Abstract
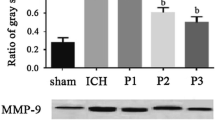
Our previous studies demonstrated that propofol protects rat brain against focal cerebral ischemia. However, whether propofol attenuates early brain injury after subarachnoid hemorrhage in rats remains unknown until now. The present study was performed to evaluate the effect of propofol on early brain injury after subarachnoid hemorrhage in rats and further explore the potential mechanisms. Sprague-Dawley rats underwent subarachnoid hemorrhage (SAH) by endovascular perforation then received treatment with propofol (10 or 50 mg/kg) or vehicle after 2 and 12 h of SAH. SAH grading, neurological scores, brain water content, Evans blue extravasation, the myeloperoxidase activity, and malondialdehyde (MDA) content were measured 24 h after SAH. Expression of nuclear factor erythroid-related factor 2 (Nrf2), nuclear factor-kappa B (NF-κB) p65, and aquaporin 4 (AQP4) expression in rat brain were detected by Western blot. Expression of cyclooxygenase-2 (COX-2) and matrix metalloproteinase-9 (MMP-9) were determined by reverse transcription-polymerase chain reaction (RT-PCR). Expressions of tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) were assessed by ELISA. Neurological scores, brain water content, Evans blue extravasation, the myeloperoxidase activity, and MDA content were significantly reduced by propofol. Furthermore, expression of Nrf2 in rat brain was upregulated by propofol, and expression of NF-κB p65, AQP4, COX-2, MMP-9, TNF-α, and IL-1β in rat brain were attenuated by propofol. Our results demonstrated that propofol improves neurological scores, reduces brain edema, blood-brain barrier (BBB) permeability, inflammatory reaction, and lipid peroxidation in rats of SAH. Propofol exerts neuroprotection against SAH-induced early brain injury, which might be associated with the inhibition of inflammation and lipid peroxidation.







Similar content being viewed by others
References
Altay O, Suzuki H, Hasegawa Y, Ostrowski RP, Tang J, Zhang JH (2014) Isoflurane on brain inflammation. Neurobiol Dis 62:365–371
Ayer RE, Zhang JH (2008) Oxidative stress in subarachnoid haemorrhage: significance in acute brain injury and vasospasm. Acta Neurochir Suppl 104:33–41
Caner B, Hou J, Altay O, Fujii M, Zhang JH (2012) Transition of research focus from vasospasm to early brain injury after subarachnoid hemorrhage. J Neurochem 123(Suppl 2):12–21
Chen G, Fang Q, Zhang J, Zhou D, Wang Z (2011) Role of the Nrf2-ARE pathway in early brain injury after experimental subarachnoid hemorrhage. J Neurosci Res 89(4):515–523
Cui D, Wang L, Qi A, Zhou Q, Zhang X, Jiang W (2012) Propofol prevents autophagic cell death following oxygen and glucose deprivation in PC12 cells and cerebral ischemia-reperfusion injury in rats. PLoS One 7(4), e35324
Ersahin M, Toklu HZ, Erzik C et al (2010) The anti-inflammatory and neuroprotective effects of ghrelin in subarachnoid hemorrhage-induced oxidative brain damage in rats. J Neurotrauma 27(6):1143–1155
Fujii M, Yan J, Rolland WB, Soejima Y, Caner B, Zhang JH (2013) Early brain injury, an evolving frontier in subarachnoid hemorrhage research. Transl Stroke Res 4(4):432–446
Garcia JH, Wagner S, Liu KF, Hu XJ (1995) Neurological deficit and extent of neuronal necrosis attributable to middle cerebral artery occlusion in rats. Statistical validation. Stroke 26(4):627–634, discussion 635
Ge M, Yao W, Wang Y et al (2015) Propofol alleviates liver oxidative stress via activating Nrf2 pathway. J Surg Res 196(2):373–381
Ji FT, Liang JJ, Miao LP, Wu Q, Cao MH (2015) Propofol postconditioning protects the blood brain barrier by decreasing matrix metalloproteinase9 and aquaporin4 expression and improves the neurobehavioral outcome in a rat model of focal cerebral ischemia reperfusion injury. Mol Med Rep 12(2):2049–2055
Li M, Ma RN, Li LH, Qu YZ, Gao GD (2013) Astragaloside IV reduces cerebral edema post-ischemia/reperfusion correlating the suppression of MMP-9 and AQP4. Eur J Pharmacol 715(1-3):189–195
Papadopoulos MC, Verkman AS (2007) Aquaporin-4 and brain edema. Pediatr Nephrol 22(6):778–784
Park S, Yamaguchi M, Zhou C, Calvert JW, Tang J, Zhang JH (2004) Neurovascular protection reduces early brain injury after subarachnoid hemorrhage. Stroke 35(10):2412–2417
Sabri M, Lass E, Macdonald RL (2013) Early brain injury: a common mechanism in subarachnoid hemorrhage and global cerebral ischemia. Stroke Res Treat 2013:394036
Sehba FA, Hou J, Pluta RM, Zhang JH (2012) The importance of early brain injury after subarachnoid hemorrhage. Prog Neurobiol 97(1):14–37
Shi SS, Yang WZ, Chen Y, Chen JP, Tu XK (2014) Propofol reduces inflammatory reaction and ischemic brain damage in cerebral ischemia in rats. Neurochem Res 39(5):793–799
Sugawara T, Ayer R, Jadhav V, Zhang JH (2008) A new grading system evaluating bleeding scale in filament perforation subarachnoid hemorrhage rat model. J Neurosci Methods 167(2):327–334
Tu XK, Yang WZ, Liang RS et al (2011) Effect of baicalin on matrix metalloproteinase-9 expression and blood–brain barrier permeability following focal cerebral ischemia in rats. Neurochem Res 36(11):2022–2028
Tu XK, Yang WZ, Chen JP et al (2014) Curcumin inhibits TLR2/4-NF-kappaB signaling pathway and attenuates brain damage in permanent focal cerebral ischemia in rats. Inflammation 37(5):1544–1551
Tu XK, Yang WZ, Chen JP et al (2015) Repetitive ischemic preconditioning attenuates inflammatory reaction and brain damage after focal cerebral ischemia in rats: involvement of PI3K/Akt and ERK1/2 signaling pathway. J Mol Neurosci 55(4):912–922
Uekawa K, Hasegawa Y, Ma M et al (2014) Rosuvastatin ameliorates early brain injury after subarachnoid hemorrhage via suppression of superoxide formation and nuclear factor-kappa B activation in rats. J Stroke Cerebrovasc Dis 23(6):1429–1439
Wang Z, Ma C, Meng CJ et al (2012a) Melatonin activates the Nrf2-ARE pathway when it protects against early brain injury in a subarachnoid hemorrhage model. J Pineal Res 53(2):129–137
Wang Z, Meng CJ, Shen XM et al (2012b) Potential contribution of hypoxia-inducible factor-1alpha, aquaporin-4, and matrix metalloproteinase-9 to blood–brain barrier disruption and brain edema after experimental subarachnoid hemorrhage. J Mol Neurosci 48(1):273–280
Wang Z, Ji C, Wu L et al (2014) Tert-butylhydroquinone alleviates early brain injury and cognitive dysfunction after experimental subarachnoid hemorrhage: role of Keap1/Nrf2/ARE pathway. PLoS One 9(5), e97685
Westermaier T, Stetter C, Raslan F, Vince GH, Ernestus RI (2012) Brain edema formation correlates with perfusion deficit during the first six hours after experimental subarachnoid hemorrhage in rats. Exp Transl Stroke Med 4(1):8
Yao X, Derugin N, Manley GT, Verkman AS (2015) Reduced brain edema and infarct volume in aquaporin-4 deficient mice after transient focal cerebral ischemia. Neurosci Lett 584:368–372
Zhang XS, Zhang X, Wu Q et al (2014) Astaxanthin offers neuroprotection and reduces neuroinflammation in experimental subarachnoid hemorrhage. J Surg Res 192(1):206–213
Zhang T, Su J, Guo B, Wang K, Li X, Liang G (2015) Apigenin protects blood–brain barrier and ameliorates early brain injury by inhibiting TLR4-mediated inflammatory pathway in subarachnoid hemorrhage rats. Int Immunopharmacol 28(1):79–87
Acknowledgments
The National Natural Science Foundation of China (81100987), the Doctoral Program Foundation of Institutions of Higher Education of China (20113518120005), the Natural Science Foundation of Fujian Province of China (2011 J05066), the Clinical Key Subject (Neurosurgery) Funding of Fujian Medical University, and the Key Laboratory (Neurosurgical Department) Funding from the Affiliated Union Hospital of Fujian Medical University, supported this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Song-sheng Shi and Hua-bin Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Shi, Ss., Zhang, Hb., Wang, Ch. et al. Propofol Attenuates Early Brain Injury After Subarachnoid Hemorrhage in Rats. J Mol Neurosci 57, 538–545 (2015). https://doi.org/10.1007/s12031-015-0634-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-015-0634-2