Abstract
The t(9;22) (BCR/ABL) and t(4;11) (AF4/MLL) positivity suggests a poor prognosis in B-acute lymphoblastic leukemia (ALL). Many western studies have highlighted the frequency and profile of BCR–ABL-positive B-ALL; however, data from the Indian subcontinent is limited. The aim of the pilot study is to present the complete clinico-hematological, immunophenotyping, and molecular profile of BCR–ABL-positive B-ALL. It is a prospective study involving a total of 15 BCR–ABL-positive B-ALL cases diagnosed by bone marrow examination, immunophenotyping, and multiplex reverse transcriptase polymerase chain reaction assay in our institute. Out of the total 15 cases enrolled in the study, ten (67 %) were adult and five (33 %), pediatric. All cases had splenomegaly, hepatomegaly, high WBC count (>20 × 109/L), and low platelet count (<100 × 109/L) at presentation. Twelve of 15 (80 %) showed aberrant myeloid marker expression, of which 11 showed expression of two myeloid antigens (CD13 and CD117/33). The BCR–ABL transcript type in our study was p210 seen in 8/15 (53.3 %) cases, while p190 in 7/15 (46.6 %) cases. Thirteen of 15 (87 %) cases received imatinib in addition to the ALL induction regimen. Ten of 13 (77 %) of these had complete hematological remission at first check marrow. The disease-free interval, however, ranged from 4–6 months in 6/15 (40 %) cases. Analysis for BCR–ABL transcript is necessary in B-ALL as the cases may benefit from addition of imatinib to the treatment regimen.
Similar content being viewed by others
Introduction
Acute lymphoblastic leukemia (ALL) is a neoplastic disease that results from multistep somatic mutations in a single lymphoid progenitor cell at one of the several discrete stages of development. Although diagnosis and classification of ALL is generally based on the morphologic, cytochemical, and immunologic features of the blasts, cytogenetic and molecular studies are frequently needed to confirm the diagnosis, predict clinical behavior, and stratify patients for therapy. In the present WHO 2008 classification for B-cell precursor ALL, t(9;22) BCR–ABL and t(4;11) AF4–MLL transcripts carry a poor prognosis, and it is considered mandatory to test for these recurrent genetic abnormalities in B-ALL cases by fluorescent in situ hybridization or reverse transcriptase polymerase chain reaction assay (RT-PCR).
The incidence of BCR–ABL positivity in adult ALL cases ranges from 20 to 25 %, while in children it is 3–5 %. It is further seen that the p190 product of BCR–ABL is noted in around 70 % in adult ALLs and 95 % in pediatric ALLs, while the p210 (b3a2/b2a2) product is seen in 28–30 % in adult ALLs and 3–5 % in pediatric ALLs [1]. The atypical BCR–ABL transcripts, i.e., e1a3, e13a3, and e6a2 are noted in only 2 % ALLs [1].
A recent paper by the authors on the incidence of common chimeric fusion transcripts in B-ALL has been accepted for publication [2]. The present study is an extension of a part of the same and focuses on the complete profile of BCR–ABL-positive B-ALLs, as there are very few studies from our subcontinent highlighting the frequency and characteristics of BCR–ABL-positive B-ALLs.
Materials and methods
A prospective analysis of BCR–ABL-positive B-ALLs diagnosed in the department of hematology of a tertiary care research institute of northern India, over a period of 1.5 years (May 2010–Oct 2011), was conducted. Bone marrow examination, cytochemistry, immunophenotyping, and multiplex RT-PCR for common BCR–ABL transcripts (p190 and p210-b3a2, b2a2) were carried out in all cases. For multiplex RT-PCR, RNA was first extracted from 1–2-ml bone marrow aspirate in EDTA using a commercial kit (Qiagen Miniamp RNA Blood kit) according to manufacturer’s instructions. The RNA quality was checked in each case by absorbance at 260 nm in a spectrophotometer and by running a 1 % formaldehyde gel for 18 s and 28 s RNA bands. This was followed by cDNA synthesis according to the commercial kit protocol (Fermentas cDNA kit). The quality of cDNA was checked using primers for β-Actin housekeeping gene. Multiplex RT-PCR was finally carried out using primers specific for p210 (b3a2 and b2a2) and p190 transcripts of BCR–ABL as per the study of Jones et al. [3]. This protocol has been standardized in our laboratory since 2004 for detection of BCR–ABL transcripts in CML cases. The primer sequences and the product base sizes are highlighted in the Table 1. The multiplex RT-PCR was carried out in the following conditions: pre-denaturation, 94 °C, 4 min, 1 cycle; denaturation, 94 °C, 1 min; annealing, 63 °C, 2 min; and extension, 72 °C, 3 min for 32 cycles. This was followed by one cycle of final extension at 72 °C for 10 min. The PCR products were run on agarose gel and stained with ethidium bromide and visualized under UV-Gel doc for b3a2-p210 (385 bp), b2a2-p210 (310 bp), and e1a2-p190 (481 bp) bands. A sample for cytogenetic analysis was also taken at the initial diagnostic work-up. Giemsa-stained bone marrow and peripheral blood slides were also reviewed at first check marrow (day 14 for pediatric cases and day 28 for adult cases) for remission status (standard remission criteria: Hb > 10 g/dl; TLC 4–12 × 109/L; platelet 150–450 × 109/L; bone marrow blasts <5 %; no Auer rod; and no blasts in peripheral blood), and follow-up data wherever available was analyzed. All cases with long history of symptoms at presentation or massive splenomegaly and/or known cases of CML with lymphoid blast crisis were excluded from the study.
Ethical justification
Prior informed consent was taken from all patients/guardians before withdrawal of sample. The bone marrow or peripheral blood sample taken during the study period was a part of the routine diagnostic tap for acute leukemia cases as per the institute protocol.
Results
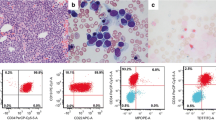
Multiplex RT-PCR was carried out in a total of 120 cases of B-ALL over the study period. The frequency of BCR–ABL transcript positivity was 15/120 (12.5 %). Of the 15 positive cases, ten (66.6 %) were adults and five (33.3 %) were pediatric. The M/F ratio was 2.3:1 for adults, while all five pediatric cases were males. The age ranged from 18–49 years (median age, 40 years) in adults and 0.9–11 years (median age, 7 years) in pediatrics. The clinico-hematological features and aberrant immunophenotype profile of all 15 cases are highlighted in the Table 2. Fifteen of 15 (100 %) cases had hepatosplenomegaly at presentation, while 3/5 (60 %) pediatric cases also had generalized lymphadenopathy. All positive cases had high WBC count (>20 × 109/L) and low platelet count (<100 × 109/L). Twelve of 15 (80 %) cases expressed two or more aberrant myeloid antigens on immunophenotyping, with 9/12 (75 %) expressing CD13 and CD 33 both, 2/12(17 %) CD13 and CD117, and 1/12 (8 %) CD13, 33, and 117. Figures 1 and 2 highlight a case of BCR–ABL-positive ALL with blast morphology on aspirate smears and the characteristic immunophenotype profile, respectively. Cytogenetic analysis was conclusive in 5/15 (33.3 %) cases at the initial work-up, while six were positive with a repeat peripheral blood sample taken at 2–3 weeks post-diagnosis when TLC was <25 × 109/L. In the rest of the cases, metaphases were inconclusive for diagnosis. No other cytogenetic abnormality was reported in any of the positive cases. The predominant BCR–ABL transcript in our study was p210 seen in 8/15 (53.3 %) cases, while p190, in 7/15 (46.6 %) cases. Table 3 highlights the BCR–ABL transcript-type in adult and pediatric cases, and Figs. 3 and 4 shows the multiplex RT-PCR gels with positivity for p210 and p190 products.
Thirteen of 15 (87 %) cases received imatinib in addition to the ALL induction regimen (at a dose of 300 mg/day). Ten of 13 (77 %) cases had complete hematological remission (CHR) at day 28/35 check marrow for adults and day 14 for pediatric cases. Of the 10 positive adult cases, nine received imatinib therapy. Six of the nine (66.6 %) had CHR at day 28 marrow, two were not in remission, of which one went into CHR at 4 months, and one was in partial remission (PR) at day 28. At post-induction (8 weeks), 4/6 were still in CHR, while two relapsed due to CNS disease. Five of 10 (50 %) adult cases were lost to follow-up, of which two were due to disease relapse at 2 months, two had persistent nonresponsive disease, and one in PR, who left treatment at 6 weeks of induction. Five of 10 (50 %) adult cases on imatinib therapy are still on follow-up with a median follow-up period of 6 months. Of the five positive pediatric cases, four received imatinib therapy. Three of four (75 %) showed rapid early response with CHR at day 14, while one had slow early response (SER) with CHR at day 35 check marrow. At post-induction, 3/4 were still in CHR, while one with SER had relapsed. However, out of the three cases in CHR post-induction, two were lost to follow-up, of which one was due to disease relapse at 5 months, another one was due to febrile neutropenia at 4 months, and one died at 6 weeks of induction therapy due to staph sepsis. One case did not receive treatment at the institute, while one with SER and relapse is on regular follow-up at 4 months post-induction.
Discussion
Of the 120 ALL cases evaluated for BCR–ABL expression during the study period, 76/120 (63 %) were pediatric and 44/120 (37 %) were adult cases. The frequency of BCR–ABL positivity in adults was 10/44 (22.7 %) and in pediatrics, 5/76 (6.5 %). This is in accordance with most of the studies reported from our subcontinent as well as few studies from the West [4–12]. Clinico-hematological features associated with BCR–ABL-positive ALL include high WBC count, older age, male predominance, and FAB L2 blast morphology. In the present study, too, male preponderance was noted, and all positive cases presented with high WBC count. In addition, low platelet count was also noted in all positive cases, with counts <50 × 109/L seen in 12/15 (80 %) cases.
Many studies have outlined specific immunophenotype patterns for the recurrent genotypic abnormalities seen in B cell ALL [13, 14]. ALL cases carrying BCR/ABL transcripts are CD45neg/dim, CD34pos/neg, CD10bright, CD15neg, CD13 pos, and CD33 positive. In our study, aberrant myeloid antigen expression (CD 13 and 33) was noted in 12/15 (80 %) BCR–ABL-positive cases which is in concordance with the study by Hrus et al. [13]. Bright positivity for CD 10 was noted in 13/15 (87 %) cases, one case was dim positive for CD10 and one was negative.
Contrary to literature review, p210 was the more common transcript subtype in positive cases in 8/15 (53 %) as compared to p190 transcript 7/15 (47 %). The frequency of p210 transcript in adult cases was 5/10 (50 %) and in pediatric cases, it was 3/5 (60 %), while that of p190 was 5/10 (50 %) in adults and 2/5 (40 %) in pediatric age group. Indeed, two studies [15, 16], based on retrospective analysis, have reported a higher percentage of adult ALL cases with p210 positivity as compared to p190 transcript. Kantarjian et al. [17] found the p190 type of breakpoint in 12 out of 24 adult ALL patients and the p210 in 11 patients, which is quiet comparable to the frequency noted in our study. In a study by Li Hui-yu et al. [7], BCR–ABL positivity in ALL was 31.5 %, and all of their nine positive cases had p210 transcript.
A prospective study [18] incorporating imatinib with the hyper-CVAD (cyclophosphamide, vincristine, infusional doxorubicin [Adriamycin], and dexamethasone) regimen in BCR–ABL-positive ALL has shown higher response rates and excellent tolerability, thereby substantially improving event-free survival. In our center, BCR–ABL-positive ALLs are treated with a combination of imatinib and chemotherapy (modified BFM protocol in adults and modified UK MRC ALL 2003 protocol in children). Imatinib is started at a dose of 300 mg/day and continued into consolidation and maintenance phase. Several studies [18–21] have reported increased CHR with the addition of imatinib to chemotherapy regimen. Our study results also show a high CHR rate of 77 % (10/13) with use of imatinib in induction regimen. However, sustained remission was observed in only 5/10 (50 %) cases, with three having early relapse in consolidation phase, one dying of staph sepsis, and one developing febrile neutropenia. This poor response was more pronounced in the pediatric group as, despite 3/4 cases achieving rapid early response at day 14 check marrow, none had sustained remission. None of the cases with relapse or partial response underwent allogeneic hematopoietic stem cell transplant and were lost to follow-up. The number of cases enrolled in our study is few, and the follow-up period was very short to make any conclusive statements.
Conclusion
Identification of BCR–ABL transcript is important, especially in B-ALL cases as a significant proportion of them are positive and fair poorly with standard chemotherapy regimens. Multiplex RT-PCR is a sensitive screening assay and should be used to detect both the common BCR–ABL transcript types (p210 and p190). Future prospective studies addressing patient survival data are needed to comprehensively assess the role of imatinib and chemotherapy combination in our cohort of patients.
References
Burmeister T, Schwartz S, Taubald A et al (2007) Atypical BCR-ABL mRNA transcripts in adult acute lymphoblastic leukemia. Hematologica 92(12):1699–1702
Bhatia P, Binota J, Varma N, Bansal D, Trehan A, Marwaha RK, Malhotra P, Varma S (2012) Incidence of common chimeric fusion transcripts in B-cell acute lymphoblastic leukemia: an Indian perspective. Acta Haematol. doi:10.1159/000338260
Jones CD, Yeung C, Zehnder JL (2003) Comprehensive validation of a real-time quantitative BCR-ABL assay for clinical laboratory use. Am J Clin Pathol 120:42–48
Pakakasama S, Kajanachumpol S, Kanjanapongkul S et al (2008) Simple multiplex RT-PCR for identifying common fusion transcripts in childhood acute leukemia. Int J Lab Hemat 30:286–291
Siraj AK, Ozbek U, Sazawal S et al (2002) Preclinical validation of a monochrome real-time multiplex assay for translocations in childhood acute lymphoblastic leukemia. Clin Cancer Res 8:3832–3840
Ibrahim K, Daud SS, Seah Y-L, Yeoh AE, Ariffin H (2008) Rapid detection of prognostically important childhood acute lymphoblastic leukemia chimeric transcripts using multiplex SYBR green real-time reverse transcription PCR. Ann Clin Lab Sci 38(4):338–343
Sazawal S, Bhatia K, Gutierrez MI, Saxena R, Arya LS, Bhargava M (2004) Paucity of TEL-AML 1 translocation, by Multiplex RT-PCR, in B-Lineage acute lymphoblastic leukemia (ALL) in Indian patients. Am J Hematol 76:80–82
Siraj AK, Kamat S, Gutierrez MI et al (2003) Frequencies of the major subgroups of precursor B-cell acute lymphoblastic leukemia in Indian children differ from the West. Leukemia 17:1192–1193
Hui-yu L, Guo-Lin Z, Ping Z (2001) The study on BCR/ABL fusion gene in adult acute lymphoblastic leukemia. Wuhan Univ J Nat Sci 6(3):742–746
Preudhomme C, Fenaux P, Lal JL et al (1993) Philadelphia negative, BCR-ABL positive adult acute lymphoblastic leukemia (ALL) in 2 of 39 patients with combined cytogenetic and molecular analysis. Leukemia 7(7):1054–1057
Radich J, Gehly G, Lee A et al (1997) Detection of bcr-abl transcripts in Philadelphia chromosome-positive acute lymphoblastic leukemia after bone marrow transplantation. Blood 89:2602–2609
Burmeister T, Schwartz S, Bartram CR, Gökbuget N, Hoelzer D, Thiel E (2008) Patients’ age and BCR-ABL frequency in adult B-precursor ALL: a retrospective analysis from the GMALL study group. Blood 112(3):918–919
Hruša’k O, Porwit-MacDonald A (2002) Antigen expression patterns reflecting genotype of acute leukemias. Leukemia 16:1233–1258
Tien HF, Wang CH, Lee FY et al (1992) Cytogenetic study of acute lymphoblastic leukemia and its correlation with immunophenotype and genotype. Cancer Genet Cytogenet 59(2):191–198
Annino L, Ferrari A, Cedrone M et al (1994) Adult Philadelphia-chromosome-positive acute lymphoblastic leukemia: experience of treatments during a ten-year period. Leukemia 8:664–667
Radich JP, Kopecky KJ, Boldt DH, Head et al (1994) Detection of BCR-ABL fusion genes in adult acute lymphoblastic leukemia by the polymerase chain reaction. Leukemia 8:1688–1695
Kantarjian HM, Talpaz M, Dhingra K et al (1991) Significance of the P210 versus P190 molecular abnormalities in adults with Philadelphia chromosome-positive acute leukemia. Blood 78:2411–2418
Thomas DA, Faderl S, Cortes J et al (2004) Treatment of Philadelphia chromosome-positive acute lymphoblastic leukemia with hyper-CVAD and imatinib mesylate. Blood 103:4396–4407
Towatari M, Yanada M, Usui N et al (2004) Combination of intensive chemotherapy and imatinib can rapidly induce high-quality complete remission for a majority of patients with newly diagnosed BCR-ABL-positive acute lymphoblastic leukemia. Blood 104:3507–3512
Wassmann B, Pfeifer H, Goekbuget N et al (2006) Alternating versus concurrent schedules of imatinib and chemotherapy as front-line therapy for Philadelphia-positive acute lymphoblastic leukemia (Ph + ALL). Blood 108(5):1469–1477
de Labarthe A, Rousselot P, Huguet-Rigal F et al (2007) Imatinib combined with induction or consolidation chemotherapy in patients with de novo Philadelphia chromosome positive acute lymphoblastic leukemia: results of the GRAAPH-2003 study. Blood 109(4):1408–1413
Conflict of interest
The authors declare they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhatia, P., Binota, J., Varma, N. et al. Clinico-hematological, immunophenotyping, and molecular transcript profile of BCR–ABL-positive B cell acute lymphoblastic leukemias. J Hematopathol 6, 19–24 (2013). https://doi.org/10.1007/s12308-012-0171-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12308-012-0171-7