Abstract
Introduction
There has been recent widespread enthusiasm in epidermal growth factor (EGFR) as a molecularly active target in esophageal adenocarcinoma (EAC). However, there is limited data on the extent of EGFR expression in EAC. Thus, the aim of this study was to evaluated EGFR, pErk1/2, and total Erk1/2 expression in malignant and benign specimens.
Methods
Baseline expression of EGFR in the human normal squamous, Barrett’s, and EAC cell lines were determined as well as after bile acid treatment and curcumin pretreatment. In addition, EGFR expression was also evaluated in 60 matched normal and malignant EAC resected specimens.
Results
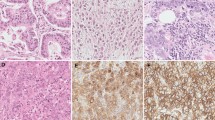
The in vitro studies in the Het-1a, BarT, and OE19 cell lines failed to show any measurable expression of EGFR via Western blot technique. The marker serving as the positive control for the study, MnSOD, showed expression in each cell line for all three treatment regimens at approximately 24 kDa EGFR, showing moderate staining in the malignant tumor specimens and low staining in the benign tissue specimens. pErk1/2 showed low staining in the malignant tumor specimens and no staining in the benign tissue specimens. Total Erk1/2 showed high staining in both the malignant tumor specimens and benign tissue specimens. The differences in the mean staining scores for the malignant versus benign tissue specimens for pErk1/2 and total Erk1/2 are not statistically significant (p = 0.0726 and p = 0.7054, respectively).
Conclusion
Thus, in conclusion, EGFR expression has been confirmed to be limited to non-existent in EAC and thus its use as a clinically active target is limited at best. Prior to the use of these expensive anti-EGFR therapies, confirmation of overexpression should be verified.






Similar content being viewed by others
References
Chen X, Yang CS. Esophageal adenocarcinoma: a review and perspectives on the mechanism of carcinogenesis and chemoprevention. Carcinogenesis 2001; 22:1119–1129.
Schiffman SC, Li Y, Xiao D et al. The resistance of esophageal adenocarcinoma to bile salt insult is associated with manganese superoxide dismutase expression. J Surg Res 2010. doi:10.1016/j.jss.2010.04.038.
Feagins LA, Souza RF. Molecular targets for treatment of Barrett's esophagus. Dis Esophagus 2005; 18:75–86.
Mebratu Y, Tesfaigzi Y. How ERK1/2 activation controls cell proliferation and cell death: is subcellular localization the answer? Cell Cycle 2009; 8:1168–1175.
Li Y, Wo JM, Liu Q et al. Chemoprotective effects of Curcuma aromatica on esophageal carcinogenesis. Ann Surg Oncol 2009; 16:515–523.
O'Sullivan-Coyne G, O'Sullivan GC, O'Donovan TR et al. Curcumin induces apoptosis-independent death in oesophageal cancer cells. Br J Cancer 2009; 101:1585–1595.
Hartojo W, Silvers AL, Thomas DG et al. Curcumin promotes apoptosis, increases chemosensitivity, and inhibits nuclear factor kappaB in esophageal adenocarcinoma. Transl Oncol 2010; 3:99–108.
Mukaida H, Toi M, Hirai T et al. Clinical significance of the expression of epidermal growth factor and its receptor in esophageal cancer. Cancer 1991; 68:142–148.
Takaoka M, Harada H, Andl CD et al. Epidermal growth factor receptor regulates aberrant expression of insulin-like growth factor-binding protein 3. Cancer Res 2004; 64:7711–7723.
al-Kasspooles M, Moore JH, Orringer MB et al. Amplification and over-expression of the EGFR and erbB-2 genes in human esophageal adenocarcinomas. Int J Cancer 1993; 54:213–219
Rygiel AM, Milano F, Ten Kate FJ et al. Gains and amplifications of c-myc, EGFR, and 20.q13 loci in the no dysplasia–dysplasia–adenocarcinoma sequence of Barrett's esophagus. Cancer Epidemiol Biomarkers Prev 2008; 17:1380–1385.
Miller CT, Moy JR, Lin L et al. Gene amplification in esophageal adenocarcinomas and Barrett's with high-grade dysplasia. Clin Cancer Res 2003; 9:4819–4825.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Harper, N., Li, Y., Farmer, R. et al. Epidermal Growth Factor Expression in Esophageal Adenocarcinoma: A Clinically Relevant Target?. J Gastrointest Surg 16, 946–955 (2012). https://doi.org/10.1007/s11605-011-1778-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-011-1778-1