Abstract
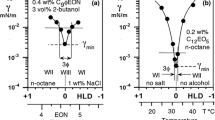
Inspired by the concept of lipophilic and hydrophilic linkers, extended surfactants have been proposed as highly desirable candidates for the formulation of microemulsions with high solubilization capacity and ultralow interfacial tension (IFT), especially for triglyceride oils. The defining characteristic of an extended surfactant is the presence of one or more intermediate-polarity groups between the hydrophilic head and the hydrophobic tail. Currently only limited information exists on extended surfactants; such knowledge is especially relevant for cleaning and separation applications where the cost of the surfactant and environmental regulations prohibit the use of concentrated surfactant solutions. In this work, we examine surfactant formulations for a wide range of oils using dilute solutions of the extended surfactant classes sodium alkyl polypropyleneoxide sulfate (R-(PO) x −SO4Na), and sodium alkyl polypropyleneoxide-polyethyleneoxide sulfate (R-(PO) y -(EO) z −SO4Na). The IFT of these systems was measured as a function of electrolyte and surfactant concentration for polar and nonpolar oils. The results show that these extended surfactant systems have low critical micelle concentrations (CMC) and critical microemulsion concentrations (CμC) compared with other surfactants. We also found that the unique structure of these extended surfactants allows them to achieve ultralow IFT with a wide range of oils, including highly hydrophobic oils (e.g., hexadecane), triolein, and vegetable oils, using only ppm levels of these extended surfactants. It was also found that the introduction of additional PO and EO groups in the extended surfactant yielded lower IFT and lower optimum salinity, both of which are desirable in most formulations. Based on the optimum formulation conditions, it was found that the triolein sample used in these experiments behaved as a very polar oil, and all other vegetable oils displayed very hydrophobic behavior. This unexpected triolein behavior is suspected to be due to uncharacterized impurities in the triolein sample, and will be further evaluated in future research.
Similar content being viewed by others
Abbreviations
- ASC:
-
average number of carbons in saturated chain
- CμC:
-
critical microemulsion concentration
- CMC:
-
critical micelle concentration
- DB:
-
average number of double bonds of unsaturated chains
- EACN:
-
equivalent alkane carbon number
- IFT:
-
interfacial tension
- SP:
-
solubilization parameter
- UC:
-
fraction of unsaturated chains
References
Bourrel, M., and R. Schecter, Microemulsions and Related Systems, Marcel Dekker, New York, 1988.
Salager, J.L., R.E. Antón, D.A. Sabatini, J.H. Harwell, E.J. Acosta, and L.I. Tolosa, Enhancing Solubilization in Microemulsions—State of the Art and Current Trends, J. Surfact. Deterg. 8:3–21 (2005).
Graciaa, A., J. Lachaise, C. Cucuphat, M. Bourrel, and J.L. Salager, Improving Solubilization in Microemulsions with Additives. 1. The Lipophilic Linker Role, Langmuir 9:669–672 (1993).
Graciaa, A., J. Lachaise, C. Cucuphat, M. Bourrel, and J.L. Salager, Improving Solubilization in Microemulsions with Additives. 2. Long Chain Alcohols as Lipophilic Linkers Langmuir 9:3371–3374.
Uchiyama, H., E. Acosta, S. Tran, D.A. Sabatini, and J.H. Harwell, Supersolubilization in Chlorinated Hydrocarbon Microemulsions: Solubilization Enhancement by Lipophilic and Hydrophilic Linkers, Ind. Eng. Chem. Res. 39:2704–2708 (2000).
Acosta, E., H. Uchiyama, D.A. Sabatini, and J.H. Harwell, The Role of Hydrophilic Linkers, J. Surfact. Deterg. 5:151–157 (2002).
Sabatini, D.A., E.J. Acosta, and J.H. Harwell, Linker Molecules in Surfactant Mixtures, J. Colloid Interface Sci. 8:316–326 (2003).
Acosta, E., S. Tran, H. Uchiyama, D.A. Sabatini, and J.H. Harwell, Formulating Chlorinated Hydrocarbon Microemulsions Using Linker Molecules, Environ. Sci. Technol. 36:4618–4624 (2002).
Acosta, E.J., J.H. Harwell, and D.A. Sabatini, Self-assembly in Linker-Modified Microemulsions, J. Colloid Interface Sci. 274:652–664 (2004).
Miñana-Pérez, M., A. Graciaa, J. Lachaise, and J.L. Salager, Solubilization of Polar Oils in Microemulsion Systems, Prog. Colloid Polym. Sci. 98:177–179 (1995).
Miñana-Pérez, M., R.E. Antón, A. Graciaa, J. Lachaise, and J.L. Salager, Solubilization of Polar Oils with Extended Surfactants, Colloid Surfaces A 100:217–224 (1995).
Scorzza, C., P. Godé, P. Martin, M. Miñana-Pérez, J.L. Salager, and P. Villa, Synthesis and Surfactant Properties of a New “Extended” Glucidoamphiphile Made from d-Glucose, J. Surfact. Deterg. 5:331 (2002).
Scorzza, C., P. Godé, G. Goethals, P. Martin, M. Miñana-Pérez, J.L. Salager, A. Usubillaga, and P. Villa, Another New Family of “Extended” Glucidoamphiphiles. Synthesis and Surfactant Properties for Different Sugar Head Groups and Spacer Arm Lengths, J. Surfact. Deterg. 5:337 (2002).
Huang, L., A. Lips, and C. Co, Microemulsification of Triglyceride Sebum and the Role of Interfacial Structure on Bicontinuous Phase Behavior, Langmuir 20:3559–3563 (2004).
Childs, J., E. Acosta, J.F. Scamehorn, and D.A. Sabatini, Surfactant-Enhanced Treatment of Oil-Based Drill Cuttings, J. Energy Res. Technol. 127:153–162 (2005).
Yanatatsaneejit, U., P. Rangsunvigit, J.F. Scamehorn, and S. Chavadej, Diesel Removal by Froth Flotation Under Low Interfacial Tension Conditions I: Foam Characteristics, Coalescence Time, and Equilibration Time, Sep. Sci. Tech. 40:1537 (2005).
Christov, N.C., N.D. Denkov, P.A. Kralchevsky, G. Broze, and A. Mehreteab, Kinetics of Triglyceride Solubilization by Micellar Solutions of Nonionic Surfactant and Triblock Copolymer1. Empty and Swollen Micelles, Langmuir 18:7880–7886 (2002).
Huh, C., Interfacial Tensions and Solubilizing Ability of a Microemulsion Phase That Coexists with Oil and Brine, J. Colloid Interface Sci. 71:408–426 (1979).
Tongcumpou, C., E.J. Acosta, L.B. Quencer, A.F. Joseph, J.F. Scamehorn, D.A. Sabatini, S. Chavadej, and N. Yanumet, Microemulsion Formation and Detergency with Oily Soils: II. Detergency Formulation and Performance, J. Surfact. Deterg. 6:205–214 (2003).
Aveyard, R., B.P. Binks, and P.D.I. Fletcher, Interfacial Tensions and Aggregate Structure in Pentaethylene Glycol Monododecyl Ether/Oil/Water Microemulsion Systems, Langmuir 5:1210 (1989).
Salager, J.L., J. Morgan, R.S. Schechter, W.H. Wade, and E. Vasquez, Optimum Formulation of Surfactant-Oil-Water Systems for Minimum Tension and Phase Behavior, Soc. Petrol. Eng. J. 19:107–115 (1979).
Salager, J.L., N. Márquez, A. Graciaa, and J. Lachaise, Partitioning of Ethoxylated Octylphenol Surfactants in Microemulsion-Oil-Water Systems: Influence of Temperature and Relation Between Partitioning Coefficient and Physicochemical Formulation, Langmuir 16:5534–5539 (2000).
Campbell, E., Baker, N., and Bandurraga, M., eds., Food Fats and Oils, Institute of Shortening and Edible Oils, Washington, DC, 1999, p. 27.
Rosen, M.J., Surfactants and Interfacial Phenomena, 2nd ed., John Wiley and Sons, New York, 1989.
Lange, H., and M.J. Schwuger, Micelle Formation and Kraft-Points in a Homologous Series of Sodium-N-Alkyl Sulfates Including Odd-Numbered Members, Kolloid Z.Z. Polym. 223;145 (1968).
Dahanayake, M., A.W. Cohen, and M.J. Rosen, Relationship of Structure to Properties of Surfactants: 13 Surface and Thermodynamic Properties of Some Oxyethylenated Sulfates and Sulfonates, J. Phys. Chem. 90:2413–2418 (1986).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Witthayapanyanon, A., Acosta, E.J., Harwell, J.H. et al. Formulation of ultralow interfacial tension systems using extended surfactants. J Surfact Deterg 9, 331–339 (2006). https://doi.org/10.1007/s11743-006-5011-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11743-006-5011-2