Abstract
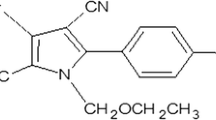
Persistence of fluopicolide and propamocarb in tomato was studied following three applications of a combination formulation of Infinito 68.75 SC (fluopicolide 6.25 % + propamocarb 62.5 %) at 1500 and 3000 mL ha−1 by 7 days interval, starting the spray at fruit development stage. QuEChERS method included extraction of sample with ethyl acetate and cleanup of dispersive solid-phase extraction was used for the determination of fluopicolide and propamocarb residues on tomato and soil. Residues of fluopicolide and propamocarb in tomato were estimated by gas–liquid chromatography and gas chromatograph–mass spectrometry, respectively. Half-lives for fluopicolide were found to be 2.58 and 2.31 days, whereas for propamocarb these values were observed to be 1.49 and 2.08 days at single and double the application rates, respectively. Residues of fluopicolide dissipated below its limit of quantification (LOQ) of 0.01 mg kg−1 after 7 and 10 days at single and double the application dosage, respectively. Similarly, residues of propamocarb took 5 and 7 days to reach LOQ of 0.10 mg kg−1, at single and double dosages, respectively. Soil samples collected after 15 days of the last application did not show the presence of fluopicolide and propamocarb at their detection limit of 0.01 and 0.10 mg kg−1, respectively. The initial deposit of residues of propamocarb and fluopicolide for both the dosages were below the prescribed codex maximum residue limit values of 2 and 1 mg kg−1, respectively. Therefore, a 1-day waiting period was suggested to reduce human health risks before consumption of tomato fruits.





Similar content being viewed by others
References
Al-Rahman SHA, Almaz MM, Ahmed NS (2012) Dissipation of fungicides, insecticides and acaricide in tomato using HPLC-DAD and QuEChERS methodology. Food Anal Methods 5:564–570
Anastassiades M, Lehotay SJ, Stajnbaher D, Schenck FJ (2003) Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J AOAC Int 86:412–431
Anastassiades M, Tasdelen B, Scherbaum E, Stajnbaher D (2007) Recent developments in QuEChERS methodology for pesticide multiresidue analysis. In: Ohkawa H, Miyagawa H, Lee PW (eds) Pesticide chemistry: crop protection, public health, environmental safety. Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Anonymous (2008) Package of practices for cultivation of vegetables. In: Malhi NS, Mahindra K. (eds) Punjab Agricultural University, Ludhiana, India, p 38–41
Arienzo M, Cataldo D, Ferrara L (2013) Pesticide residues in fresh-cut vegetable from integrated pest management by ultra performance liquid chromatography coupled to tandem mass spectrometry. Food Control 31:108–115
Aysal P, Ambrus A’, Lehotay SJ, Cannavan A (2007) Validation of an efficient method for the determination of pesticide residues in fruits and vegetables using ethyl acetate for extraction. J Environ Sci Hlth B 42:481–490
Bardsley E, Wegener M, Tafforeau S (2006) Field development of Infinito for late blight control in potatoes. Pflanzenschutz-Nachr Bayer 59:281–291
Bardsley RA, Shattock RC, Day JP (1996) Studies investigating the sensitivity of Phytophthora infestans to propamocarb hydrochloride. Brighton Crop Prot. Conf.—Pests and diseases, Alton, UK. BCPC Publications. p 719–724
Bardsley RA, Shattock RC, Day JP (1998) Studies comparing the sensitivity of European and USA isolates of Phytophthora infestans to propamocarb hydrochloride. Brighton Crop Prot. Conf. – Pests and diseases, Alton, UK. BCPC. P 523–528
Bates R (2002) Pesticide residues and risk assessment. Pestic Outlook 13:142–145
Bayer CropScience (2011) http://www.bayercropscience.co.uk/assets/Document-Store/0251-BCS-Product-Manual-2011.pdf available online
Causon R (1997) Validation of chromatographic methods in biomedical analysis viewpoint and discussion. J Chromatogr B 689:175–180
Charalampos KM, Zisis V, Euphemia PM (2012) Biodegradation of soil applied pesticides by selected strains of plant growth-promoting rhizobacteria (PGPR) and their effects on bacterial growth. Biodegradation 23:297–310
Codex Alimentarius Commission (2007) Guide to Codex Maximum Limits for Pesticide Residues. Food and Agricultural Organization, Rome. (http://www.codexalimentarius.net/pestres/data/pesticides/ details.html?id=148)
Codex Alimentarius Commission (2010) Guide to Codex Maximum Limits for Pesticide Residues. Food and Agricultural Organization, Rome. (http://www.codexalimentarius.net/pestres/data/pesticides/details.html;jsessionid=FEB5E2641084D2820077251DC785C430?d-16497-o=2&id=235 &d-16497-s=3)
Cohen Y, Coffey MD (1986) Systemic fungicides and control of oomycetes. Annu Rev Phytopathol 24:311–318
Cooke LR, Little G (2006) Evaluation of fluopicolide-containing formulations for the control of potato late blight in Northern Ireland. Pflanzenschutz-Nachr-Bayer 59:303–316
Diez C, Traag WA, Zommer P, Marinero P, Atienza J (2006) Comparison of an acetonitrile extraction/partitioning and “dispersive solid-phase extraction” method with classical multi-residue methods for the extraction of herbicide residues in barley samples. J Chromatogr A 1131:11–23
Englander L, Merlino JA, McGuire JJ (1980) Efficacy of two new systemic fungicides and etazole for control of Phytophthora root rot of rhododendron, and spread of Phtophthora cinnamomi in propagation benches. Phytopathology 70:1175–1179
EPA (2013) Food safety http://www.epa.gov/agriculture/tfsy.html available online March 14, 2013
FAO (2009) Joint meeting on pesticide residues: fluopicolide, pesticide residues in food. FAO plant prod protect pap 235:350–406
Francotte E, Davatz A, Richert P (1996) Development and validation of chiral high-performance liquid chromatographic methods for the quantitation of valsartan and of the tosylate of valinebenzyl ester. J Chromatogr B 686:77–83
Gouot LM (2006) Field efficacy of Profiler, a fluopicolide and fosetyl-Al fungicide combination for the control of grape downey mildew (Plasmopara viticola). Pflanzenschutz-Nachric Bayer 59:293–302
Hoskins WM (1966) Mathematical treatment of the rate of loss of pesticide residues. FAO Plant Protect Bull 9:163–168
Latorse MP, Holah D, Bardsley R (2006) Fungicidal properties of fluopicolide-based products. Pflanzenschutz-Nachric Bayer 59:185–200
May LL, Kimati H (2000) Controle de Phtophthora parasitica com fungicide e efeito desses produtos no crescimento micelial de Trichoderma. Sum Phytopathol 26:52–57
Mohapatra S, Ahuja AK, Deepa M, Jagdish GK, Rashmi N, Kumar S, Prakash GS (2011) Persistence and dissipation of fluopicolide in/on grape berries and soil under semi arid tropical climatic conditions of India. Bull Environ Contam Toxicol 86:238–241
Papavizas GC, O’Neill NR, Lewis JA (1978) Fungistatic activity of propyl-N-(adimethylaminopropyl)-carbamate on Pythium spp., and its reversal by strols. Phytopathology 68:1667–1671
Pieroh EA, Krass W, Hemmen C (1978) Propamocarb, einneues Fungizid zur Abwehr von Oomyceten im Zierpflaanzen-und Gemusebau. Meded Fac Landbouw Rijksuniv Gent 43:933–942
Rapp L, Richter J (1982) Effect of propamocarb-hydrochloride (Previcur N) on several isolates of some Pythium and Phtophthora species in vitro and in vivo. J Plant Dis Prot 43:933–942
Reilly JJ (1980) Chemical control of black shank of tobacco. Plant Dis 64:274–277
Rupprecht JK (2006) Behaviour of flupicolide in plants. Pflanzenschutz-Nachric Bayer 59:261–280
Sahoo SK, Battu RS, Singh B (2011) Development and validation of QuEChERS method for estimation of propamocarb residues in tomato (Lycopersicon esculentum Mill) and soil. Am J Anal Chem 2:26-31
Tomlin CDS (2000) The pesticide manual, 12th edn. British Crop Protection Council, Bracknell, 769
Toquin V, Barja F, Sirven C, Gamet S, Latorse MP, Zundel JL, Schmitt F, Beffa R (2006) A new mode of action for fluopicolide: modification of the cellular localization of a spectrin-like protein. Pflanzenschutz-Nachric Bayer 59:171–184
Wilde TH (1990) Propamocarb-HCl, a fungicide suitable for integrated pest management. Tomato and Pepper Production in the Tropics. Proc. Intl. Sympos. Integrated Management Practices, Taiwan, p 306.
Acknowledgments
The authors are thankful to Indian Council of Agricultural Research, India for sponsoring the project and also to the Head, Department of Entomology, Punjab Agricultural University, Ludhiana, Punjab, India, for providing necessary research facilities.
Conflict of Interest
Sanjay Kumar Sahoo has no conflict of interest. Kousik Mandal has no conflict of interest. Rajinder Kumar has no conflict of interest. Balwinder Singh has no conflict of interest. This article does not contain any studies with human or animal subjects.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sahoo, S.K., Mandal, K., Kumar, R. et al. Analysis of Fluopicolide and Propamocarb Residues on Tomato and Soil Using QuEChERS Sample Preparation Method in Combination with GLC and GCMS. Food Anal. Methods 7, 1032–1042 (2014). https://doi.org/10.1007/s12161-013-9709-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-013-9709-2