feasible? J Clin Med. 2020;9(8). https://doi.org/10.3390/jcm9082372.
Hess S, Alavi A, Werner T, Høilund-Carlsen PF. Molecular imaging of bacteria in patients is an attractive Fata Morgana, not a realistic option. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2018;59(4):716–7. https://doi.org/10.2967/jnumed.117.207001.
Marjanovic-Painter B, Kleynhans J, Zeevaart JR, Rohwer E, Ebenhan T. A decade of ubiquicidin development for PET imaging of infection: a systematic review. Nucl Med Biol. 2023;116–117:108307. https://doi.org/10.1016/j.nucmedbio.2022.11.001.
Auletta S, Galli F, Lauri C, Martinelli D, Santino I, Signore A. Imaging bacteria with radiolabelled quinolones, cephalosporins and siderophores for imaging infection: a systematic review. Clin Transl Imaging. 2016;4:229–52. https://doi.org/10.1007/s40336-016-0185-8.
• Auletta S, Varani M, Horvat R, Galli F, Signore A, Hess S. PET radiopharmaceuticals for specific bacteria imaging: a systematic review. Journal of clinical medicine. 2019;8(2). https://doi.org/10.3390/jcm8020197. A comprehensive overview of potential novel bacteria-specific PET-tracers based in a systematic review of the literature.
Rousset O, Rahmim A, Alavi A, Zaidi H. Partial volume correction strategies in PET. PET Clin. 2007;2(2):235–49. https://doi.org/10.1016/j.cpet.2007.10.005.
Alavi A, Werner TJ, Høilund-Carlsen PF. What can be and what cannot be accomplished with PET to detect and characterize atherosclerotic plaques. J Nucl Cardiol: Off Publ Am Soc Nucl Cardiol. 2018;25(6):2012–5. https://doi.org/10.1007/s12350-017-0977-x.
Slart R, Tsoumpas C, Glaudemans A, Noordzij W, Willemsen ATM, Borra RJH, et al. Long axial field of view PET scanners: a road map to implementation and new possibilities. Eur J Nucl Med Mol Imaging. 2021;48(13):4236–45. https://doi.org/10.1007/s00259-021-05461-6.
van Sluis J, Borra R, Tsoumpas C, van Snick JH, Roya M, Ten Hove D, et al. Extending the clinical capabilities of short- and long-lived positron-emitting radionuclides through high sensitivity PET/CT. Cancer Imag: Off Publ Int Cancer Imag Soc. 2022;22(1):69. https://doi.org/10.1186/s40644-022-00507-w.
Glaudemans A, Gheysens O. Expert opinions in nuclear medicine: finding the “holy grail” in infection imaging. Front Med (Lausanne). 2023;10:1149925. https://doi.org/10.3389/fmed.2023.1149925.
Author information
Authors and Affiliations
Contributions
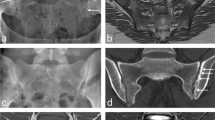
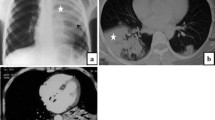
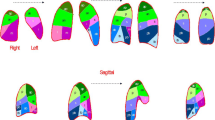
All authors contributed to conceptualizing this work and contributed individual sections of the main text. K.R. prepared figures 1 & 2. All authors reviewed the manuscript and approved the final version.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thorsen, M.E., Singh, S.B., Rewers, K. et al. Molecular Imaging Techniques in the Diagnosis and Monitoring of Infectious Diseases. Curr Treat Options Infect Dis (2024). https://doi.org/10.1007/s40506-024-00274-1
Accepted:
Published:
DOI: https://doi.org/10.1007/s40506-024-00274-1