Abstract
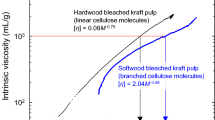
Softwood and hardwood bleached kraft pulps (SBKP and HBKP, respectively) and highly crystalline native celluloses such as algal, tunicate, bacterial and cotton lint celluloses were dissolved in 8 % (w/v) LiCl/N,N-dimethylacetamide (DMAc) after ethylenediamine (EDA) pretreatment. Complete dissolution of SBKP and other highly crystalline native celluloses in 8 % LiCl/DMAc was achieved after solvent exchange from EDA to DMAc through methanol. Neutral sugar composition analysis showed no significant differences between the original and EDA-treated pulps. A combination of size-exclusion chromatography and multi-angle laser light scattering (SEC–MALLS) was used to analyze the cellulose solutions after dilution to 1 % (w/v) LiCl/DMAc. The 0.05 % (w/v) solutions of highly crystalline cellulose in 1 % (w/v) LiCl/DMAc contained entangled molecules, and therefore 0.025 % (w/v) cellulose solutions in 1 % (w/v) LiCl/DMAc were used in the SEC–MALLS analysis to obtain reliable conformation plots (or double-logarithmic plots of molecular mass vs. root-mean-square radius). All the cellulose samples except SBKP gave conformation plots with slope values of 0.56–0.57, showing that these cellulose molecules had random-coil conformations. In contrast, SBKP gave a slope value of 0.35, indicating that some branched structures were present in the high-molecular-mass fraction. Double-logarithmic plots of the reduced viscosities of the cellulose solutions in 1 % (w/v) LiCl/DMAc versus the molecular mass were linear, except for SBKP, also suggesting the presence of anomalous cellulose structures in SBKP.







Similar content being viewed by others
References
Berggren R, Berthold F, Sjöholm E, Lindström M (2003) Improved methods for evaluating the molar mass distributions of cellulose in kraft pulp. J Appl Polym Sci 88:1170–1179
Dupont A, Harrison G (2004) Conformation and dn/dc determination of cellulose in N,N-dimethylacetamide containing lithium chloride. Carbohydr Polym 58:233–243
Kuramae R, Saito T, Isogai A (2014) TEMPO-oxidized cellulose nanofibrils prepared from various plant holocelluloses. React Funct Polym 85:126–133
Larsson PT, Hult EL, Wickholm K, Pettersson E, Tommy I (1999) CP/MAS 13C-NMR spectroscopy applied to structure and interaction studies on cellulose I. Solid State Nucl Magn Reson 15:31–40
Okita Y, Saito T, Isogai A (2010) Entire surface oxidation of various cellulose microfibrils by TEMPO-mediated oxidation. Biomacromolecules 22:1696–1700
Ono Y, Hiraoki R, Fujisawa S, Saito T, Isogai A (2015) SEC–MALLS analysis of wood holocelluloses dissolved in 8% LiCl/1,3-dimethyl-2-imidazolidinone: challenges and suitable analytical conditions. Cellulose 22:3347–3357
Ono Y, Ishida T, Soeta H, Saito T, Isogai A (2016) Reliable dn/dc values of cellulose, chitin, and cellulose triacetate dissolved in LiCl/N,N-dimethylacetamide for molecular mass analysis. Biomacromolecules 17:192–199
Potthast A, Radosta S, Saake B, Lebioda S, Heinze T, Henniges U, Isogai A, Koschella A, Kosma P, Rosenau T, Schiehser S, Sixta H, Strlić M, Strobin G, Vorwerg W, Wetzel H (2015) Comparison testing of methods for gel permeation chromatography of cellulose: coming closer to a standard protocol. Cellulose 22:1591–1613
Schult T, Hjerde T, Optun OI, Kleppe PJ, Moe S (2002) Characterization of cellulose by SEC-MALLS. Cellulose 9:149–158
Siller M, Ahn K, Pircher N, Rosenau T, Potthast A (2014) Dissolution of rayon fibers for size exclusion chromatography: a challenge. Cellulose 21:3291–3301
Tanaka R, Saito T, Ishii D, Isogai A (2014) Determination of nanocellulose fibril length by shear viscosity measurement. Cellulose 21:1581–1589
Tanaka R, Saito T, Hondo H, Isogai A (2015) Influence of flexibility and dimensions of nanocelluloses on the flow properties of their aqueous dispersions. Biomacromolecules 16:2127–2131
Wada M, Kwon GJ, Nishiyama Y (2008) Structure and thermal behavior of a cellulose I-ethylenediamine complex. Biomacromolecules 9:2898–2904
Wada M, Heux L, Nishiyama Y, Langan P (2009) The structure of the complex of cellulose I with ethylenediamine by X-ray crystallography and cross-polarization/magic angle spinning 13C nuclear magnetic resonance. Cellulose 16:943–957
Wise LE, Murphy M, D’Addieco AA (1946) A chlorite holocellulose, its fractionation and bearing on summative wood analysis and studies on the hemicellulose. Paper Trade J 122:35–43
Yamamoto M, Kuramae R, Yanagisawa M, Ishii D, Isogai A (2011) Light-scattering analysis of native wood holocelluloses totally dissolved in LiCl–DMI solutions: high probability of branched structures in inherent cellulose. Biomacromolecules 12:3982–3988
Yanagisawa M, Isogai A (2007) Size exclusion chromatographic and UV-VIS absorption analyses of unbleached and bleached softwood kraft pulps using LiCl/1,3-dimethyl-2-imidazolidinone as a solvent. Holzforschung 61:236–241
Yanagisawa M, Shibata I, Isogai A (2005) SEC-MALLS analysis of softwood kraft pulp using LiCl/1,3-dimethyl-2-imidazolidinone as an eluent. Cellulose 12:151–158
Acknowledgments
This research was supported by Core Research for Evolutional Science and Technology (CREST) of the Japan Science and Technology Agency (JST).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ono, Y., Tanaka, R., Funahashi, R. et al. SEC–MALLS analysis of ethylenediamine-pretreated native celluloses in LiCl/N,N-dimethylacetamide: softwood kraft pulp and highly crystalline bacterial, tunicate, and algal celluloses. Cellulose 23, 1639–1647 (2016). https://doi.org/10.1007/s10570-016-0948-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-016-0948-4