Abstract
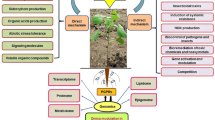
This study aimed to assess the role of two Plant growth promoting rhizobacteria (PGPR), Pseudomonas stutzeri (KX574858) and Pseudomonas putida (KX574857) against charcoal rot instigated by Macrophomina phaseolina in soybean (Glycine max L.) varieties; Ajmeri and NARC grown in pots under greenhouse condition. Macrophomina inocula were added to the soil at the time of sowing. Disease incidence and severity were recorded on 90th day of sowing. Seeds were inoculated with PGPR prior to sowing. Growth parameters such as germination index, shoot height and shoot fresh weight were measured at flowering stage. P. stutzeri significantly (p < 0.05) increased germination index (147% and 115%), shoot height (117% and 103%) and shoot fresh weight (120% and 100%) in cv. Ajmeri and cv. NARC, respectively, in infected plants. Both P. stutzeri (76% and 60%) and P. putida (23% and 22%) significantly decreased the disease severity index of charcoal rot in cv. Ajmeri and cv. NARC, respectively. P. stutzeri induced polyphenol oxidase (435% and 386%), phenylalanine ammonia-lyase (257% and 180%), superoxide dismutase (290% and 240%), peroxidase (733% and 666%) and catalase activities (1867% and 1424%) were linearly increased in cv. Ajmeri and cv. NARC, respectively, after 90 days of infection. Significantly higher accumulation of leaf proline and soluble proteins was recorded in both varieties due to P. stutzeri under infected condition. PGPR enhanced the availability of macronutrients in the rhizosphere of infested soil. The antioxidant and defense enzymes in plant were significantly correlated with disease suppression. The PGPR can be used as a supplement with fungicides to combat adverse effect of disease.











Similar content being viewed by others
References
Aebi, H. (1984). Catalase in vitro. Methods in Enzymolgy, 105, 121–126.
Akladious, S. A., & Abbas, S. M. (2014). Application of Trichoderma harzianum T22 as a biofertilizer potential in maize growth. Journal of Plant Nutrition, 37(1), 30–49.
Altinok, H. H., Dikilitas, M., & Yildiz, H. N. (2013). Potential of pseudomonas and Bacillus isolates as biocontrol agents against fusarium wilt of eggplant. Biotechnology and Biotechnological Equipment, 27(4), 3952–3958.
Arias, S. G., Pons, R. R., Stowasser, V., & Sanfuentes, E. (2013). Temporal analysis of charcoal root rot in forest nurseries under different pathogen inoculum densities and soil moisture content. Tropical Plant Pathology, 38(3), 179–187.
Bates, L. S., Waldren, R. P., & Teare, I. D. (1973). Rapid determination of free proline for water-stress studies. Plant and Soil, 39(1), 205–207.
Beauchamp, C., & Fridovich, I. (1971). Superoxide dismutase: Improved assays and assay applicable to acrylamide gels. Analytical Biochemistry, 44, 276–278.
Beneduzi, A., Ambrosini, A., & Passaglia, L. M. (2012). Plant growth-promoting rhizobacteria (PGPR): Their potential as antagonists and biocontrol agents. Genetics and Molecular Biology, 35(4), 1044–1051.
Chiang, K. S., Liu, H. I., & Bock, C. H. (2017). A discussion on disease severity index values. Part I: warning on inherent errors and suggestions to maximise accuracy. Annals of Applied Biology, 171(2), 139–154.
Corwin, D. L., & Yemoto, K. (2017). Salinity: Electrical conductivity and total dissolved solids. Methods of soil analysis, (msaonline2017). https://doi.org/10.2136/msa2015.0039.
Cotxarrera, L., Trillas-Gay, M. I., Steinberg, C., & Alabouvette, C. (2002). Use of sewage sludge compost and Trichoderma asperellum isolates to suppress Fusarium wilt of tomato. Soil Biology and Biochemistry, 34(4), 467–476.
Doubledee, M. D., Rupe, J. C., Rothrock, C. S., & Bajwa, S. G. (2018). Effect of root infection by Macrophomina phaseolina on stomatal conductance, canopy temperature and yield of soybean. Canadian Journal of Plant Pathology, 40(2), 272–283.
Ganeshamoorthi, P., Anand, T., Prakasam, V., Bharani, M., Ragupathi, N., & Samiyappan, R. (2008). Plant growth promoting rhizobacterial (PGPR) bioconsortia mediates induction of defense-related proteins against infection of root rot pathogen in mulberry plants. Journal of Plant Interactions, 3(4), 233–244.
Godbold, D. L., & Huttermann, A. (1988). Inhibition of photosynthesis and transpiration in relation to mercury-induced root damage in spruce seedlings. Physiologia Plantarum, 74(2), 270–275.
Gouda, S., Kerry, R. G., Das, G., Paramithiotis, S., Shin, H. S., & Patra, J. K. (2018). Revitalization of plant growth promoting rhizobacteria for sustainable development in agriculture. Microbiological Research, 206, 131–140.
Groth, J. V., Ozmon, E. A., & Busch, R. H. (1999). Repeatability and relationship of incidence and severity measures of scab of wheat caused by Fusarium graminearum in inoculated nurseries. Plant Disease, 83, 1033–1038.
Gupta, G. K., Sharma, S. K., & Ramteke, R. (2012). Biology, epidemiology and management of the pathogenic fungus Macrophomina phaseolina (Tassi) Goid with special reference to charcoal rot of soybean (Glycine max (L.) Merrill). Journal of Phytopathology, 160(4), 167–180.
Hashem, A., Abd Allah, E. F., Alqarawi, A. A., Radhakrishnan, R., & Kumar, A. (2017). Plant defense approach of Bacillus subtilis (BERA 71) against Macrophomina phaseolina (Tassi) Goid in mung bean. Journal of Plant Interactions, 12(1), 390–401.
Hernandez-Montiel, L. G., Chiquito Contreras, C. J., Murillo Amador, B., Vidal Hernandez, L., Aguilar, Q., Evanjelina, E., & Chiquito Contreras, R. G. (2017). Efficiency of two inoculation methods of Pseudomonas putida on growth and yield of tomato plants. Journal of Soil Science and Plant Nutrition, 17(4), 1003–1012.
Hershman, D. E. (2011). Charcoal Rot of Soybean. http://plantpathology.ca.uky.edu/files/ppfs-ag-s-02.pdf.
Jamal, Q., Seong, L. Y., Deok, J. H., & Young, K. K. (2018). Effect of plant growth-promoting bacteria Bacillus amylliquefaciens Y1 on soil properties, pepper seedling growth, rhizosphere bacterial flora and soil enzymes. Plant Protection Science, 54(3), 129–137.
Jones, J. B. (2001). Laboratory guide for conducting soil tests and plant analysis. Boca Raton: CRC Press. https://doi.org/10.1201/9781420025293.
Karthikeyan, V., Sankaralingam, A., & Nakkeeran, S. (2006). Management of groundnut root rot with biocontrol agents and organic amendments. Archives of Phytopathology and Plant Protection, 39(3), 215–223.
Khan, S. N. (2007). Macrophomina phaseolina as causal agent for charcoal rot of sunflower. Myco-Phytopathological, 5(2), 111–118. http://pu.edu.pk/images/journal/impp/previousissue/Mycopath-9.pdf.
Khan, M. A., & Ungar, I. A. (1996). Influence of salinity and temperature on the germination of Haloxylon recurvum bunge ex. boiss. Annals of Botany (Lond), 78, 547–551.
Khare, E., & Arora, N. K. (2010). Effect of indole-3-acetic acid (IAA) produced by Pseudomonas aeruginosa in suppression of charcoal rot disease of chickpea. Current Microbiology, 61(1), 64–68.
Kingston, H. M. S., Dengwei, H., Yusheng, L., & Chalk, S. (1998). Accuracy in species analysis: Speciated isotope dilution mass spectrometry (SIDMS) exemplified by the evaluation of chromium species. Spectrochimica Acta Part B: Atomic Spectroscopy, 53(2), 299–309.
Kishor, P. K., Sangam, S., Amrutha, R. N., Sri Laxmi, P., Naidu, K. R., Rao Sreenath Rao, K. R. S. S., Reddy, K. J., Theriappan, P., & Sreenivasulu, N. (2005). Regulation of proline biosynthesis, degradation, uptake and transport in higher plants: its implications in plant growth and abiotic stress tolerance. Current Science, 88(3), 424–438.
Kunwar, I. K., Singh, T., Machado, C. C., & Sinclair, J. B. (1986). Histopathology of soybean seed and seedling infection by Macrophomina phaseolina. Phytopathology, 76(5), 532–535. https://www.apsnet.org/publications/phytopathology/backissues/Documents/1986Articles/Phyto76n05_532.pdf.
Liang, X., Zhang, L., Natarajan, S. K., & Becker, D. F. (2013). Proline mechanisms of stress survival. Antioxidants & Redox Signaling, 19(9), 998–1011.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. Journal of Biological Chemistry, 193(1), 265–275.
Markwell, J., Osterman, J. C., & Mitchell, J. L. (1995). Calibration of the Minolta SPAD-502 leaf chlorophyll meter. Photosynthesis Research, 46, 467–472.
Mengistu, A., Ray, J. D., Smith, J. R., & Paris, R. L. (2007). Charcoal rot disease assessment of soybean genotypes using a colony-forming unit index. Crop Science, 47(6), 2453–2461.
Mengistu, A., Yin, X., Bellaloui, N., McClure, A. M., Tyler, D. D., & Reddy, K. N. (2016). Potassium and phosphorus have no effect on severity of charcoal rot of soybean. Canadian Journal of Plant Pathology, 38(2), 174–182.
Meyer, W. A., Sinclair, J. B., & Khare, M. N. (1974). Factors affecting charcoal rot of soybean seedlings. Phytopathology, 64(6), 845–849.
Mihail, J. D., & Taylor, S. J. (1995). Interpreting variability among isolates of Macrophomina phaseolina in pathogenicity, pycnidium production, and chlorate utilization. Canadian Journal of Botany, 73(10), 1596–1603.
Orhan, E., Esitken, A., Ercisli, S., Turan, M. & Sahin, F. (2006). Effects of plant growth promoting rhizobacteria (PGPR) on yield, growth and nutrient contents in organically growing raspberry. Scientia Horticulturae, 111(1), 38–43.
Pastor, N., Masciarelli, O., Fischer, S., Luna, V., & Rovera, M. (2016). Potential of Pseudomonas putida PCI2 for the protection of tomato plants against fungal pathogens. Current Microbiology, 73(3), 346–353.
Pham, V. T., Rediers, H., Ghequire, M. G., Nguyen, H. H., De Mot, R., Vanderleyden, J., & Spaepen, S. (2017). The plant growth-promoting effect of the nitrogen-fixing endophyte Pseudomonas stutzeri A15. Archives of Microbiology, 199(3), 513–517.
Pine, L., Hoffman, P. S., Malcolm, G. B., Benson, R. F., & Keen, M. G. (1984). Determination of catalase, peroxidase, and superoxide dismutase within the genus legionella. Journal of Clinical Microbiology, 20(3), 421–429.
Rais, A., Jabeen, Z., Shair, F., Hafeez, F. Y. & Hassan, M. N. (2017). Bacillus spp., a bio-control agent enhances the activity of antioxidant defense enzymes in rice against Pyricularia oryzae. PloS one, 12(11).
Rojas-Solís, D., Zetter-Salmón, E., Contreras-Perez, M., Del Carmen Rocha-Granados, M., Macías-Rodríguez, L., & Santoyo, G. (2018). Pseudomonas stutzeri E25 and Stenotrophomonas maltophilia CR71 endophytes produce antifungal volatile organic compounds and exhibit additive plant growth-promoting effects. Biocatalysis and Agricultural Biotechnology, 13, 46–52.
Romeiro, R. S., Lanna Filho, R., Macagnan, D., Garcia, F. A., & Silva, H. S. (2010). Evidence that the biocontrol agent Bacillus cereus synthesizes protein that can elicit increased resistance of tomato leaves to Corynespora cassiicola. Tropical Plant Pathology, 35(1), 011–015.
Sadasivam, S. & Manickam, A. (1992). Biochemical methods for agricultural sciences. Wiley Eastern Ltd, New Delhi, p 246. http://iari.bestbookbuddies.com/cgi-bin/koha/opac-detail.pl?biblionumber=70247.
Sangeetha, G., Thangavelu, R., Rani, S. U., Muthukumar, A., & Udayakumar, R. (2010). Induction of systemic resistance by mixtures of antagonist bacteria for the management of crown rot complex on banana. Acta Physiologiae Plantarum, 32(6), 1177–1187.
Scandiani, M. M., Luque, A. G., Razori, M. V., Ciancio Casalini, L., Aoki, T., O donnell, K., Cervigni, G. D., & Spampinato, C. P. (2014). Metabolic profiles of soybean roots during early stages of Fusarium tucumaniae infection. Journal of Experimental Botany, 66(1), 391–402.
Singh, K. (2011). Organic amendments to soil inoculated arbuscular mycorrhizal fungi and Pseudomonas fluorescens treatments reduce the development of root-rot disease and enhance the yield of Phaseolus vulgaris L. European Journal of Soil Biology, 47(5), 288–295.
Singh, R. J., Nelson, R. L., & Chung, G. H. (2007). Soybean (Glycine max (L.) Merr.). Genetic resources, chromosome engineering, and crop improvement. Oilseed Crops, 4, 13–50.
Singh, J. S., Pandey, V. C., & Singh, D. P. (2011). Efficient soil microorganisms: a new dimension for sustainable agriculture and environmental development. Agriculture, Ecosystems & Environment, 140(3–4), 339–353.
Singh, A., Sarma, B. K., Upadhyay, R. S., & Singh, H. B. (2013). Compatible rhizosphere microbes mediated alleviation of biotic stress in chickpea through enhanced antioxidant and phenylpropanoid activities. Microbiological Research, 168(1), 33–40.
Siranidou, E., Kang, Z., & Buchenauer, H. (2002). Studies on symptom development, phenolic compounds and morphological defence responses in wheat cultivars differing in resistance to Fusarium head blight. Journal of Phytopathology, 150(4–5), 200–208.
Souza, T. P., Dias, R. O., & Silva-Filho, M. C. (2017). Defense-related proteins involved in sugarcane responses to biotic stress. Genetics and Molecular Biology, 40(1), 360–372.
Staple, W. J., & Lehane, J. J. (1962). Variability in soil moisture sampling. Canadian Journal of Soil Science, 42(1), 157–164.
Sutter, V. L., Barry, A. L., Wilkins, T. D., & Zabransky, R. J. (1979). Collaborative evaluation of a proposed reference dilution method of susceptibility testing of anaerobic bacteria. Antimicrobial Agents and Chemotherapy, 16(4), 495–502.
Thompson, D. C., Clarke, B. B., & Kobayashi, D. Y. (1996). Evaluation of bacterial antagonists for reduction of summer patch symptoms in Kentucky bluegrass. Plant Disease, 80(8), 856–862.
Van Assche, F., Cardinaels, C., & Clijisters, H. (1988). Induction of enzyme capacity in plants as a result of heavy metal toxicity: dose-response relations in Phaseolus vulgaris L. treated with zinc and cadmium. Environmental Pollution, 52, 103–115.
Vidhyasekaran, P., & Muthamilan, M. (1995). Development of formulation of Pseudomonas fluorescens for control of chickpea wilt. Plant Disease, 79(8), 782–786.
Walkley, A., & Black, I. A. (1934). An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Science, 37(1), 29–38.
Whetten, R. W., & Sederoff, R. R. (1992). Phenylalanine ammonia-lyase from loblolly pine. Plant Physiology, 98(1), 380–386.
Wightwick, A., Walters, R., Allinson, G., Reichman, S. & Menzies, N. (2010). Environmental risks of fungicides used in horticultural production systems. In Fungicides. InTech. https://doi.org/10.5772/13032.
Yang, G., & Huang, T. S. (1994). Human face detection in a complex background. Pattern Recognition, 27, 53–63.
Yang, X. B., & Navi, S. S. (2005). First report of charcoal rot epidemics caused by Macrophomina phaseolina in soybean in Iowa. Plant Disease, 89(5), 526–526.
Zhao, L., Xu, Y., & Lai, X. (2018). Antagonistic endophytic bacteria associated with nodules of soybean (Glycine max L.) and plant growth-promoting properties. Brazilian Journal of Microbiology, 49(2), 269–278.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human participants and animal studies
This research did not involve human participants or any animal experimentation.
Rights and permissions
About this article
Cite this article
Mufti, R., Bano, A. PGPR-induced defense responses in the soybean plant against charcoal rot disease. Eur J Plant Pathol 155, 983–1000 (2019). https://doi.org/10.1007/s10658-019-01828-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-019-01828-6