Abstract
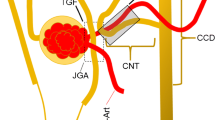
In previous studies, we used a mathematical model of the thick ascending limb (TAL) to investigate nonlinearities in the tubuloglomerular feedback (TGF) loop. That model does not represent other segments of the nephron, the water, and NaCl transport along which may impact fluid flow rate and NaCl transport along the TAL. To investigate the extent to which those transport processes affect TGF mediation, we have developed a mathematical model for TGF signal transduction in a short loop nephron. The model combines a simple representation of the renal cortex with a highly-detailed representation of the outer medulla (OM). The OM portion of the model is based on an OM urine concentrating mechanism model previously developed by Layton and Layton (Am. J. Renal 289:F1346–F1366, 2005a). When perturbations are applied to intratubular fluid flow at the proximal straight tubule entrance, the present model predicts oscillations in fluid flow and solute concentrations in the cortical TAL and interstitium, and in all tubules, vessels, and interstitium in the OM. Model results suggest that TGF signal transduction by the TAL is a generator of nonlinearities: if a sinusoidal oscillation is added to constant intratubular fluid flow, the time required for an element of tubular fluid to traverse the TAL is oscillatory, but nonsinusoidal; those results are consistent with our previous studies. As a consequence, oscillations in NaCl concentration in tubular fluid alongside the macula densa (MD) will be nonsinusoidal and contain harmonics of the original sinusoidal frequency. Also, the model predicts that the oscillations in NaCl concentration at the loop-bend fluid are smaller in amplitude than those at the MD, a result that further highlights the crucial role of TAL in the nonlinear transduction of TGF signal from SNGFR to MD NaCl concentration.
Similar content being viewed by others
References
Armsen, T., Reinhardt, H.W., 1971. Transtubular movement of urea at different degrees of water diuresis. Pflügers Arch. 326, 270–280.
Budu-Grajdeanu, P., Moore, L.C., Layton, H.E., 2007. Effect of tubular inhomogeneities on filter properties of thick ascending limb of Henle’s loop. Math. Biosci. 209(2), 564–592.
Casellas, D., Dupont, M., Bouriquet, N., Moore, L.C., Artuso, A., Mimran, A., 1994. Anatomic pairing of afferent arterioles and renin cell distribution in rat kidneys. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 267(36), F931–F936.
Chen, Y.-M., Yip, K.-P., Marsh, D.J., Holstein-Rathlou, N.-H., 1995. Magnitude of TGF-initiated nephron–nephron interaction is increased in SHR. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 269(38), F198–F204.
Danielson, R.A., Schmidt-Nielsen, B., 1972. Recirculation of urea analogs from renal collecting ducts of high- and low-protein-fed rats. Am. J. Physiol. 223(1), 130–137.
Holstein-Rathlou, N.-H., 1987. Synchronization of proximal intratubular pressure oscillations: evidence for interaction between nephrons. Pflügers Arch. 408, 438–443.
Holstein-Rathlou, N.-H., Leyssac, P.P., 1986. TGF-mediated oscillations in proximal intratubular pressure: Differences between spontaneously hypertensive rats and Wistar–Kyoto rats. Acta Physiol. Scand. 126, 333–339.
Holstein-Rathlou, N.-H., Marsh, D.J., 1990. A dynamic model of the tubuloglomerular feedback mechanism. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 258(27), F1448–F1459.
Jamison, R.L., Kriz, W., 1982. Urinary Concentrating Mechanism: Structure and Function. Oxford University Press, New York.
Källskog, Ö., Marsh, D.J., 1990. TGF-initiated vascular interactions between adjacent nephrons in the rat kidney. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 259(28), F60–F64.
Knepper, M.A., 1983. Urea transport in nephron segments from medullary rays of rats. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 244(13), F622–F627.
Kriz, W., Der architektonische and funktionelle Aufbau der Rattenniere. Z. Zellforsch. 82, 1967.
Kriz, W., Schnermann, J., Koepsell, H., 1972. The position of short and long loops of Henle in the rat kidney. Z. Anat. Entwickl.-Gesch. 138, 301–319.
Layton, A.T., Layton, H.E., 2002. A semi-Lagrangian semi-implicit numerical method for models of the urine concentrating mechanism. SIAM J. Sci. Comput. 23(5), 1528–1548.
Layton, A.T., Layton, H.E., 2003. A region-based model framework for the rat urine concentrating mechanism. Bull. Math. Biol. 65(5), 859–901.
Layton, A.T., Layton, H.E., 2005a. A region-based mathematical model of the urine concentrating mechanism in the rat outer medulla: I. Formulation and base-case results. Am. J. Physiol. Renal Physiol. 289, F1346–F1366.
Layton, A.T., Layton, H.E., 2005b. A region-based mathematical model of the urine concentrating mechanism in the rat outer medulla: II. Parameter sensitivity and tubular inhomogeneity. Am. J. Physiol. Renal Physiol. 289, F1367–F1381.
Layton, A.T., Moore, L.C., Layton, H.E., 2006. Multistability in tubuloglomerular feedback and spectral complexity in spontaneously hypertensive rats. Am. J. Physiol. Renal Physiol. 291, F79–F97.
Layton, A.T., Moore, L.C., Layton, H.E., 2009. Tubuloglomerular feedback signal transduction in a compliant thick ascending limb. Am. J. Physiol. Renal Physiol., submitted.
Layton, H.E., Pitman, E.B., 1994. A dynamic numerical method for models of renal tubules. Bull. Math. Biol. 56(3), 547–565.
Layton, H.E., Pitman, E.B., Moore, L.C., 1991. Bifurcation analysis of TGF-mediated oscillations in SNGFR. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 261(30), F904–F919.
Layton, H.E., Pitman, E.B., Moore, L.C., 1997a. Nonlinear filter properties of the thick ascending limb. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 273(42), F625–F634.
Layton, H.E., Pitman, E.B., Moore, L.C., 1997b. Spectral properties of the tubuloglomerular feedback system. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 273(42), F635–F649.
Lemley, K.V., Kriz, W., 1991. Anatomy of the renal interstitium. Kidney Int. 39, 370–381.
Leyssac, P.P., Baumbach, L., 1983. An oscillating intratubular pressure response to alterations in Henle loop flow in the rat kidney. Acta Physiol. Scand. 117, 415–419.
Marsh, D.J., Sosnovtseva, O.V., Mosekilde, E., Holstein-Rathlou, N.-H., 2007. Vascular coupling induces synchronization, quasiperiodicity, and chaos in a nephron tree. Chaos 17, 015114-1–015114-10.
Mason, J., Gutsche, H.U., Moore, L.C., Müller-Suur, R., 1979. The early phase of experimental acute renal failure. IV. The diluting ability of the short loops of Henle. Pflügers Arch. 379, 11–18.
Nielsen, S., Pallone, T., Smith, B.L., Christensen, E.I., Agre, P., Maunsbach, A.B., 1995. Aquaporin-1 water channels in short and long loop descending thin limbs and in descending vasa recta in rat kidney. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 268(8), F1023–F1037.
Pannabecker, T.L., Dantzler, W.H., Layton, H.E., Layton, A.T., 2008. Role of three-dimensional architecture in the urine concentrating mechanism of the rat renal inner medulla. Am. J. Physiol. Renal Physiol. 295, F1217–F1285.
Pfaller, W., 1982. Structure Function Correlation on Rat Kidney: Quantitative Correlation of Structure and Function in the Normal and Injured Rat Kidney. Springer, New York.
Sakai, T., Craig, D.A., Wexler, A.S., Marsh, D.J., 1986. Fluid waves in renal tubules. Biophys. J. 50, 805–813.
Sands, J.M., Layton, H.E., 2000. Urine concentrating mechanism and its regulation. In: Seldin, D.W., Giebisch, G. (Eds.), The Kidney: Physiology and Pathophysiology (3rd edn.), pp. 1175–1216. Lippincott Williams & Wilkins, Philadelphia.
Schnermann, J., Briggs, J., Schubert, G., 1982. In situ studies of the distal convoluted tubule in the rat. I. Evidence for NaCl secretion. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 243(12), F160–F166.
Schnermann, J., Briggs, J.P., 2000. Function of the juxtaglomerular apparatus: Control of glomerular hemodynamics and renin secretion. In: Seldin, D.W., Giebisch, G. (Eds.), The Kidney: Physiology and Pathophysiology (3rd edn.), pp. 945–980. Lippincott Williams & Wilkins, Philadelphia
Schnermann, J., Chou, C.L., Ma, T., Traynor, T., Knepper, M.A., Verkman, A.S., 1998. Defective proximal tubular fluid reabsorption in transgenic aquaporin-1 null mice. Proc. Natl. Acad. Sci. USA 95, 9660–9664.
Silverman, M., Whiteside, C., Lumsden, C.J., Steinhart, H., 1989. In vivo indicator dilution kinetics of PAH transport on dog kidney. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 256(25), F255–F265.
Steinhausen, M., Hill, E., Parekh, N., 1976. Intravital microscopical studies of the tubular urine flow in the conscious rat. Pflügers Arch. 362, 261–264.
Stumpe, K.O., Lowitz, H.D., Ochwadt, B., 1970. Fluid reabsorption in Henle’s loop and urinary excretion of sodium and water in normal rat and rats with chronic hypertension. J. Clin. Invest. 49, 1200–1212.
Verkman, A.S., 1999. Lessons on renal physiology from transgenic mice lacking aquaporin water channels. J. Am. Soc. Nephrol. 10, 1126–1135.
Wade, J.B., Lee, A.J., Ecelbarger, C.A., Mitchell, C., Bradford, A.D., Terris, J., Kim, G.-H., Knepper, M.A., 2000. UT-A2: a 55-kDa urea transporter in thin descending limb whose abundance is regulated by vasopressin. Am. J. Physiol. Renal Physiol. 278, F52–F62.
Wexler, A.S., Kalaba, R.E., Marsh, D.J., 1991. Three-dimensional anatomy and renal concentrating mechanism. I. Modeling results. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 260(29), F368–F383.
Yip, K.-P., Holstein-Rathlou, N.-H., Marsh, D.J., 1991. Chaos in blood flow control in genetic and renovascular hypertensive rats. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 261(30), F400–F408.
Yip, K.-P., Holstein-Rathlou, N.-H., Marsh, D.J., 1992. Dynamics of TGF-initiated nephron–nephron interactions in normotensive rats and SHR. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 262(31), F980–F988.
Zhai, X.-Y., Thomsen, J.S., Birn, H., Kristoffersen, I.B., Andreasen, A., Christensen, E.I., 2006. Three-dimensional reconstruction of the mouse nephron. J. Am. Soc. Nephrol. 17, 77–88.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Layton, A.T., Edwards, A. Tubuloglomerular Feedback Signal Transduction in a Short Loop of Henle. Bull. Math. Biol. 72, 34–62 (2010). https://doi.org/10.1007/s11538-009-9436-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-009-9436-4