Abstract
Stripe rust (or yellow rust), caused by Puccinia striiformis f. sp. tritici, is one of the most destructive diseases of wheat worldwide. Growing resistant cultivars is the best approach to control the disease. To identify and map genes for stripe rust resistance in wheat cultivar ‘Wuhan 2', an F2 population was developed from a cross between the cultivar and susceptible cultivar Mingxian 169. The parents, 179 F2 plants and their derived F2:3 lines were evaluated for responses to Chinese races CYR30 and CYR31 of the pathogen in a greenhouse. A recessive gene for resistance was identified. DNA bulked segregant analysis was applied and resistance gene analog polymorphism (RGAP) and simple sequence repeat (SSR) techniques were used to identify molecular markers linked to the resistance gene. A genetic map consisting of five RGAP and six SSR markers was constructed. The recessive gene, designated Yrwh2, was located on the short arm of chromosome 3B and flanked by SSR markers Xwmc540 and Xgwm566 at 5.9 and 10.0 cM, respectively. The chromosomal location of the resistance gene and its close marker suggest that the locus is different from previously reported stripe rust resistance genes Yr30, QYr.ucw-3BS, Yrns-B1, YrRub and QYrex.wgp-3BL previously mapped to chromosome 3B. Yrwh2 and its closely linked markers are potentially useful for developing stripe rust resistance wheat cultivars if used in combination with other genes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stripe rust (or yellow rust), a worldwide wheat disease caused by fungus Puccinia striiformis Westend. f. sp. tritici Erikss. (Pst), significantly reduces wheat yields and grain quality when developed to an epidemic (Smith et al. 1986; Dimmock and Gooding 2002). Like other rust fungi, uredospores of Pst can be disseminated by wind in a long distance, leading to large-scale epidemics. Stripe rust is a major biological disaster affecting food security in all wheat production regions (Li and Zeng 2000; Chen 2005). The disease often causes 10–30 % yield loss, and under favorable conditions for the fungus the loss can even be 100 %. In China, stripe rust epidemics caused yield losses of more than one million tons in 1950, 1964, 1990 and 2002 (Wan et al. 2004, 2007).
Growing resistant cultivars is the most effective way to control wheat stripe rust. However, seedling resistance (also called all-stage resistance) is usually race-specific and can be overcome by new races of the pathogen. Growing cultivars with a race-specific resistance gene accelerates the selection of new Pst races (Chen et al. 2010; Wang et al. 2012). Since the 1950s, several large-scale cultivar replacements occurred in China due to the appearance of virulent races which rendered previously resistant cultivars susceptible. This phenomenon also explains the lack of stripe rust resistant germplasm sources in China (Yang et al. 2003; Wan et al. 2007). Exploring new sources with resistance genes and developing new cultivars with different types of resistance, stripe rust can be effectively controlled.
Molecular markers closely linked to resistance genes can be used in gene pyramiding, facilitating breeding resistant cultivars (Young 1996). To date, over 50 genes for stripe rust resistance have been officially named (McIntosh et al. 2011; Xu et al. 2013). Only few of the genes are effective against the recent Chinese Pst population (Li and Zeng 2000; Wan et al. 2004; Zhou et al. 2011) (Table 1). Therefore, it is urgent to identify new genes for resistance to stripe rust and to develop molecular markers for efficient incorporation and pyramiding of new genes into wheat cultivars.
Cultivar ‘Wuhan 2' was resistant to most tested Pst races, but which gene(s) control the resistance to stripe rust in this cultivar were not identified. The objective of this study was to characterize the gene(s) conferring resistance in Wuhan 2 through genetic analysis and molecular mapping.
Materials and methods
Plant material
Wuhan 2, a hexaploid winter wheat (Triticum aestivum L.) cultivar exhibited resistance to all tested Chinese Pst races, when tested in the seedling and adult-plant stage in the greenhouse (Kang et al. unpublished data). It was also resistant in the field tests at Yangling (Shaanxi province) and Tianshui (Gansu province) under natural infections since 2008 (Kang et al. unpublished data). Mingxian 169 (M169) is a Chinese winter wheat cultivar that is susceptible to all Pst races in China. Wuhan 2 as the male parent was crossed with M169. In 2010, F1 plants were grown and selfed to produce F2 seeds in a field, and 179 F2 plants from a single F1 plant were grown in the greenhouse and tested with race CYR30. After scoring stripe rust data, the F2 plants were transferred into the open during the 2011–2012 winter for vernalization, and subsequently transplanted in the field with five plants per row of 1.0 m in length spaced 20 cm apart for seed production in 2012. One hundred and seventy nine plants were successfully harvested to produce enough F2:3 seeds. The 179 F2:3 lines were tested with race CYR31 for confirming the phenotypes and determine the genotypes of the F2 plants.
Evaluation of stripe rust response phenotype
To determine reactions of Wuhan 2 to various Pst races at the seedling and adult-plant stages, eight races (CYR25, CYR29, CYR30, CYR31, CYR32, CYR33, PST-Su5 and PST-CH42) predominant from 1980s to 2010 (Wan et al. 2007; He et al. 2011) were used in this study (Table 1). For the seedling tests, the inoculations were done at the two-leaf stage and for the adult-plant tests, the inoculations were done at the heading stage in a greenhouse. The 179 F2 plants together with the parents were inoculated with CYR30. Because of the failure for increasing spores of CYR30, the 179 F2:3 lines together with the parents were inoculated with CYR31 at the seedling stage in the greenhouse. About 20 seeds each of the F2:3 lines and the parents were planted in 8 × 8 × 8 cm3 pots. The plants were dust-inoculated with a mixture of fresh uredospores and talc at a 1:50 ratio and kept in a dew (humid) chamber at 10 °C without light. After 24 h, the inoculated plants were transferred into an environmentally controlled greenhouse with a daily cycle of 16 h of light at 16 °C and 8 h of darkness at 12 °C. Infection type (IT) on a 0–9 scale (Line and Qayoum 1992) was recorded at 16 days after inoculation. To distinguish homozygous and heterozygous lines, a single IT was recorded if all plants of a F2:3 line had uniform IT; whereas different ITs on plants of a line showing segregation of F2:3 plants within a line indicated the heterozygosity of their parental F2 plant (Zhou et al. 2011).
During the 2012 crop season, Wuhan 2 and M169, together with other wheat germplasm, were tested for stripe rust resistance at two field locations at Yangling. On March 20, one of the field nurseries was inoculated with CYR32 and another with a mixture of CYR31, CYR32, CYR33 and PST-CH42.
DNA extraction, PCR amplification, electrophoresis and silver staining
Genomic DNA was extracted from fresh leaves of the parents and each F2 plant using the CTAB method (Yan et al. 2003). DNA quality and concentration were determined using a NanoDrop ND-1000 (Thermo scientific, Wilmington, DE, USA). Stock DNA solutions were diluted to 50 ng/μl as templates for use in PCR amplification. Twenty-seven RGA primers (Table 2) designed by Dr. Meinan Wang, Washington State University based on cereal resistance gene sequences were used in random pairs. Resistance gene analog polymorphism (RGAP) reaction mixtures were made as described by Chen et al. (1998). PCR amplification, electrophoresis and gel visualization were done following the standard procedures and conditions previously described (Chen et al. 1998). The primer sequences and chromosomal location information of simple sequence repeat (SSR) markers were obtained from the Graingenes website (http://wheat.pw.usda.gov/) and the application conditions specific for individual primer pairs were followed.
Bulk segregant analysis
Based on phenotypic data of F2 plants and their single-seed descent F2:3 lines, the five most resistant (IT 2 and DS 5 %) and the five most susceptible (IT 9 and DS 90–100 %) F2 plants were selected for bulk segregant analysis to screen for RGAP and SSR markers potentially linked to the resistance locus (Michelmore et al. 1991; Chen et al. 1998). Resistant and susceptible bulks were constructed with equal amount of DNA from each selected F2 plant. Polymorphic bands specific to the resistant parent and the resistant bulk, or the susceptible parent and the susceptible bulk, were used to genotype the 179 F2 plants.
Chromosomal locations of molecular markers and the linked resistance gene
Chinese Spring (CS) and 21 nulli-tetrasomic (NT) lines were used to determine the chromosomal locations of RGAP markers linked to the stripe rust resistance gene. If the target band was present in CS, the complete set of CS nulli-tetrasomic lines was tested with the markers. The chromosome absent in the nulli-tetrasomic line not showing the band was determined to carry the RGAP marker locus. SSR markers (http://wheat.pw.usda.gov/) for the specific chromosomes were chosen to screen for more markers using bulk segregant analysis, and selected linkage markers were tested with DNA of the 179 F2 plants.
Map construction
Using software quantitative trait locus (QTL) IciMapping V3.2 (Wang 2009; http://www.isbreeding.net/download_software_ICIM.aspx), a genetic map was developed using the marker and stripe rust data of the 179 F2 plants. The Kosambi map function was applied to calculate genetic distances in centiMorgans (cM) (Kosambi 1944). A LOD threshold of 3.0 was used for grouping, and the algorithm “nnTwo Opt” was used for ordering. Linkage groups were assigned to chromosomes and compared with previously published wheat consensus map (Somers et al. 2004; http://wheat.pw.usda.gov).
Results
Stripe rust evaluated in the greenhouse
Under controlled greenhouse conditions, M169 was consistently susceptible (IT 9) with abundant uredia in all of the race tests at seedling and adult-plant stages. Wuhan 2 was resistant (IT 2) to all races except CYR29 and CYR32 (IT 8) at seedling and adult-plant stages (Table 3).
Stripe rust evaluated in the fields
Results from the field test with race mixture showed that Wuhan 2 was resistant (IT 2-4) with disease severity of 30–50 %, with spot necrosis and short stripes, and with some sporulation on leaves at adult-plant tests in Yangling 2012. It was fully susceptible (IT 8-9) to race CYR32 and had disease severity of 60–100 % at every note-taking at another field location in Yangling. The field and greenhouse data were consistent. The results indicated that Wuhan 2 has race-specific resistance to most tested Pst races.
Inheritance of stripe rust resistance in Wuhan 2 investigated on the 179 F2 plants
Of the 179 F2 plants from cross M169/Wuhan 2 were tested with CYR30 at seedling stage in the greenhouse as mentioned in above. The numbers of F2 plants and F2:3 lines with different infection types or categories are presented in Table 3. The 179 F2 plants segregated in 43 resistant (IT 1–4) and 136 susceptible (IT 5–9) ones, fitting a 1:3 ratio (χ2 = 0.3, P = 0.53), suggesting that Wuhan 2 has a recessive gene conferring resistance. Of the 179 F2:3 lines, 40 were homozygous resistant, 97 segregated and 42 were homozygous susceptible, fitting a 1:2:1 ratio (χ2 = 1.3, P = 0.52), confirming one gene for resistance. Almost all F2:3 lines derived from resistant F2 plants were homozygous resistant and most F2:3 lines from F2 plants with intermediate (IT 5–6) or susceptible (IT 7–9) reactions segregated (Table 3). Both the F2 and F2:3 phenotypes indicate a recessive gene, which is temporarily designated as Yrwh2, for resistance to both races CYR30 and CYR31.
Mapping of the all-stage resistance gene
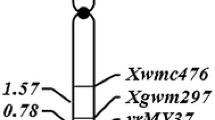
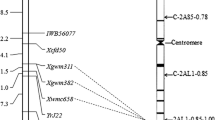
A total of 205 RGA primer pairs were screened on the parents and bulks. Five primer pairs produced strong and repeatable bands which appeared to be associated with the resistance gene locus in the bulked segregant analysis. When the five RGAP markers (Xwgp5475, Xwgp6670, Xwgp5467, Xwgp5672 and Xwgp5759) were used to genotype the 179 F2 plants, they were found to be linked to the resistance gene in Wuhan 2. Xwgp6670 and Xwgp5759 were co-dominant and the remaining three (Xwgp5475, Xwgp5672 and Xwgp5467) were dominant (Table 4). To determine the chromosomal location of the resistance gene, three RGAP markers, Xwgp5467, Xwgp5759 and Xwgp6670, which were also present in CS, were used to test the 21 CS nulli-tetrasomic lines. Xwgp5467, Xwgp5759 and Xwgp6670 amplified the target fragments of 148, 152 and 477 bp in CS (Table 4). The three markers produced the target bands in all 21 nulli-tetrasomic lines, except N3B/T3A, indicating that the resistance gene was located on chromosome 3B. As an example, marker Xwgp5759 was shown in Fig. 1. After the resistance gene was localized on chromosome 3B using the nulli-tetrasomic CS lines tested with RGAP markers, 66 SSR markers on chromosome 3B were screened in bulk segregant analysis to enrich the marker density. Six markers showed clear polymorphisms associated to the resistance locus. Linkage analysis using the six markers tested with the 179 F2 plants indicated that they were linked to the resistance gene locus. Of the six SSR markers, five (Xwmc540, Xgwm566, Xwmc366, Xgwm376 and Xcfd4) were located on the short arm and one (Xwmc182) on the long arm of chromosome 3B. All of the markers including five RGAP and six SSR markers fit a 3:1 (for dominant markers) or 1:2:1 (for co-dominant markers) ratio with P values ranging from 0.36 to 0.96 (data not shown). As examples, RGAP marker Xwgp5467 and SSR marker Xwmc366 are shown in Fig. 2. Using these markers, a linkage group spanning 122.34 cM was constructed around the resistance gene locus (Fig. 3a). The gene was mapped on the short arm of chromosome 3B, flanked by SSR markers Xwmc540 and Xgwm566 at distances of 5.9 and 10.0 cM, respectively (Fig. 3a).
Genetic map showing the location of the Yrwh2 locus on the short arm of wheat chromosome 3B based on 179 F2 plants with their phenotypes confirmed and genotypes determined by the single-seed descent F2:3 lines of cross M169/Wuhan 2 (a). The location of Yrwh2 in relation to previously identified genes on chromosome 3B based on molecular markers (b). References: Yrns-B1 (Börner et al. 2000), Yr30 (Suenaga et al. 2003), YrRub (Bansal et al. 2010), QYr.ucw-3BS (Loweet et al. 2011), and QYrex.wgp-3BL (Lin and Chen 2009)
Discussion
In this study, we identified a gene conferring all-stage race-specific resistance to stripe rust and mapped it to the short arm of chromosome 3B in Wuhan 2. The order of six linked SSR markers was the same as in the previously published wheat chromosome 3B consensus map (Somers et al. 2004; http://wheat.pw.usda.gov).
Several genes for adult-plant or high-temperature adult-plant (HTAP) resistance to stripe rust were previously mapped to the short arm of chromosome 3B in other wheat genotypes. Börner et al. (2000) reported Yrns-B1 on 3BS determining non-race specific adult-plant resistance against stripe rust, and marker Xgwm533.1 was linked to Yrns-B1. Yr30 confers adult-plant resistance to stripe rust (McIntosh et al. 2001). In contrast, Yrwh2 confers all-stage resistance. Sr2, a slow-rusting stem rust resistance gene associated with Yr30, was completely linked to Xgwm533.1 (Singh et al. 2000). Yr30 was closely linked to Xgwm389 and Xgwm533.1 (Suenaga et al. 2003), but these markers were not polymorphic in the mapping population in the present study. A QTL for stripe rust resistance in cultivar Rubric (YrRub) was mapped to the distal region of chromosome 3BS (Bansal et al. 2010). However, YrRub was mapped 10.1 ± 4.2 cM distal of Xgwm533.1, and the distance suggests that the location of YrRub is outside of the Yrwh2 region described in the present study. Lowe et al. (2011) reported QYr.ucw-3BS on chromosome 3BS. It was mapped 3.6 cM distal of Xgwm493 (Fig. 3b). In our study, we could not determine the distance between Yrwh2 and Xgwm493 as the marker was not polymorphic. Lin and Chen (2009) detected a QTL on 3BL (QYrex.wgp-3BL) for HTAP resistance to stripe rust in spring wheat cultivar Express. Yrwh2 and QYrex.wgp-3BL should be different because they confer different types of resistance and are located on different arms of chromosome 3B. QYrex.wgp-3BL was located between Xgwm299 and Xwgp66 (Fig. 3b), while Yrwh2 was not linked with these markers. Therefore, Yrwh2 is likely different from all previously reported genes for resistance to stripe rust in wheat genotypes. However, the genetic distances of Yrwh2 with these genes on chromosome 3BS need further studies.
In the year 2000, stripe rust occurred at an epidemic level in China, mainly by races CYR32 and CYR33, which are virulent on most previously reported resistance genes (Xia et al. 2007; Chen et al. 2009). Among officially named Yr genes, only a small number of genes (e.g. Yr5, Yr10, and Yr15) provide effective resistance in China (Yang et al. 2003; Han et al. 2010; Zhan et al. 2012). Because Yr24/Yr26, which are now considered to be the same gene, are effective against CYR32, CYR33 and against those identified before the two races, the gene has been widely used in breeding programs in China and as a result, many Chinese wheat cultivars carry Yr24/Yr26 (He et al. 2011; Zhang et al. 2013). Since 2008, wheat cultivars Chuanmai 42 that carry Yr26 have become susceptible due to the appearance of a new Pst race PST-CH42 (or PST-V26). This race would likely overcome resistance in many cultivars with Yr26. Therefore, any genes conferring resistance to PST-CH42 will be useful. In the present study, we identified a gene, Yrwh2, effective against this new race.
Although the gene Yrwh2 in Wuhan 2 is ineffective against races CYR29 and CYR32, it is effective against the currently most predominant race, CYR33 and the new emerging race, PST-CH42. Therefore, Yrwh2 should be used in combination with genes effective against CYR29 and CYR32, or more preferably with genes effective against all races or genes for non-race specific resistance. The closely linked molecular markers can be used in marker-assisted selection for pyramiding genes for resistance to stripe rust in wheat breeding programs. Because of its race-specificity, Yrwh2 should be useful also in monitoring different races of the stripe rust pathogen.
References
Bansal U, Hayden M, Gill M, Bariana H (2010) Chromosomal location of an uncharacterised stripe rust resistance gene in wheat. Euphytica 171:121–127
Börner A, Röder MS, Unger O, Meinel A (2000) The detection and molecular mapping of a major gene for non-specific adult-plant disease resistance against stripe rust (Puccinia striiformis) in wheat. Theor Appl Genet 100:1095–1099
Chen XM (2005) Epidemiology and control of stripe rust (Puccinia striiformis f. sp tritici) on wheat. Can J Plant Pathol 27:314–337
Chen XM, Line RF, Leung H (1998) Genome scanning for resistance-gene analogs in rice, barley and wheat by high-resolution electrophoresis. Theor Appl Genet 97:345–355
Chen WQ, Wu LR, Liu TG, Xu SC, Jin SL, Peng YL, Wang BT (2009) Race dynamics, diversity, and virulence evolution in Puccinia striiformis f. sp. tritici, the causal agent of wheat stripe rust in China from 2003 to 2007. Plant Dis 93:1093–1101
Chen XM, Penman L, Wan AM, Cheng P (2010) Virulence races of Puccinia striiformis f. sp. tritici in 2006 and 2007 and development of wheat stripe rust and distributions, dynamics, and evolutionary relationships of races from 2000 to 2007 in the United States. Can J Plant Pathol 32:315–333
Dimmock JPRE, Gooding MJ (2002) The influence of foliar diseases, and their control by fungicides, on the protein concentration in wheat grain: a review. J Agric Sci 138:349–366
Han DJ, Wang QL, Zhang L, Kang ZS (2010) Evaluation of resistance of current wheat cultivars to stripe rust in northwest China, north China and the middle and lower reaches of Changjiang River epidemic area. Scientia Agricultura Sinica 43:2889–2896
He ZH, Lan CX, Chen XM, Zou YC, Zhuang QS, Xia XC (2011) Progress and perspective in research of adult-plant resistance to stripe rust and powdery mildew in wheat. Scientia Agricultura Sinica 44:2193–2215
Kosambi DD (1944) The estimation of map distances from recombination values. Ann Eugen 12:172–175
Li ZQ, Zeng SM (2000) Wheat rusts in China. China Agricultural Press, Beijing
Lin F, Chen XM (2009) Quantitative trait loci for non-race-specific, high-temperature adult-plant resistance to stripe rust in wheat cultivar Express. Theor Appl Genet 118:631–642
Line RF, Qayoum A (1992) Virulence, aggressiveness, evolution, and distribution of races of Puccinia striiformis (the cause of stripe rust of wheat) in North America, 1968–87. U. S. Department of Agriculture Technical Bulletin No. 1788, p 44
Lowe I, Jankuloski LC, Chao SM, Chen XM, See D, Dubcovsky J (2011) Mapping and validation of QTL which confer partial resistance to broadly virulent post-2000 North American races of stripe rust in hexaploid wheat. Theor Appl Genet 123:143–157
McIntosh RA, Devos KM, Dubcovsky J, Rogers WJ (2001) Catalogue of gene symbols for wheat: 2001 supplement [online]. Available from http://grain.jouy.inra.fr/ggpages/wgc/2001upd.html. Accessed 15 Jan 2009
McIntosh RA, Dubcovsky J, Rogers WJ, Morris C, Appels R, Xia XC (2011) Catalogue of gene symbols for wheat: 2011 supplement, http://www.shigen.nig.ac.jp/wheat/komugi/genes/macgene/supplement2011.pdf. Accessed 20 May 2012
Michelmore RI, Paran R, Kesseli RV (1991) Identification of markers closely linked to disease-resistance genes by bulked segregant analysis: a rapid method to detect markers in specific genomic regions by using segregating populations. Proc Natl Acad Sci USA 88:9828–9832
Röder MS, Korzun V, Wendehake K, Plaschke J, Tixier MH, Leroy P, Ganal MW (1998) A microsatellite map of wheat. Genetics 149:207–333
Singh RP, Nelson JC, Sorrells ME (2000) Mapping Yr28 and other genes for resistance to stripe rust in wheat. Crop Sci 40:1148–1155
Smith RCG, Heritage AD, Stapper M, Barrs HD (1986) Effect of stripe rust (Puccinia striiformis West.) and irrigation on the yield and foliage temperature of wheat. Field Crop Res 14:39–51
Somers DJ, Isaac P, Edwards K (2004) A high-density microsatellite consensus map for bread wheat (Triticum aestivum L.). Theor Appl Genet 109:1105–1114
Sourdille P, Gandon B, Chiquet V, Nicot N, Somers D, Murigneux A, Bernard M (2005) Wheat genoplante SSR mapping data release: a new set of markers and comprehensive genetic and physical mapping data. http://wheat.pw.usda.gov/GG2/index.shtml. Accessed 26 Nov 2011
Suenaga K, Singh RP, Huerta-Espino J, William HM (2003) Microsatellite markers for genes Lr34/Yr18 and other quantitative trait loci for leaf rust and stripe rust resistance in bread wheat. Phytopathology 93:881–890
Wan AM, Zhao ZH, Chen XM, He ZH, Jin SL (2004) Wheat stripe rust epidemic and virulence of Puccinia striiformis f. sp. tritici in China in 2002. Plant Dis 88:896–904
Wan AM, Chen XM, He ZH (2007) Wheat stripe rust in China. Aust J Agric Res 58:605–619
Wang JK (2009) Inclusive composite interval mapping of quantitative trait genes. Acta Agronomica Sinica 35:239–245
Wang MN, Chen XM, Xu LS, Cheng P, Bockelman H (2012) Registration of 70 common spring wheat germplasm lines resistant to stripe rust. J Plant Regist 6:1–7
Xia XC, Li ZF, Li GQ, Singh RP (2007) Stripe rust resistance in Chinese bread wheat cultivars and lines. In: Buck HT (ed) Wheat production in stressed environments. Springer, Dordrecht, pp 77–82
Xu LS, Wang MN, Cheng P, Kang ZS, Hulbert SH, Chen XM (2013) Molecular mapping of Yr53, a new gene for stripe rust resistance in durum wheat accession PI 480148 and its transfer to common wheat. Theor Appl Genet 126:523–533
Yan GP, Chen XM, Line RF, Wellings CR (2003) Resistance gene-analog polymorphism markers co-segregating with the Yr5 gene for resistance to wheat stripe rust. Theor Appl Genet 106:636–643
Yang ZM, Xie CJ, Sun QX (2003) Situation of the sources of stripe rust resistance of wheat in the post-CYR32 era in China. Acta Agron Sin 29:161–168
Young ND (1996) QTL mapping and quantitative disease resistance in plants. Annu Rev Phytopathol 34:479–501
Zhan GM, Chen XM, Kang ZS, Huang LL, Wang MN, Wan AM, Cheng P, Cao SQ, Jin SL (2012) Virulence and molecular comparison of Puccinia striiformis f. sp. tritici populations in China and the United States. Fungal Biol 116:643–653
Zhang XJ, Han DJ, Zeng QD, Duan YH, Yuan FP, Shi JD, Wang QL, Wu JH, Huang LL, Kang ZS (2013) Fine mapping of wheat stripe rust resistance gene Yr26 based on collinearity of wheat with Brachypodium distachyon and rice. PLoS One 8(3):e57885
Zhou XL, Wang WL, Wang LL, Hou DY, Jing JX, Wang Y, Xu ZQ, Yao Q, Yin JL, Ma DF (2011) Genetics and molecular mapping of genes for high-temperature resistance to stripe rust in wheat cultivar Xiaoyan 54. Theor Appl Genet 123:431–438
Acknowledgments
This study was supported by the National Key Basic Research Program of China (2013CB127700), China Post-doctoral Science Foundation (2012M512036), the National Natural Science Foundation of China (31201497) and the 111 Project from the Ministry of Education of China (No. B07049). The authors are grateful to Prof. Xianming Chen for critical review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
X. L. Zhou and D. J. Han have made equal contributions towards this study.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Zhou, X.L., Han, D.J., Gou, H.L. et al. Molecular mapping of a stripe rust resistance gene in wheat cultivar Wuhan 2. Euphytica 196, 251–259 (2014). https://doi.org/10.1007/s10681-013-1028-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-013-1028-6