Abstract
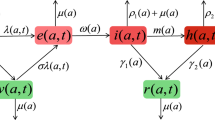
We develop a multi-group epidemic framework via virtual dispersal where the risk of infection is a function of the residence time and local environmental risk. This novel approach eliminates the need to define and measure contact rates that are used in the traditional multi-group epidemic models with heterogeneous mixing. We apply this approach to a general n-patch SIS model whose basic reproduction number \({\mathcal {R}}_0 \) is computed as a function of a patch residence-time matrix \({\mathbb {P}}\). Our analysis implies that the resulting n-patch SIS model has robust dynamics when patches are strongly connected: There is a unique globally stable endemic equilibrium when \({\mathcal {R}}_0>1 \), while the disease-free equilibrium is globally stable when \({\mathcal {R}}_0\le 1 \). Our further analysis indicates that the dispersal behavior described by the residence-time matrix \({\mathbb {P}}\) has profound effects on the disease dynamics at the single patch level with consequences that proper dispersal behavior along with the local environmental risk can either promote or eliminate the endemic in particular patches. Our work highlights the impact of residence-time matrix if the patches are not strongly connected. Our framework can be generalized in other endemic and disease outbreak models. As an illustration, we apply our framework to a two-patch SIR single-outbreak epidemic model where the process of disease invasion is connected to the final epidemic size relationship. We also explore the impact of disease-prevalence-driven decision using a phenomenological modeling approach in order to contrast the role of constant versus state-dependent \({\mathbb {P}}\) on disease dynamics.







Similar content being viewed by others
References
Anderson RM, May RM (1982) Directly transmitted infections diseases: control by vaccination. Science 215:1053–1060
Anderson RM, May RM (1991) Infectious diseases of humans. Dynamics and control. Oxford Science Publications, New York
Arino J (2009) Diseases in metapopulations. In: Ma Z, Zhou Y, Wu J (eds) Modeling and dynamics of infectious diseases, vol 11. World Scientific, Singapore
Arino J, Davis J, Hartley D, Jordan R, Miller J, van den Driessche P (2005) A multi-species epidemic model with spatial dynamics. Math Med Biol 22:129–142
Arino J, van den Driessche P (2003) The basic reproduction number in a multi-city compartmental model. Lecture notes in control and information science, vol 294, pp 135–142
Arino J, van den Driessche P (2006) Disease spread in metapopulations. In: Zhao X-O, Zou X (eds) Nonlinear dynamics and evolution equations, vol 48. Fields Institute Communications, AMS, Providence, pp 1–13
Berman A, Plemmons RJ (1994) Nonnegative matrices in the mathematical sciences, vol 9 of classics in applied mathematics. Society for Industrial and Applied Mathematics (SIAM), Philadelphia, PA. Revised reprint of the 1979 original
Bernoulli D (1766) Essai d’une nouvelle analyse de la mortalité causée par la petite vérole, Mem. Math. Phys. Acad. R. Sci. Paris, pp. 1–45
Blythe SP, Castillo-Chavez C (1989) Like-with-like preference and sexual mixing models. Math Biosci 96:221–238
Brauer F (2008) Epidemic models with heterogeneous mixing and treatment. Bull Math Biol 70:1869–1885
Brauer F, Castillo-Chavez C (1994) Basic models in epidemiology. In: Steele J, Powell T (eds) Ecological time series. Raven Press, New York, pp 410–477
Brauer F, Castillo-Chávez C (2012) Mathematical models in population biology and epidemiology. In: Marsden JE, Sirovich L, Golubitski M (eds) Applied mathematics, vol 40. Springer, New York
Brauer F, Castillo-Chavez C, Velasco-Herná ndez JX (1996) Recruitment effects in heterosexually transmitted disease models. In: Kirschner D (ed) Advances in mathematical modeling of biological processes, vol 3:1. Int J Appl Sci Comput, pp 78–90
Brauer F, Feng Z, Castillo-Chavez C (2010) Discrete epidemic models. Math Biosci Eng 7:1–15
Brauer F, van den Driessche P (2001) Models for transmission of disease with immigration of infectives. Math Biosci 171:143–154
Brauer F, van den Driessche P, Wang L (2008) Oscillations in a patchy environment disease model. Math Biosci 215:1–10
Brauer F, Watmough J (2009) Age of infection epidemic models with heterogeneous mixing. J Biol Dyn 3:324–330
Castillo-Chavez C, Busenberg S (1991) A general solution of the problem of mixing of subpopulations and its application to risk-and age-structured epidemic models for the spread of AIDS. Math Med Biol 8:1–29
Castillo-Chavez C, Cooke K, Huang W, Levin SA (1989) Results on the dynamics for models for the sexual transmission of the human immunodeficiency virus. Appl Math Lett 2:327–331
Castillo-Chavez C, Hethcote H, Andreasen V, Levin S, Liu W (1989) Epidemiological models with age structure, proportionate mixing, and cross-immunity. J Math Biol 27:233–258
Castillo-Chavez C, Huang W (1999) Age-structured core group modeland its impact on STD dynamics. In Mathematical approaches for emerging and reemerging infectious diseases: models, methods, and theory (Minneapolis, MN, 1999), vol. 126 of IMA, Math Appl, Springer, New York, 2002, pp. 261–273
Castillo-Chavez C, Huang W, Li J (1996) Competitive exclusion in gonorrhea models and other sexually transmitted diseases. SIAM J Appl Math 56:494–508
Castillo-Chavez C, Huang W, Li J (1999) Competitive exclusion and coexistence of multiple strains in an SIS STD model. SIAM J Appl Math 59:1790–1811 (electronic)
Castillo-Chavez C, Thieme HR (1995) Asymptotically autonomous epidemic models. In: Arino ADE, Kimmel O, Kimmel M (eds) Mathematical population dynamics: analysis of heterogeneity, volume one: theory of epidemics. Wuerz, Winnipeg
Castillo-Chavez C, Velasco-Hernández JX, Fridman S (1994) Modeling contact structures in biology. In: Levin SA (ed) Frontiers in mathematical biology, vol 100. Springer, Berlin ch. 454–491
Chowell D, Castillo-Chavez C, Krishna S, Qiu X, Anderson KS (2015) Modelling the effect of early detection of Ebola. Lancet 15:148–149
Diekmann O, Heesterbeek JAP, Metz JAJ (1990) On the definition and the computation of the basic reproduction ratio \(R_0\) in models for infectious diseases in heterogeneous populations. J Math Biol 28:365–382
Dietz K, Heesterbeek J (2002) Daniel Bernoulli’s epidemiological model revisited. Math Biosci 180:1–21
Dietz K, Schenzle D (1985) Mathematical models for infectious disease statistics. In: Atkinson, Anthony, Fienberg, Stephen E (eds) A celebration of statistics. Springer, New York, pp 167–204
Fall A, Iggidr A, Sallet G, Tewa J-J (2007) Epidemiological models and Lyapunov functions. Math Model Nat Phenom 2:62–68
Fenichel E, Castillo-Chavez C, Ceddia MG, Chowell G,Gonzalez Parra P, Hickling GJ, Holloway G, Horan R, Morin B, Perrings C, Springborn M, Valazquez L, Villalobos C (2011) Adaptive human behavior in epidemiological models. PNAS 208(15):6306–6311
Guo H, Li M, Shuai Z (2006) Global stability of the endemic equilibrium of multigroup models. Can Appl Math Q 14:259–284
Guo H, Li M, Shuai Z (2008) A graph-theoretic approach to the method of global Lyapunov functions. Proc Am Math Soc 136(8):2793–2802
Hadeler K, Castillo-Chavez C (1995) A core group model for disease transmission. Math Biosci 128:41–55
Heiderich KR, Huang W, Castillo-Chavez C (2002) Nonlocal response in a simple epidemiological model. In: Appli IVM (ed) Mathematical approaches for emerging and reemerging infectious diseases: an introduction, vol 125. Springer, New York, pp 129–151
Hernandez-Ceron N, Feng Z, Castillo-Chavez C (2013) Discrete epidemic models with arbitrary stage distributions and applications to disease control. Bull Math Biol 75:1716–1746
Hethcote HW (1976) Qualitative analyses of communicable disease models. Math Biosci 28:335–356
Hethcote HW (2000) The mathematics of infectious diseases. SIAM Rev 42:599–653 (electronic)
Hethcote HW, Thieme HR (1985) Stability of the endemic equilibrium in epidemic models with subpopulations. Math Biosci 75:205–227
Hethcote HW, Yorke J (1984) Gonorrhea: transmission dynamics and control, vol 56. Lecture notes in biomathematics. Springer
Hirsch M (1984) The dynamical system approach to differential equations. Bull AMS 11:1–64
Horan DR, Fenichel EP (2007) Economics and ecology of managing emerging infectious animal diseases. Am J Agric Econ 89:1232–1238
Horan DR, Fenichel EP, Melstrom RT (2011) Wildlife disease bioeconomics. Int Rev Environ Resour Econ 5:23–61
Horan DR, Fenichel EP, Wolf CA, Graming BM (2010) Managing infectious animal disease systems. Annu Rev Resour Econ 2:101–124
Hsu Schmitz S-F (2000a) Effect of treatment or/and vaccination on HIV transmission in homosexual with genetic heterogeneity. Math Biosci 167:1–18
Hsu Schmitz S-F (2000b) A mathematical model of HIV transmission in homosexuals with genetic heterogeneity. J Theor Med 2:285–296
Hsu Schmitz S-F (2007) The influence of treatment and vaccination induced changes in the risky contact rate on HIV transmisssion. Math Popul Stud 14:57–76
Huang W, Cooke K, Castillo-Chavez C (1992) Stability and bifurcation for a multiple-group model for the dynamics of HIV/AIDS transmission. SIAM J Appl Math 52:835–854
Huang W, Cooke KL, Castillo-Chavez C (1992) Stability and bifurcation for a multiple-group model for the dynamics of HIV/AIDS transmission. SIAM J Appl Math 52:835–854
Hutson V (1984) A theorem on average Lyapunov functions. Monatshefte für Mathematik 98:267–275
Iggidr A, Sallet G, Tsanou B (2012) Global stability analysis of a metapopulation SIS epidemic model. Math Popul Stud 19:115–129
Jacquez JA, Simon CP (1993) The stochastic SI model with recruitment and deaths I. Comparison with the closed SIS model. Math Biosci 117:77–125
Jacquez JA, Simon CP, Koopman J (1991) The reproduction number in deterministic models of contagious diseases. Comment Theor Biol 2:159–209
Jacquez JA, Simon CP, Koopman J, Sattenspiel L, Perry T (1988) Modeling and analyzing HIV transmission: the effect of contact patterns. Math Biosci 92:119–199
Kuniya T, Muroya Y (2014) Global stability of a multi-group SIS epidemic model for population migration. DCDS Ser B 19(4):1105–1118
Lajmanovich A, Yorke J (1976) A deterministic model for gonorrhea in a nonhomogeneous population. Math Biosci 28:221–236
Lin X, So JW-H (1993) Global stability of the endemic equilibrium and uniform persistence in epidemic models with subpopulations. J Aust Math Soc Ser B 34:282–295
Morin B, Castillo-Chavez C (2003) SIR dynamics with economically driven contact rates. Nat Resour Model 26:505–525
Mossong J, Hens N, Jit M, Beutels P, Mikolajczyk R, Massari M, Salmaso S, Tomba GS, Wallinga J, Heijne J, Sadkowska-Todys M, Rosinska M, Edmunds WJ (2008) Social contacts and mixing patterns relevant to the spread of infectious diseases. PLoS Med 5:381–391
Nold A (1980) Heterogeneity in disease-transmission modeling. Math Biosci 52:227
Perrings C, Castillo-Chavez C, Chowell G, Daszak P, Fenichel EP, Finnoff D, Horan RD, Kilpatrick AM, Kinzig AP, Kuminoff NV, Levin S, Morin B, Smith KF, Springborn M (2014) Merging economics and epidemiology to improve the prediction and management of infectious disease. Ecohealth 11(4):464–475
Ross R (1911) The prevention of malaria. John Murray, London
Rushton S, Mautner A (1955) The deterministic model of a simple epidemic for more than one community. Biometrika 42(1/2):126–132
Sattenspiel L, Dietz K (1995) A structured epidemic model incorporating geographic mobility among regions. Math Biosci 128:71–91
Sattenspiel L, Simon CP (1988) The spread and persistence of infectious diseases in structured populations. Math Biosci 90:341–366 [Nonlinearity in biology and medicine (Los Alamos, NM, 1987)]
Shuai Z, van den Driessche P (2013) Global stability of infectious disease models using lyapunov functions. SIAM J Appl Math 73:1513–1532
Simon CP, Jacquez JA (1992) Reproduction numbers and the stability of equilibria of SI models for heterogeneous populations. SIAM J Appl Math 52:541–576
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48
Velasco-Hernández JX, Brauer F, Castillo-Chavez C (1996) Effects of treatment and prevalence-dependent recruitment on the dynamics of a fatal disease. IMA J Math Appl Med Biol 13:175–192
Vidyasagar M (1980) Decomposition techniques for large-scale systems with nonadditive interactions: stability and stabilizability. IEEE Trans Autom Control 25:773–779
Yorke JA, Hethcote HW, Nold A (1978) Dynamics and control of the transmission of gonorrhea. Sex Transm Dis 5:51–56
Acknowledgments
These studies were made possible by grant #1R01GM100471-01 from the National Institute of General Medical Sciences (NIGMS) at the National Institutes of Health. The contents of this manuscript are solely the responsibility of the authors and do not necessarily represent the official views of DHS or NIGMS. Research of Y.K. is partially supported by NSF-DMS (1313312). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. The authors are grateful to two anonymous referees for helpful comments and suggestions which led to an improvement of this paper.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1: Computation of \({\mathcal {R}}_0\)
Proof
The general SIS model with residence time is described by the system (6)
The right-hand member of the above system can be clearly decomposed as \({\mathcal {F}}+{\mathcal {V}}\) where
The Jacobian matrix at the DFE of \({\mathcal {F}}\) and \({\mathcal {V}}\) is given by:
The basic reproduction number \({\mathcal {R}}_0\) is given by the spectral radius of the next-generation matrix \(-FV^{-1}\) (Diekmann et al. 1990; Driessche and Watmough 2002). Hence, we deduce that
\(\square \)
Appendix 2: Proof of Theorem 2.1
The proof uses the method in Iggidr et al. (2012) which is based on Hirsch’s theorem (Hirsch 1984).
Theorem 5.1
(Hirsch 1984) Let \(\dot{x}=F(x)\) be a cooperative differential equation for which \({\mathbb {R}}^n_+\) is invariant , the origin is an equilibrium, each DF(x) is irreducible, and that all orbits are bounded. Suppose that
Then, all orbits in \({\mathbb {R}}^n_+\) tend to zero or there is a unique equilibrium \(p^*\) in the interior of \({\mathbb {R}}^n_+\) and all orbits in \({\mathbb {R}}^n_+\) tend to \(p^*\).
Proof of Theorem 2.1
Equation (6) can be written as:
where \(F=\text {diag}(\bar{N}){\mathbb {P}}\text {diag}({\mathcal {B}})\text {diag}(\tilde{N})^{-1}{\mathbb {P}}^t\) and \(V=-\text {diag}(d_I+\gamma _I)\), as defined in “Appendix 1.” Let us denote by X(I) the semi-flow induced by (14). Hence,
where \(W(I_1,I_2)={\text {diag}}({\mathbb {P}}{\text {diag}}({\mathcal {B}}){\text {diag}}(\tilde{N})^{-1}{\mathbb {P}}^t I)\). Since \({\mathbb {P}}\) is irreducible and \(I\le \bar{N}\), DX(I) is clearly Metzler irreducible matrix. That means, the flow is strongly monotone. Plus, DX(I) is clearly decreasing with respect of I. Hence, by Hirsch’s theorem all trajectories either go to zero or go to an equilibrium point \(\bar{I}\gg 0\). From the relation (15), we have \(DX(0)=F+V\) where F and V are the one defined previously in “Appendix 1.” However, since F a nonnegative matrix and V is Metzler, we have the following equivalence
where \(\alpha (F+V)\) is the stability modulus, i.e., the largest real part of eigenvalues, of \(F+V\) and \(\rho (-FV^{-1})\) the spectral radius of \(-FV^{-1}\). Hence, the DFE is globally asymptotically stable if \({\mathcal {R}}_0=\rho (-FV^{-1})<1\). And if \({\mathcal {R}}_0>1\), i.e., \(\alpha (F+V)>0\), the DFE is unstable (Driessche and Watmough 2002). Since, we have proved that DX(I) is a Metzler matrix, to prove the local stability of the endemic equilibrium \(\bar{I}\gg 0\), we only need to prove that it exists \(w\gg 0\) such that \(DX(\bar{I})w<0\) (Berman and Plemmons 1994). The endemic equilibrium \(\bar{I}\gg 0\) satisfies the equation
Hence,
Hence, with \(w=\bar{I}\), we deduce that \(\bar{I}\) is locally stable. With the attractivity of \(\bar{I}\) guaranteed Hirsh’s theorem, we conclude that the endemic equilibrium \(\bar{I}\gg 0\) is globally asymptotically stable if \({\mathcal {R}}_0>1\).
Finally, if \({\mathcal {R}}_0=1\), we have \(\alpha (F+V)=0\). It exists \(c\gg 0\) such that \((F+V)^tc=0\). By considering the Lyapunov function \(V=\left\langle c |I\right\rangle \). This function is definite positive and its derivation along the trajectories if (14) is
Plus \(\dot{V}=0\) only at the DFE. Hence, the DFE is GAS if \({\mathcal {R}}_0=1\). This completes the proof of the Theorem 2.1. \(\square \)
Appendix 3: Proof of Theorem 2.2
Proof
Since System (6) has an attracting compact \(\Omega \), then according to Theorem (2.1), we can expect that \(\lim _{t\rightarrow \infty } I_i(t)<\frac{b_i}{d_i}\); thus, for time large enough, we can have \(\frac{b_i}{d_i}-I_i>0\), therefore we have
which indicates that when \({\mathcal {R}}_0^i({\mathbb {P}})>1 \)
Then apply the average Lyapunov Theorem (Hutson 1984), we can conclude that \(\liminf _{t\rightarrow \infty } I_i(t)>0\); i.e., the disease in the residence Patch i is persistent if \({\mathcal {R}}_0^i({\mathbb {P}})>1 \) .
If \(p_{ij}>0\) and \(p_{kj}=0\) for all \(k=1,\ldots ,n, \text{ and } k\ne i\), this implies that if there is a portion of the residence Patch i population flowing into the residence Patch j, then there is no other residence Patch k where \(k\ne j\), i.e.,
which also implies that
then we can conclude that Model (6) can have an equilibrium since under these conditions,
Therefore, if the conditions \(p_{kj}=0\) for all \(k=1,\ldots ,n, \text{ and } k\ne j\) whenever \(p_{ij}>0\) hold, then we have
Therefore, \(I_i=0\) is the invariant manifold for Model (6).
On the other hand, when these conditions hold, then we have
Therefore, if \({\mathcal {R}}_0^i({\mathbb {P}})=R_0^i \times \sum _{j=1}^n \left( \frac{\beta _j}{\beta _i}\right) p_{ij}<1\), then we have the following inequality:
Therefore, we have \(\lim _{t\rightarrow \infty } I_i(t)=0\); i.e., there is no endemic in the residence Patch i. \(\square \)
Rights and permissions
About this article
Cite this article
Bichara, D., Kang, Y., Castillo-Chavez, C. et al. SIS and SIR Epidemic Models Under Virtual Dispersal. Bull Math Biol 77, 2004–2034 (2015). https://doi.org/10.1007/s11538-015-0113-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-015-0113-5
Keywords
- Epidemiology
- SIS–SIR models
- Dispersal
- Residence times
- Global stability
- Adaptive behavior
- Final size relationship