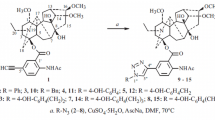
Derivatives of lappaconitine containing styryl, hetarylvinyl, 3-oxobutenyl, and acetyl substituents on the aromatic ring were synthesized using a Heck reaction of 5′-iodolappaconitine and various olefines.


Similar content being viewed by others
References
V. E. Romanov, E. E. Shul’ts, M. M. Shakirov, and G. A. Tolstikov, Zh. Org. Khim., 47, 578 (2011).
T. G. Tolstikova, E. E. Shul’ts, A. O. Bryzgalov, M. V. Khvostov, V. E. Romanov, S. A. Osadchii, and G. A. Tolstikov, Khim. Interesakh Ustoich. Razvit., 15, 599 (2007).
S. A. Osadchii, E. E. Shul’ts, E. V. Polukhina, M. M. Shakirov, and G. A. Tolstikov, Izv. Akad. Nauk, Ser. Khim., 1038 (2006).
A. V. Belovodskii, E. E. Shul’ts, M. M. Shakirov, V. E. Romanov, B. Zh. Elmuradov, Kh. M. Shakhidoyatov, and G. A. Tolstikov, Khim. Prir. Soedin., 747 (2010).
I. Fritzson, B. Svensson, S. Al-Karadaghi, B. Walse, U. Wellmar, U. J. Nilsson, D. da Graca Thrige, and S. Jonsson, Chem. Med. Chem., 5, 608 (2010).
J. Patterson, R. Lum, and R. Wayne, U.S. Pat. No. 6,479,548 B2, “Substituted Stilbenes as Glucose Uptake Enhancers,” (2002).
C.-M. Anderson and A. Hallberg, J. Org. Chem., 52, 3529 (1987).
V. T. Bauman, E. E. Shults, M. M. Shakirov, and G. A. Tolstikov, Izv. Akad. Nauk, Ser. Khim., 1206 (2007).
S. A. Osadchii, N. A. Pankrushina, M. M. Shakirov, E. E. Shults, and G. A. Tolstikov, Izv. Akad. Nauk, Ser. Khim., 552 (2000).
Y. Takahashi, T. Ito, S. Sakai, and Y. Ishii, Chem. Commun., 1065 (1970).
Acknowledgment
The work was supported financially by the Russian Foundation for Basic Research (Project No. 11-03-00242).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 1, January–February, 2013, pp. 58–61.
For No. XVIII, see [1].
Rights and permissions
About this article
Cite this article
Romanov, V.E., Shul’ts, E.E., Shakirov, M.M. et al. Alkaloids of the Flora of Siberia and Altai. XIX. Synthesis of new lappaconitine derivatives on the aromatic ring. Chem Nat Compd 49, 66–69 (2013). https://doi.org/10.1007/s10600-013-0506-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-013-0506-3