Abstract
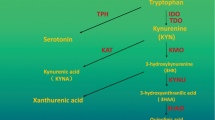
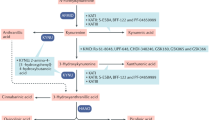
Tryptophan-2, 3-dioxygenase (TDO) is a heme-containing protein catalyzing the first reaction in the kynurenine pathway, which incorporates oxygen into the indole moiety of tryptophan and catalyzes it into kynurenine (KYN). The activation of TDO results in the depletion of tryptophan and the accumulation of kynurenine and its metabolites. These metabolites can affect the function of neurons and inhibit the proliferation of T cells. Increasing evidence demonstrates that TDO is a potential therapeutic target in the treatment of brain diseases as well as in the antitumor and transplant fields. Despite its growing popularity, there are few reviews only focusing on TDO. Hence, we herein review TDO by providing a comprehensive overview of TDO, including its biological functions as well as the evolution, structure and catalytic process of TDO. Additionally, this review will focus on the role of TDO in the pathology of three groups of brain diseases: Schizophrenia, Alzheimer’s disease (AD) and Glioma. Finally, we will also provide an opinion regarding the future developmental directions of TDO in brain diseases, especially whether TDO has a potential role in other brain diseases as well as the development and applications of TDO inhibitors as treatments.



Similar content being viewed by others
References
Adams S, Teo C, Mcdonald KL, et al. (2014) Involvement of the Kynurenine pathway in human glioma pathophysiology. PLoS One 9(11):e112945–e112945
Allikmets LH, Vahing VA, Lapin IP (1969) Dissimilar influences of imipramine, benactyzine and promazine on effects of microinj- ections of noradrenaline, acetylcholine and serotonin into the amygdala in the cat. Psychopharmacologia 15:392–403
Alsadany MA, Shehata HH, Mohamad MI, Mahfouz RG (2013) Histone deacetylases enzyme, copper, and IL-8 levels in patients with Alzheimer’s disease. Am J Alzheimers Dis Other Demen 28:54–61
Altman K, Greengard O (1966) Correlation of kynurenine excretion with liver tryptophan pyrrolase levels in disease and after hydr- ocortisone induction. J Clin Invest 45:1527–1534
Amori L, Wu H-Q, Marinozzi M, Pellicciari R, Guidetti P, Schwarcz R (2009) Specific inhibition of kynurenate synthesis enhances extracellular dopamine levels in the rodent striatum. Neuroscience 159:196–203
Andreasen NC (1996) Pieces of the schizophrenia puzzle fall into place. Neuron 16:697–700
Ball HJ, Jusof FF, Bakmiwewa SM, Hunt NH, Yuasa HJ (2014) Tryptophan-catabolizing enzymes-party of three. Front Immunol 5(485):1–10
Beal MF, Matson WR, Swartz KJ, Gamache PH, Bird ED (1990) Kynurenine pathway measurements in Huntington’s disease str- iatum:evidence for reduced formation of kynurenic acid. J Neurochem 55:1327–1339
Betzen C, White R, Zehendner CM, Pietrowski E, Bender B, HJ L n, CRW K (2009) Oxidative stress upregulates the NMDA receptor on cerebrovascular endothelium. Free Radic Biol Med 47:1212–1220
Blasko I, Veerhuis R, Stampfer-Kountchev M, Saurwein Teissl M, Eikelenboom P, GrubeckLoebenstein B (2000) Costimulatory effects of interferon-gamma and interleukin1beta or tumor necrosis factor alpha on the synthesis of Abeta1-40 and Abeta1-42 by human astrocytes. Neurobiol Dis7:682–689
Bonda DJ, Mailankot M, Stone JG, Garrett MR, Staniszewska M, Castellani RJ, et al. (2010) Indoleamine 2, 3-dioxygenase and 3-hydroxykynurenine modifications are found in the neuropath- ology of Alzheimer’s disease. Redox Rep 15:161–168
Bowers MB Jr, Hoffman FJ (1984) Homovanillic acid in rat caudate and prefrontal cortex following phencyclidine and amphetami- ne. Psychopharmacology 84:137–138
Braidman IP, Rose DP (1971) Effects of sex hormones on three glucocorticoidinducible enzymes concerned with amino acid metabolism in rat liver. Endocrinology 89:1250–1255
Brady FO, Monaco ME, Forman HJ, Schutz G, Feigelson P (1972) On the role of copper in activation of and catalysis by tryptophan-2,3-dioxygenase. J Biol Chem 247(24):7915–7922
Britan A, Maffre V, Tone S, Vet JR (2006) Quantitative and spatial differences in the expression of tryptophan metabolizing enzy- mes in mouse epididymis. Cell Tissue Res 324(2):301–310
Bromet EJ, Fennig S (1999) Epidemiology and natural history of schizophrenia. Biol Psychiatry 46:871–881
Bunney WE Jr, Davis JM (1965) Norepinephrine in depressive reactions. Arch Gen Psychiatry 13:483–494
Campesan S, Green EW, Breda C, Sathyasaikumar KV, Muchowski PJ, Schwarcz R, et al. (2011) The kynurenine pathway modulates neurodegeneration in a drosophila model of Huntington’s disease. Curr Biol 21:961–966
Carpenedo R, Pittaluga A, Cozzi A, Attucci S, Galli A, Raiteri M, et al. (2001) Presynaptic kynurenate-sensitive receptors inhibit glutamate release. Eur J Neuro sci 13:2141–2147966
Carpenter WT Jr, Buchanan RW (1994) Schizophreni a. New Engl J Med 330:681–690
Chong Y (1997) Effect of a carboxy-terminal fragment of the Alzheimer’s amyloid precursor protein on expression of proinflammatory cytokines in rat glial cells. Life Sci 61:2323e2333
Chapin DS, Campbell B, Strick C, and Kozak (2010) The impact of a KATII inhibitor on performance in the rat sustained attention task and conditioned avoidance responding. Soc Neurosci Abstr 472.16
Christiane A, Opitz, Ulrike M, Litzenburger, Felix Sahm, Martina Ott, Isabel Tritschler, SaskiaTrump, Theresa Schumacher, Leonie Jestaedt, Dieter Schrenk, Michael Weller, Manfred Jugold, Gilles J. Guillemin, Christine L. Miller, Christian Lutz, Bernhard Radlwimmer, Irina Lehmann, Andreas von Deimling, Wolfgang Wick & Michael Platten (2011) Anendogenous tumour-promting ligand of the human aryl hydrocarbon receptor. Nature Volume 478, Issue 7368, pp. 197–203
Chiang SS, Riedel M, Schwarz M, Mueller N (2013) Is T helper type2 shift schizophrenia specific? Primary results from a comparison of related psyciatric disorders and healthy controls. Psychiatry Clin Neurosci 67:228–236
Civen M, Knox WE (1960) The specificity of tryptophan analogues as inducers, substrates, inhibitors, and stabilizers of liver tryptophan pyrrolase. J Biol Chem 235(6):1716–1718
Cochran SM, Kennedy M, McKerchar CE, Steward LJ, Pratt JA, Morris BJ (2003) Induction of metabolic hypofunction and neurochemical deficits after chronic intermittent exposure to phencyclidine: differential modulation by antipsychotic drugs. Neuropsychopharmacology 28:265–275
Comings DE, Muhleman D, Dietz G, et al. (1995) Sequence of human tryptophan 2, 3-dioxygenase (TDO2): presence of a glucocorticoid response-like element composed of a GTT repeat and an intronic CCCCT repeat. Genomics 29(2):390–396
Comings DE, Gade R, Muhleman D, et al. (1996) Exon and intron varints in the human tryptophan 2, 3 dioxgenase gene:potential association with Tourette syndrome, substance abuse and other disor ders. Pharmacogenet Genomics 6(4):307–318
Coyle JT (2012) NMDA receptor and schizophrenia:abriefhistory. Schizophr Bull 38:920–926
Csaba J’s, Diane C, Chugani, Muzik O, et al. (2006) In vivo uptake and metabolism of a-[11C] methyl-L-tryptophan in human brain tumors. J Cereb Blood Flow Metab 26:345–357
Danesch U, Gloss B, Schmid W, Schutz G, Schule R, Renkawitz R (1987) Glucocorticoid induction of the rat tryptophan oxygena- se gene is mediated by two widely separated glucocorticoid-responsive elements. EMBO J 6:625–630
Danesch U, Hashimoto S, Renkawitz R, Schutz G (1983) Transcriptional regulation of the tryptophan oxygenase gene in rat liver by glucocorticoids. J Biol Chem 258(4):750–4753
Del Bo R, Angeretti N, Lucca E, De Simoni MG, Forloni G (1995) Reciprocal control of inflammatory cytokines, IL-1 and IL-6, and beta amyloid production in cultures. Neurosci Lett 188:70–74
Deutch AY, Tam SY, Freeman AS, Bowers MB Jr, Roth RH (1987) Mesolimbic and mesocortical dopamine activation induced by phencyclidine:contrasting response to striatal response. Eur J Pharmacol 134:257–264
Dipanwita B, Syun-Ru Y (2007) Human tryptophan dioxygenase:a comparison to indoleamine-2, 3- dioxygenase. J Am Chem Soc 129:15690–15701
Doherty JD, Simonovic M, So R, Meltzer HY (1980) The effect of phencyclidine on dopamine synthesis and metabolism in rat str- iatum. Eur J Pharmacol 65:139–149
Eduard D, Pierre L, Laurence M, et al. (2011) Tryptophan-2, 3-diox- ygenase (TDO) inhibitors 3-(2- (pyridyl) ethenyl) indoles as potential anticancer immunomodulators. J Med Chem 54(15):5320–5334
Eguchi N, Watanabe Y, Kawanishi K, et al. (1984) Inhibition of indoleamine-2, 3-dioxygenase and tryptophan-2, 3-dioxygenase by beta-carboline and indole derivatives. Archives Biochem biophysics 232(2):602–609
Erhardt S, Blennow K, Nordin C, Skogh E, Lindstrom LH, Engberg G (2001) Kynurenic acid levels are elevated in the cereb- rospinal fluid of patients with schizophrenia. Neurosci Lett 313:96–98
Fabrick JA, Kanost MR, Baker JE (2004) RNAi-induced silencing of embryonic tryptophan oxygenase in the Pyralidmoth, Plodia interpunctella. J Insect Sci 4:15
Forouhar F, Anderson JLR, Mowat CG, Vorobiev SM., Hussain A, Abashidze M, Bruckmann C, Thackray SJ, Seetharaman J, Tucker T, et al (2007) Molecular insights into substrate recognition and catalysis by tryptophan 2, 3-dioxygenase. Proc Natl Acad Sci U S A 104, 473–47 8
Forrest CM, Mackay GM, Stoy N, Spiden SL, Taylor R, Stone TW, et al. (2010) Blood levels of kynurenines, interleukin-23 and soluble human leucocyte antigen-gat different stages of hunting- ton’s disease. J Neurochem 112:112–122
Gallas J (2004) Light filters using the oxidative polymerization product of 3-hydroxykynurenine (3-OHKyn). EP, US 6825975:B2
Gellért L, Varga D, Ruszka M, Toldi J, Farkas T, Szatmári I, et al. (2012) Behavioural studies with a newly developed neuroprotective kynaamide. J Neural Transm 119(2):165–172
Grant RS, Naif H, Thuruthyil SJ, Nasr N, Littlejohn T, Takikawa O, et al. (2000) Induction of indoleamine 2,3-dioxygenase in primary human macrophages by hiv-1. Redox Rep 5(2–3):105–107(3)
Gibney SM, Fagan EM, Waldron AM, O'Byrne J, Connor TJ, Harkin A (2014) Inhibition of stress-induced hepatic tryptophan 2,3-dioxygenase exhibits antidepressant activity in an animal model of depressive behaviour. Int J Neuropsychopharmacol 17(06):1–12
Gitter BD, Cox LM, Rydel RE, May PC (1995) Amyloid beta peptide potentiates cytokine secretion by interleukin-1 beta-activated human astrocytoma cells. Proc Natl Acad Sci U S A 92(1073):8–10741
Gongkai JIAO, Xiao-yan KE, Lu CHENG, Ying ZHOU, Ping CHEN, Bei-li SUN, Bing ZOU, Yue-yue HANG, Ming-hao ZHA-NG, Min-jie WANG, Zu-hong LU (2010) Positive associat ion between tryptophan-2, 3-dioxygenase gene polymorphism and autistic disorder in Chinese Han population. Chin Med J 123(2):82
Guidetti P, Luthi-Carter RE, Augood SJ, Schwarcz R (2004) Neostriatal and cortical quinolinate levels are increased in early grade Huntington’s disease. Neurobiol Dis 17:455–461
Guillemin GJ, Brew BJ (2002) Implications of the kynurenine pathway and quinolinic acid in Alzheimer’s disease. Redox Rep 7:199–206
Guillemin GJ, Brew BJ, Noonan CE, Takikawa O, Cullen KM (2005a) Indoleamine 2, 3 dioxygenase and quinolinic acid immunoreactivity in Alzheimer’s diseasehippocampus. Neuropathol Appl Neurobiol 31:395–404
Guillemin GJ, Kerr SJ, Brew BJ (2005b) Involvement of quinolinic acid in AIDS dementia complex. Neurotox Res 7:103–123
Guillemin GJ, Smythe GA, Veas LA, Takikawa O, Brew BJ (2003) A beta1–42 induces production of quinolinic acid by human macrophages and microglia. Neuroreport 14:2311–2315
Haber R, Bessette D, Hulihan-Giblin B, Durcan MJ, Goldman D (1993) Identification of tryptophan-2, 3-dioxygenase RNA in rodent brain. J Neurochem 60:1159–1162
Hayaishi O (1976) Properties and function of indoleamine-2, 3-dioxygenase. J Biochem (Tokyo) 79:13P–21P
Hamann M, Sander SE, Richter A (2008) Effects of the kynurenine 3-hydroxylase inhibitor ro61-8048 after intrastriatal injections on the severity of dystonia in the dt(sz) mutant. Eur J Pharmacol 586:156–159
Hertel P, Mathe JM, Nomikos GG, Iurlo M, Mathe AA, Svensson TH (1996) Effects of damphetamine and phencyclidine on behavior and extracellular concentrations of neurotensin and dopamine in the ventral striatum and medial prefrontal cortex of the rat. Behav Brain Res 72:103–114
Heyes MP, Saito K, Crowley JS, Davis LE, Deitrack MA, et al. (1992) Quinolinic acid and kynurenine pathway metabolism in inflammatory and noninflammatory neurological disease. Brain 115(Pt5):1249–1273
Heyes MP, Saito K, Milstien S, Shiff SJ (1995) Quinolinic acid in tumors, hemorrhage and bacterial infections of the central nervous system in children. J Neurol Sci 133:112–118
Hirata F, Hayaishi O (1972) New degradative routes of 5-hydroxy-tryptophan and serotonin by intestinal tryptophan 2, 3-dioxygenase, Biochem. Biophys. Res Commun 47:1112–1119
Hu X, Bao Z, Hu J, Shao M, Zhang L, Bi K, Zhan A, Huang X (2006) Cloning and characterization of tryptophan-2, 3-dioxygenase gene of Zhikong scallop Chlamys farreri (Jones and Preston1904). Aquac Res 37:1187–1194
Huang CW, Lui CC, Chang WN, Lu CH, Wang YL, et al. (2009) Elevated basal cortisol level predicts lower hippocampal volume and cognitive decline in Alzheimer’s disease. J Clin Neurosci 16(128):3–1286
Huell M, Strauss S, Volk B, Berger M, Bauer J (1995) Interleukin-6 is present in early stages of plaqueformation and is restricted to the brains of Alzheimer’s disease patients. Acta Neuropathol 89:544–551
Iwamoto Y, Lee IS, Tsubaki M, Kido R (1995) Tryptophan-2, 3-dioxygenase in Saccharomyces cerevisiae. Can J Microbiol 141:19–26
Jauch D, Urbanska EM, Guidetti P, Bird ED, Vonsattel JP, Whetsell WO, et al. (1995) Dysfunction of brain kynurenic acid metabolism in Huntington’s disease:focus on kynurenine aminotransferas- es. J Neurol Sci 130:39–47
Jentsch JD, Elsworth JD, Redmond DE Jr, Roth RH (1997) Phencyclidine increases forebrain monoamine metabolism in rats and monkeys: Modulation by the isomers of HA966. J Neurosci 17(1):769–1776
Jentsch JD, Roth RH (1999) The neuropsychopharmacology of mphencyclidine: from NMDA receptor hypofunction to the dopa- mine hypothesis of schizophrenia. Neuropsychopharmacology 20:201–225
Knox WE, Mehler AH (1951) The adaptive increase of the tryptophan peroxidase-oxidase system of liver. Science 113:237–238
Konradi C, Heckers S (2003) Molecular aspects of glutamate dysregulation: implications for schizophrenia and its treatment. Pharmacology Therapeutics 97(2):153–179
Kotake Y, Masayama IZ (1936) Uber den Mechanismus der kynureninbildung aus tryptophan. Z Physiol Chem 243:237–244
Kuhn R (1958) The treatment of depressive states with g22355 (imipramine hydrochloride). Am J Psychiatr 115(5):459–464
Lapin IP, Oxenkrug GF, Osipova SV, Uskova NV (1970) The frog as a subject for screening thymoleptic drugs. J Pharm Pharmacol 22:781–782
Liimatainen S, Lehtimaki K, Raitala A, Peltola M, Oja SS, Peltola J, et al. (2011) Increased indo leamine 2, 3-dioxygenase (IDO) activity in idiopathic generalized epilepsy. Epilepsy Res 94:206–212
Linderholm K.R, Skogh E, Olsson SK, Dahl ML, Holtze M., Engberg G, et al (2012) Increased levels of kynurenine and kynurenic acid in the CSF of patients with schizophrenia. Schizophr Bull 38, 426–432
Löb S, Königsrainer A, Rammensee HG, Opelz G, Terness P (2009) Inhibitors of indoleamine-2, 3-dioxygenase for cancer therapy: can we see the wood for the trees? Nat Rev Cancer 9:445–452
Lorenzen MD, Brown SJ, Denell RE, Beeman RW (2002) Cloning and characterization of the Tribolium castaneum eye-color genes encoding tryptophan oxygenase and kynurenine 3-mono- xygenase. Genetics 160:225–234
Lue LF, Rydel R, Brigham EF, Yang LB, Hampel H., Murphy GM et al (2001) Inflammatory repertoire of Alzheimer’s disease and nondemented elderly microglia in vitro Glia 35, 72–79
Matsumura M, Osada K, Aiba S (1984) L-tryptophan-2, 3-dioxygenase of a moderate thermophile, bacillus brevis, purification, properties and a substrate-mediated stabilization of the quaternary structure. Biochim Biophys Acta 786:9–17
Mcgeer PL, Mcgeer EG (2002) Local neuroinflammation and the progression of alzheimer's disease. J Neuro 8(6):529–538
Mellor A (2005) Indoleamine 2,3 dioxygenase and regulation of T cell immunity. Biochem Biophys Res Commun 338(1):20–24
Mellor AL, Munn DH (2004) IDO expression by dendritic cells: toleranceand tryptophan catabolism. Nat Rev Immunol 4(10):762–774
Meng B, Wu D, Gu J, et al. (2014) Structural and functional analyses of human tryptophan 2,3 dioxygenase. Proteins structure Function Bioinformatics 82(11):3210–3216(7)
Moghaddam B, Javitt D (2012) From revolution to evolution:the glutamate hypothesis of schizophrenia and it simplication for treatment. Neuropsychopharmacology 37:4–15
Möller M, Preez JLD, Viljoen FP, et al. (2013) Social isolation rearing induces immunological, neurochemical, mitochondrial and behavioural deficits in rats, and is reversed by clozapine or N-acetyl cysteine. Brain Behav Immun 30(9):156–167
Monaco F, Fumero S, Mondino A, Mutani R (1979) Plasma and cerebrospinal fluid tryptophan in multiple sclerosis and degene- rative disease. J Neurol Neurosurg Psychiatry 42(7):640–641
Miller C, Llenos IC, Dulay JR, Barillo M.M, Yolken RH., and Weis S (2004) Expression of the kynurenine pathway enzyme trypto- phan 2, 3-dioxygenase is increased in the frontal cortex of individuals with schizophrenia. Neurobiol Dis 15(3):618–629
Miller CL, Llenos IC, Dulay JR, et al. (2006) Upregulation of the initiating step of the kynurenine pathway in postmortem anterior cingulate cortex from individuals with schizophrenia and bipolar disorder. Brain Res 1073-1074(3):25–37
Miller CL, Llenos IC, Cwik M, Walkup J, Weis S (2008) Alterations in kynurenine precursor and product levels in schizophrenia and bipolar disorder. Neurochem Int 52(6):1297–1503
Miller CL, Murakami P, Ruczinski I, Ross RG, Sinkus M, Sullivan B, Leonard S (2009) Two complex genotypes relevant to the kynurenine pathway and melanotropin function show association with schizophrenia and bipolar disorder. Schizophr Res 113:259–267
Mukabayire O, Cornel AJ, Dotson EM, Collins FH, Besansky NJ (1996) The tryptophan oxygenase gene of Anopheles gambiae. Insect Biochem Mol Biol 26:525–528
Myint AM, Schwarz MJ, Verkerk R, Mueller HH, Zach J, Scharpé S, Steinbusch HW, Leonard BE, Kim YK (2011) Reversal of imbalance between kynurenic acid and 3-hydroxykynurenine by antipsychotics in medication-naive and medication-free schizophrenic patients. Brain Behav Immun 25(8):1576–1581
Nakamura T, Shinno HI, Chihara A (1980) Insulin and glucagon as a new regulator system for tryptophan oxygenase activity demonstrated in primary cultured rat hepatocytes. J Biol Chem 255:7533–7535
Newcomer JW, Farber NB, JevtovicTodorovic V, Selke G, Melson AK, Hershey T, et al. (1999) Ketamine-induced NMDA receptor hypofunction as a model of memory impairment and psychosis. Neurol Psychiatr 20:106–118
Nilsson LK, Linderholm KR, Engberg G, Paulson L, Blennow K, Lindstrom LH, et al. (2005) Elevated levels of kynurenic acid in the cerebrospinal fluid of male patients with schizophrenia. Schizophr Res 80:315–322
Niranjan R (2013) Molecular basis of etiological implications in Alzheimer’s disease:focus on neuroinflammation. Mol Neurobiol 48:412–428
Oxenkrug GF, Lapin IP (1971) Effect of dimethyl and monomethyl tricyclic antidepressants on central 5-hydroxytryptamine processes in the frog. J Pharm Pharmacol 23:971–972
Oxenkrug GF (2011) Interferon-gamma inducible kynurenines pteridies inflammation cascade:implications for aging and aging-associated psychiatric and medical disorders. J Neural Transm 118(1):75–85
Oung SN (1991) Use of tryptophan in combination with other antidepressant treatments:a review. J Psychiatry Neurosci 16:241–246
Pearson SJ, Reynolds GP (1992) Increased brain concentrations of aneurotoxin, 3-hydroxykynurenine, in Huntington’s disease. Neurosci Lett 144:199–201
Phillips R (2011) Structure, mechanism, and substrate specificity of kynureninase. Biochimicaet biophysica. Acta Protozool 1814:1481–1488
Pilotte L, Larrieu P, Stroobant V, Colau D, Dolusic E, Frééick R, De Plaen E, Uyttenhove C, Wouters J, Masereel B, Van den Eynde BJ (2012) Reversal of tumoural immune resistance by inhibition of tryptophan 2, 3-dioxygenase. Proc Natl Acad Sci U S A 109:2497–2502
Rahman A, Ting K, Cullen KM, et al. (2009) The excitotoxin quinolinic acid induces tau phosphorylation in human neurons. PLoS One 4(7):e6344–e6344
Rassoulpour A, Wu H-Q, Ferre S, Schwarcz R (2005) Nanomolar concentrations of kynurenic acid reduce extracellular dopamine levels in the striatum. J Neurochem 93:762–765
Reed RD, Nagy LM (2005) Evolutionary redeployment of a biosynthetic module: expression of eyepigment genes vermilion, cinnabar, and white in butterflywing development. Evol Dev 7:301–311
Richter A, Hamann M (2003) The kynurenine 3-hydroxylase inhibitor improves dystoniaina genetic model of paroxysmaldyskines- ia. Eur J Pharmacol 47(8):47–52
Rojo LE, Fernandez JA, Maccioni AA, Jimenez JM, Maccioni RB (2008) Neuroinflammation: implications for the pathogenesis and molecular diagnosis of Alzheimer's disease. Arch Med Res 39:1–16
Rond SL, Gonzalez A, Gonzalez ASL, Carosella ED, Rouas Freiss N (2005) Indoleamine 2, 3 dioxygenase and human leucoc- yte antigen-G inhibit the t-cell alloproliferative response through two independent pathways. Immunology 116(3):297–307
Salter M, Hazelwood R, Pogson CI, Iyer R, Madge DJ (1995) The effects of a novel and selective inhibitor of tryptophan 2, 3-dio- xygenase on tryptophan and serotonin metabolism in the rat. Biochem Pharmacol 49:1435–1442
Schwartz TL,Sachdeva S,Stahl SM (2012) Glutamate neurocircuitry: theoretical underpinnings in schizophrenia. Front Pharmacol 3,195. http://dx.doi.org/10.3389/fphar.2012.00195.
Schildkraut JJ (1967) The catecholamine hypothesis of affective disorders.A review of supporting evidence. International J Psychiatry 4(5):509–522
Schutz G, Killewich L, Chen G, Feigelson P (1975) Control of the mRNA for hepatic tryptohan oxygenase during hormonal and substrate induction. Proc Natl Acad Sci U S A 72:1017–1020
Schwarcz R, Okuno E, White RJ, Bird ED and Whetsell, WO Jr (1988) 3-Hydroxyanthranilate oxygenase activity is increased in the brains of Huntington disease victims. Proc Natl Acad Sci U S A 85, 4079–4081
Schwarcz R, Rassoulpour A, Wu HQ, Medoff D, Tamminga CA, Roberts RC (2001) Increased cortical kynurenate contentin sch- izophrenia. Biol Psychiatry 50:521–530
Schwarcz R, Whetsell WO Jr, Mangano RM (1983) Quinolinic acid: an endogenous metabolite that produces axonsparing lesions in rat brain. Science 219:316–318
Searles LL, Voelker RA (1986) Molecular characterization of the drosophila vermilion locus and its suppressible alleles. ProcNatl Acad Sci USA 83:404–408
Shimizu T, Nomiyama S, Hirata F, Hayaishi O (1978) Indoleamine 2, 3-dioxygenase. Purification some properties J Biol Chem 253:4700–4706
Stone TW, Perkins MN (1981) Quinolinic acid: a potent endogenous excitant at amino acid receptors in CNS. Eur J Pharmacol 72(4):11–412
Stone TW (2001) Endogenous neurotoxins from tryptophan. Toxicon Official J Inter Society Toxinology 39(1):61–73(13)
Stoy N, Mackay GM, Forrest CM, Christofides J, Egerton M, Stone TW, et al. (2005) Tryptophan metabolism and oxidative stress inpatients with Huntington’s disease. J Neurochem 93:611–623
Straub RE, Jiang Y, Maclean CJ, Ma Y, Webb BT, Myakishev MV, et al. (2002) Genetic variation in the 6p22.3 gene dtnbp1, the human ortholog of the mouse dysbindin gene, is associated with schizophrenia. Am J Hum Genet 71(2):337–348
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, et al. (2005) Radiotherapy plus concomitant and adjuvant temozol- omide for glioblastoma. N Engl J Med 352:987–996
Tatsumi K, Higuchi T, Fujiwara H,.Nakayama T, Egawa H & Itoh K. et al (2000) Induction of tryptophan 2, 3-dioxygenase in the mouse endometrium during implantation. Biochemical Biophysical Res Communications 274 (1), 166–170
Thackray SJ, Mowat CG, Chapman SK (2008) Exploring the mechanism of tryptophan 2, 3-dioxygenase. Biochem Soc Trans 36(Pt6):1120–1123
Thomson J, Rankin H, Ashcroft GW, Yates CM, McQueen JK, Cummings SW (1982) The treatment of depression in general practice:a comparison of L-tryptophan, amitriptyline and a combination of L-tryptophan and amitriptyline with placebo. Psychol Med 12:741–751
Torrey EF, Yolken RH, Zito M, Heyes M (1998) Increased CSF and brain quinolinic acid in schizophrenia and bipolar disorder. Schizophr Res 29:91–92
Tshimura Y, Nozaki M., Hayaishi Ohe (1970) Oxygenated form of L-tryptophan-2, 3-dioxygenase as reaction intermediate. J Biol Chem 1970, 245, 3593–3602
Uyttenhove C, Pilotte L, Theate I, et al. (2003) Evidnce for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine-2, 3-dioxygenase. Nat Med 9:1269–1274
Vakhing VA, Allikmets LH, Lapin IP (1968) The development of vomiting under the influence of microinjections of serotonin into the hypothalamus, septum and amygdala of cats having previous- ly received imipramine. Biull EkspBiol Med 66:48–51
Van Os J, Kapur S (2009) Schizophrenia. Lancet 374(9690):635–645
Vander GAT, Zhu W, VazquezManrique RP, Seinstra RI, Dettmer K, Michels H, et al. (2012) Delaying aging and the aging-associated decline in protein homeostasis by inhibition of tryptophan degradation. Proc Natl Acad Sci U S A109:14912
Vasiliev GV, Merkulov VM, Kobzev VF, et al. (1999) Point mutations within 663-666 bp of intron 6 of the human TDO2 gene, associated with a number of psychiatric disorders, damage the YY-1 transcription factor binding site. FEBS Lett 462:85–88(4)
Verma A, Moghaddam B (1996) The role of excitatory amino acids in prefrontal cortex function as assessed by spatial delayed alternation performance in rats:Modulation by dopamine. J Neurosci 16:373–379
Walinder J, Skott A, Carlsson A, Nagy A, Roos BE (1976) Potentiation of the antidepressant action of clomipramine by tryptophan. Arch Gen Psychiatry 33:1384–1389
Widner B, Leblhuber F, Walli J, Tilz GP, Demel U, et al. (2000) Tryptophan degradation and immune activation in Alzheimer’s disease. J Neural Transm 107:343–353
Widner B, Leblhuber F, Fuchs D (2002) Increased neopterin production and tryptophan degradation in advanced Parkinson’s disease. J NeuralTransm 109:181–189
Wolf H, Brown RR (1971) Studies on tryptophan metabolism in male subjects treated with hydrocortisone. J Clin Endocrinol Metab 33:838–843
Wonodi I, Stine OC, Sathyasaikumar KV, Roberts RC, Mitchell BD, Hong LE, et al. (2011) Downregulated kynurenine3-monooxygenase gene expression and enzyme activity in schizophrenia and genetic association with schizophrenia endophenotypes. Arch Gen Psychiatry 68:665–674
Wu W, Nicolazzo JA, Wen L, Chung R, Stankovic R, Bao SS, et al. (2013) Expression of tryptophan-2, 3-dioxygenase and production of kynurenine pathway metabolite sintriple transgenic mice and human Alzheimer’s disease brain. PLoS One 8:e59749
Minatogawa Y, Suzuki S, Ando Y, Tone S, Takikawa O (2003) Tryptophan pyrrolering cleavage enzymes in placenta. Adv Exp Med Biol 527:425–434
Zhang Y, Kang SA, Mukherjee T, Bale S, Crane BR, Begley TP, Ealick SE (2007) Crystal structure and mechanism of tryptophan-2, 3-dioxygenase, a heme enzyme involved in tryptophan catabolism and in quinolinate biosynthesis. Biochemistry 46(145–15):5
Zwilling D, Huang SY, Sathyasaikumar KV, Notarangelo FM, Guidetti P, HQ W, et al. (2011) Kynurenine-3-monooxygenase inhibition in blood ameliorates neurodegeneration. Cell 145:863–874
Acknowledgments
We thank the participants in this study. This work was supported by grants from the National Natural Science Foundation of China (Grant No.81160248 and 81560464 to DYL), the Natural Science Foundation of JiangXi Province (Grant No. 20142BAB205069 to ZZP) and the Department of Public Health of JiangXi Province (Grant No.20121156 to ZZP).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors do not declare any conflicts of interest related to this manuscript.
Additional information
Cheng-Peng Yu and Ze-Zheng Pan contributed equally to this work
Rights and permissions
About this article
Cite this article
Yu, CP., Pan, ZZ. & Luo, DY. TDO as a therapeutic target in brain diseases. Metab Brain Dis 31, 737–747 (2016). https://doi.org/10.1007/s11011-016-9824-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-016-9824-z