Abstract
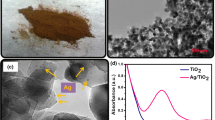
Silver nanoparticles were prepared by chemical reduction of acetaldehyde gas in the absence of protective gas, and Ag/FePO4 nanocomposites were synthesised by modified silver mirror reaction at a gas-liquid interface. A hydrogen peroxide (H2O2) electrochemical sensor was constructed through immobilizing Ag/FePO4 nanocomposites on gold (Au) electrode. The morphology and composition of the nanocomposites were characterized by transmission electron microscopy (TEM) and energy-dispersive X-ray spectroscopy (EDS). The electrochemical investigations of the sensor indicated that it exhibited excellent analytical performance with a wide linear range from 3.0×10−5 to 1.1×10−2 mol⋅L−1 and a low detection limit of 4.7 μmol⋅L −1 at a signal-to-noise ratio of 3. Meanwhile, it also showed acceptable reproducibility and anti-interference ability. This study may provide a new method for the synthesis of highly dispersed metal nanoparticles which might be used in other related fields.

The process for preparing FePO4 nanocomposites decorated with Ag NPs is described. An H2O2 electrochemical sensor was fabricated by immobilizing Ag/FePO4 nanocomposites on gold electrode. The electrochemical investigations for this sensor exhibited excellent H2O2 sensing performance.









Similar content being viewed by others
References
Sanderson W R 2000 Pure. Appl. Chem. 72 1289
Barnard J P and Stinson M W 1999 Infect. Immun. 67 6558
Karthega M, Nagarajan S and Rajendran N 2010 Electrochim. Acta 55 2201
Klassen N V, Marchington D and McGowan H C E 1994 Anal. Chem. 66 2921
Gao Y, Wang G N, Huang H, Hu J J, Shah S M and Su X G 2011 Talanta 85 1075
Steinberg S M 2013 Environ. Monit. Assess. 185 3749
Hoshino M, Kamino S, Doi M, Takada S, Mitani S, Yanagihara R and Fujita Y 2014 Spectrochim. Acta A 117 814
Shi W B, Zhang X D, He S H and Huang Y M 2011 Chem. Commun. 47 10785
Liu M M, Liu R and Chen W 2013 Biosens. Bioelectron. 45 206
Abdulrahman O 2013 Nanoscale 5 8921
Zhang J and Zheng J B 2015 Anal. Methods 7 1788
Mase K, Ohkubo K and Fukuzumi S 2013 J. Am. Chem. Soc. 135 2800
Welch C M, Banks C E, Simm A O and Compton R G 2005 Anal. Bioanal. Chem. 382 12
Long L H, Hoi A and Halliwell B 2010 Arch. Biochem. Biophys. 501 162
Landon P, Collier P J, Papworth A J, Kiely C J and Hutchings G J 2002 Chem. Commun. 18 2058
Zhang J D and Oyama M 2005 J. Electroanal. Chem. 577 273
Jiang F X, Yue R R, Du Y K, Xu J K and Yang P 2013 Biosens. Bioelectron. 44 127
Luo B B, Li X M, Yang J C, Li X L, Xue L P, Li X L, Gu J K, Wang M Z and Jiang L 2014 Anal. Methods 6 1114
Jia F F, Zhong H, Zhu F X, Li X H, Wang Y Z, Cheng Z P, Zhang L L, Sheng Z H and Guo L P 2014 Electroanal. 26 2244
Yadav D K, Gupta R, Ganesan V, Sonkar P K and Rastogi P K 2016 J. Appl. Electrochem. 46 103
Michaels A M, Jiang J and Brus L 2000 J. Phys. Chem. B 104 11965
Iga M, Seki A and Watanabe K 2004 Sens. Actuators, B 101 368
Bai W S, Nie F, Zheng J B and Sheng Q L 2014 ACS Appl. Mater. Inter. 6 5439
Huang H, Yin S C and Nazar L F 2001 Electrochem. Solid-State Lett. 4 A170
Lee J G, Kim B, Cho J, Kin Y W and Park B 2004 J. Electrochem. Soc. 151 A801
Prosini P P, Lisi M, Scaccia S, Carewska M, Cardellini F and Pasquali M 2002 J. Electrochem. Soc. 149 A297
Qing C B, Bai Y, Yang J M and Zhang W F 2011 Electrochim. Acta 56 6612
Laffont L, Delacourt C, Gibot P, Wu M Y, Kooyman P, Masquelier C and Tarascon J M 2006 Chem. Mater. 18 5520
Song Y N, Yang S F, Zavalij P Y and Whittingham M S 2002 Mater. Res. Bull. 37 1249
Okada S, Yamamoto T, Okazaki Y, Yamaki J I, Tokunaga M and Nishida T 2005 J. Power Sources 146 570
Shi Z C, Li Y X, Ye W L and Yang Y 2005 Electrochem. Solid-State Lett. 8 A396
Li M J, Wu Z L, Ma Z, Schwartz V, Mullins D R, Dai S and Overbury S H 2009 J. Catal. 266 98
Yin Y J, Wu P, Zhang H and Cai C X 2012 Electrochem. Commun. 18 1
Chen D, Wang G, Lu W, Zhang H and Li J H 2007 Electrochem. Commun. 9 2151
Masataka H, Takuro K and Hideaki K 1986 Electrochim. Acta 31 377
Xu J, Huang W H and McCreery R L 1996 Electroanal. Chem. 410 235
Lu W B, Liao F, Luo Y L, Chang G H and Sun X P 2011 Electrochim. Acta 56 2295
Zhao W, Wang H C, Qin X, Wang X S, Zhao Z X, Miao Z Y, Chen L L, Shan M M, Fang Y X and Chen Q 2009 Talanta 80 1029
Hocevar S B, Ogorevc B, Schachl K and Kalcher K 2004 Electroanalysis 16 1711
Luo Y L, Lu W B, Chang G H, Liao F and Sun X P 2011 Electrochim. Acta 56 8371
Liu S, Tian J Q, Wang L, Li H Y, Zhang Y W and Sun X P 2010 Macromolecus 43 10078
Acknowledgements
The authors gratefully acknowledge the financial support of this project by the National Science Fund of China (No. 21275116), Specialized Research Fund for the Doctoral Program of Higher Education (No. 20126101120023), the Natural Science Fund of Shaanxi Province in China (No. 2012JM2013, 2013KJXX-25), the Fund of Shaanxi Province Educational Committee of China (No. 12JK0576), the Scientific Research Foundation of Shaanxi Provincial Key Laboratory (2010JS088, 11JS080, 12JS087, 12JS088, 13JS097, 13JS098) and the Graduate Innovation Fund of Northwest University (No. YZZ12019).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information (SI)
The EDS spectrum of Ag/FePO4 nanocomposites (figure S1) is given in the supporting information available at www.ias.ac.in/chemsci.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
RAO, D., ZHANG, J. & ZHENG, J. Synthesis of silver nanoparticles-decorated FePO4 nanosphere at a gas-liquid interface for the electrochemical detection of Hydrogen peroxide. J Chem Sci 128, 839–847 (2016). https://doi.org/10.1007/s12039-016-1062-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-016-1062-8