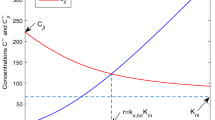
Abstract
In this paper, a typical pharmacokinetic (PK) model is studied for the case of multiple intravenous bolus-dose administration. This model, of one-compartment structure, not only exhibits simultaneous first-order and Michaelis–Menten elimination, but also involves a constant endogenous production. For the PK characterization of the model, we have established the closed-form solution of concentrations over time, the existence and local stability of the steady state. Using analytical approaches and the concept of corrected concentration, we have shown that the area under the curve (\(\hbox {AUC}^{corr}_{ss,\tau }\)) at steady state is higher compared to that at the single dose (\(\hbox {AUC}^{corr}_{0-\infty }\)). Moreover, by splitting the dose and dosing interval into halves, we have revealed that it can result in a significant decrease in the steady-state average concentration. These model-based findings, which contrast with the current knowledge for linear PK, confirm the necessity to revisit drugs exhibiting nonlinear PK and to suggest a rational way of using mathematical analysis for the dosing regimen design.






Similar content being viewed by others
References
Agarwal Ravil P (2000) Difference equations and inequalities. Theory, methods, and applications, 2nd edn., rev. and expanded. New York
Corless RM, Gonnet GH, Hare DEG, Jeffrey DJ, Knuth DE (1996) On the Lambert \(W\) function. Adv Comput Math 5:329–359
Craig M, Humphries AR, Nekka F, Bélair J, Mackey MC LJ (2015) Neutrophil dynamics during concurrent chemotherapy and G-CSF administration: mathematical modelling guides dose optimisation to minimise neutropenia. J Theor Biol 385:77–89
Craig M, Humphries AR, Mackey MC (2016) A mathematical model of granulopoiesis incorporating the negative feedback dynamics and kinetics of G-CSF/neutrophil binding and internalization. Bull Math Biol 78(12):2304–2357
Dirks NL, Meibohm B (2010) Population pharmacokinetics of therapeutic monoclonal antibodies. Clin Pharmacokinet 49(10):633–659
European Medicines Agency (2010) https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-investigation-bioequivalence-rev1_en.pdf. Accessed 11 Aug 2019
FDA Guidance (2014) Guidance for industry. Bioavailability and bioequivalence studies submitted in NDAs or INDs—general considerations. https://www.fda.gov/media/88254/download. Accessed 11 Aug 2019
Foley C, Mackey MC (2009) Mathematical model for G-CSF administration after chemotherapy. J Theor Biol 257:27–44
Frymoyer A, Juul SE, Massaro AN, Bammler TK, Wu YW (2017) High-dose erythropoietin population pharmacokinetics in neonates with hypoxic-ischemic encephalopathy receiving hypothermia. Pediatr Res 81(6):865–872
Gibaldi M, Perrier D (2007) Pharmacokinetics. Informa Healthcare USA Inc, New York
Health Canada (2018) Guidance document: conduct and analysis of comparative bioavailability studies. https://www.canada.ca/content/dam/hc-sc/documents/services/drugs-health-products/drug-products/applications-submissions/guidance-documents/bioavailability-bioequivalence/conduct-analysis-comparative.pdf. Accessed 11 Aug 2019
Jin F, Krzyzanski W (2004) Pharmacokinetic model of target-mediated disposition of thrombopoietin. AAPS Pharm Sci 6(1):E9
Keizer RJ, Huitema AD, Schellens JH, Beijnen JH (2010) Clinical pharmacokinetics of therapeutic monoclonal antibodies. Clin Pharmacokinet 49(8):493–507
Klitgaard T, Nielsen JN, Skettrup MP, Harper A, Lange M (2009) Population pharmacokinetic model for human growth hormone in adult patients in chronic dialysis compared with healthy subjects. Growth Horm IGF Res 19(6):463–470
Kloft C, Graefe EU, Tanswell P, Scott AM, Hofheinz R, Amelsberg A, Karlsson MO (2004) Population pharmacokinetics of sibrotuzumab, a novel therapeutic monoclonal antibody, in cancer patients. Invest New Drugs 22(1):39–52
Kuester K, Kovar A, Lüpfert C, Brockhaus B, Kloft C (2008) Population pharmacokinetic data analysis of three phase I studies of matuzumab, a humanised anti-EGFR monoclonal antibody in clinical cancer development. Br J Cancer 98(5):900–906
Leader B, Baca QJ, Golan DE (2008) Protein therapeutics: a summary and pharmacological classification. Nat Rev Drug Discov 7:21–39
Li J, Nekka F (2007) A pharmacokinetic formalism explicitly integrating the patient drug compliance. J Pharmacokinet Pharmacodyn 34(1):115–139
Mager DE, Jusko WJ (2001) General pharmacokinetic model for drugs exhibiting target-mediated drug disposition. J Pharmacokinet Pharmacodyn 28(6):507–532
Mager DE (2006) Target-mediated drug disposition and dynamics. Biochem Pharmacol 72(1):1–10
Mehdi B (2015) Pharmacokinetics and toxicokinetics. CRC Press, Boca Raton
Quartino AL, Karlsson MO, Lindman H, Friberg LE (2014) Characterization of endogenous G-CSF and the inverse correlation to chemotherapy-induced neutropenia in patients with breast cancer using population modeling. Pharm Res 31(12):3390–3403
Schnell S, Mendoza C (1997) Closed form solution for time dependent enzyme kinetics. J Theor Biol 187:207–212
Shi S (2014) Biologics: an update and challenge of their pharmacokinetics. Curr Drug Metab 15(3):271–290
Tang S, Xiao Y (2007) One-compartment model with Michaelis–Menten elimination kinetics and therapeutic window: an analytical approach. J Pharmacokinet Pharmacodyn 34:807–827
van der Graaf PH, Benson N, Peletier LA (2016) Topics in mathematical pharmacology. J Dyn Diff Equ 28:1337–1356
Wong H, Chow TW (2017) Physiologically based pharmacokinetic modeling of therapeutic proteins. J Pharm Sci 106(9):2270–2275
Woo S, Krzyzanski W, Jusko WJ (2007) Target-mediated pharmacokinetic and pharmacodynamic model of recombinant human erythropoietin (rHuEPO). J Pharmacokinet Pharmacodyn 34(6):849–868
Wu X, Li J, Nekka F (2015) Closed form solutions and dominant elimination pathways of simultaneous first-order and Michaelis–Menten kinetics. J Pharmacokinet Pharmacodyn 42:151–161
Wu X, Nekka F, Li J (2016) Steady-state volume of distribution of two-compartment models with simultaneous linear and saturated elimination. J Pharmacokinet Pharmacodyn 43(4):447–459
Wu X, Nekka F, Li J (2018) Mathematical analysis and drug exposure evaluation of pharmacokinetic models with endogenous production and simultaneous first-order and Michaelis–Menten elimination: the case of single dose. J Pharmacokinet Pharmacodyn 45(5):693–705
Yu RH, Cao YX (2017) A method to determine pharmacokinetic parameters based on andante constant-rate intravenous infusion. Sci Rep 7(1):13279
Zhao L, Shang EY, Sahajwalla CG (2012) Application of pharmacokinetics–pharmacodynamics/clinical response modeling and simulation for biologics drug development. J Pharm Sci 101(12):4367–4382
Acknowledgements
This research is supported by NSERC-Industrial Chair in Pharmacometrics—Novartis, Pfizer and Inventiv Health Clinical and FRQNT Projet d’équipe led by F. Nekka as well as NSERC and FRQNT (F.N. and J.L.). FRQNT Fellowship and NSFC (No. 11501358) hold by X.W. are also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
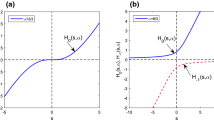
Appendix A: Introduction of X Function and Unique Real Branch in the First Quadrant in \(R^2\)
The aim of introduction of X function is to express the closed-form solution of a one-compartment PK open model exhibiting simultaneous first-order and Michaelis–Menten elimination (Wu et al. 2015). Specifically, the model is described as
where D is an intravenous bolus dose, \(k_\mathrm{el}\) is the rate constant of the first-order elimination, \(V_\mathrm{max}\) and \(K_\mathrm{{m}}\) are the corresponding parameters of Michaelis–Menten elimination representing the maximum velocity of its kinetics and the concentration value at which 50% of \(V_\mathrm{max}\) is reached, and \(V_\mathrm{{d}}\) is the apparent volume of distribution.
Integrating Eq. A.1 gives rise to the following algebraic equation
where
Using the definition of X function (see Definition 1), we are able to express C(t) as
For more details, refer to Wu et al. (2015).
Next, we show the proof of unique real branch of X function in the first quadrant in \({\mathbb {R}}^2\), where \(p>0\) and \(q>0\).
From the definition of X function, we denote
In the scope of real number in the first quadrant in \({\mathbb {R}}^2\), namely for each \(z, X>0\), we can have
By implicit function theorem, there is a unique real solution \(X(z,p,q)>0\) such that the equation \(X(z,p,q)^p(X(z,p,q)+1)^q=z\) is valid. Moreover, the derivative of X(z, p, q) with respect to z is given by
which implies the function X(z, p, q) is increasing with the variable z in the first quadrant.
Appendix B: Proof of the Relationship of Eq. 35
To prove Eq. 35, we define a continuous function as
where \(C_{\beta }^{en}\) and \(C_\mathrm{hs}\) are concentrations defined in Sect. 2.3, D is dose amount, \(V_\mathrm{{d}}\) is the apparent volume of distribution, \(C^{tr}_{ss}(D,\tau )\) and \(C^{tr}_{ss}(\tfrac{D}{2},\tfrac{\tau }{2})\) are respective steady- state trough concentrations which are greater than \(C_\mathrm{hs}\).
Taking derivative of h(x) with respect to the variable x yields
where \(g=g_1-g_2\) with
and
Considering the right hand of \(h'(x)\), the left portion before g is positive; thus, the sign of \(h'(x)\) depends on that of g alone. Furthermore, g can be rewritten as
with
and
Since \(C^{tr}_{ss}(\frac{D}{2},\frac{\tau }{2})>C^{tr}_{ss}(D,\tau )>C_\mathrm{hs}\) and \(x\in [0,C_{\beta }^{en}+C_\mathrm{hs}]\), we have \(0<w_1<w_2\), and thus, g is negative. Therefore, h(x) is a decreasing function with respect to its variable \(x\in [0,C_{\beta }^{en}+C_\mathrm{hs}]\). Subsequently, we have
Rights and permissions
About this article
Cite this article
Wu, X., Nekka, F. & Li, J. Analytical Solution and Exposure Analysis of a Pharmacokinetic Model with Simultaneous Elimination Pathways and Endogenous Production: The Case of Multiple Dosing Administration. Bull Math Biol 81, 3436–3459 (2019). https://doi.org/10.1007/s11538-019-00651-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-019-00651-8
Keywords
- Pharmacokinetic model
- Simultaneous first-order and Michaelis–Menten elimination
- Endogenous production
- Area under the curve at steady state
- Average concentration at steady state