Abstract
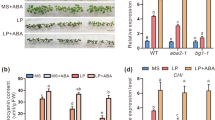
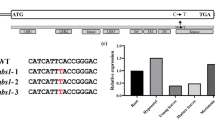
To investigate whether brassinosteroids (BR) affects cytokinin (CK)-induced anthocyanin biosynthesis, seedlings of the Arabidopsis dwarf4 (dwf4) mutants including partially suppressing coi1 (psc1) and dwf4-102, which are defective in the BR biosynthesis, and the brassinosteroid-insensitive 1–4 (bri1-4) mutant defective in BR signalling were used for the analysis of CK-induced anthocyanin accumulation and the expression of anthocyanin biosynthetic genes and WD-repeat/Myb/bHLH transcription factors. The results show that the CK-induced anthocyanin accumulation was remarkably reduced in dwf4 and bri1-4 mutants, but distinctly increased in the wild type (WT) treated with BR. Moreover, the CK-induced expressions of the late anthocyanin biosynthetic genes including dihydroflavonol reductase, leucoanthocyanidin dioxygenase, and UDP-glucose: flavonoid-3-O-glucosyl transferase were significantly reduced in bri1-4 and dwf4-102 mutants compared to WT. In addition, the expressions of transcription factors production of anthocyanin pigment 1 (PAP1), glabra 3 (GL3), and enhancer of glabra 3 (EGL3) were induced by CK in WT but not in the bri1-4 and dwf4-102 mutants. These results indicate that BR enhanced the CK-induced anthocyanin biosynthesis by up-regulating the late anthocyanin biosynthetic genes and this regulation might be mediated by the transcription factors PAP1, GL3, and EGL3.
Similar content being viewed by others
Abbreviations
- BR:
-
brassinosteroids
- CHI :
-
chalcone isomerase
- CHS :
-
chalcone synthase
- CK:
-
cytokinin
- DFR :
-
dihydroflavonol reductase
- EGL3 :
-
enhancer of glabra 3
- GL3 :
-
glabra 3
- JA:
-
jasmonate
- LDOX :
-
leucoanthocyanidin dioxygenase
- PAP1/2 :
-
production of anthocyanin pigment 1/2
- TTG1 :
-
transparent testa glabra 1
- UF3GT :
-
UDP-glucose: flavonoid-3-O-glucosyl transferase
References
Aza-González, C., Herrera-Isidrón, L., Núñez-Palenius, H.G., Martínez de la Vega, O., Ochoa-Alejo, N.: Anthocyanin accumulation and expression analysis of biosynthesisrelated genes during chili pepper fruit development. — Biol. Plant. 57: 49–55, 2013.
Bajguz, A., Hayat, S.: Effects of brassinosteroids on the plant responses to environmental stresses. — Plant Physiol. Biochem. 47: 1–8, 2009.
Cevahir, G., Yentur, S., Eryilmaz, F., Yilmazer, N.: Influence of brassinosteroids on pigment content of Glycine max L. (soybean) grown in dark and light. — J. appl. biol. Sci 2: 23–28, 2008.
Chen, S.M., Li, C.H., Zhu, X.R., Deng, Y.M., Sun, W., Wang, L.S., Chen F.D., Zhang, Z.: The identification of flavonoids and the expression of genes of anthocyanin biosynthesis in the chrysanthemum flowers. — Biol. Plant. 56: 458–464, 2012.
Choe, S., Dilkes, B.P., Fujioka, S., Takatsuto, S., Sakurai, A., Feldmann, K.A.: The DWF4 gene of Arabidopsis encodes a cytochrome P450 that mediates multiple 22α-hydroxylation steps in brassinosteroid biosynthesis. — Plant Cell 10: 231–243, 1998.
Chory, J., Nagpal, P., Peto, C.A.: Phenotypic and genetic analysis of det2, a new mutant that affects light-regulated seedling development in Arabidopsis. — Plant Cell 3: 445–459, 1991.
Choudhary, S.P., Yu, J.Q., Yamaguchi-Shinozaki, K., Shinozaki, K., Tran, L.S.P.: Benefits of brassinosteroid crosstalk. — Trends Plant Sci. 17: 594–605, 2012.
Cooney, L.J., Van Klink, J.W., Hughes, N.M., Perry, N.B., Schaefer, H.M., Menzies, I.J., Gould, K.S.: Red leaf margins indicate increased polygodial content and function as visual signals to reduce herbivory in Pseudowintera colorata. — New Phytol. 194: 488–497, 2012.
Dao, T.T.H., Linthorst, H.J.M., Verpoorte, R.: Chalcone synthase and its functions in plant resistance. — Phytochem. Rev. 10: 397–412, 2011.
Das, P.K., Shin, D.H., Choi, S.B., Park, Y.I.: Sugar-hormone cross-talk in anthocyanin biosynthesis. — Mol. Cells 34: 501–507, 2012a.
Das, P.K., Shin, D.H., Choi, S.B., Yoo, S.D., Choi, G., Park, Y.I.: Cytokinins enhance sugar-induced anthocyanin biosynthesis in Arabidopsis. — Mol. Cells 34: 93–101, 2012b.
Deikman, J., Hammer, P.E.: Induction of anthocyanin accumulation by cytokinins in Arabidopsis thaliana. — Plant Physiol. 108: 47–57, 1995.
Farooq, M., Wahid, A., Basra, S.M.A., Islam-ud-Din.: Improving water relations and gas exchange with brassinosteroids in rice under drought stress. — J. Agron. Crop Sci. 195: 262–269, 2009.
Gendron, J.M., Haque, A., Gendron, N., Chang, T., Asami, T., Wang, Z.Y.: Chemical genetic dissection of brassinosteroidethylene interaction. — Mol. Plant 1: 368–379, 2008.
Gonzalez, A., Zhao, M., Leavitt, J.M., Lloyd A.M.: Regulation of the anthocyanin biosynthetic pathway by the TTG1/bHLH/Myb transcriptional complex in Arabidopsis seedlings. — Plant J. 53: 814–827, 2008.
Gould, K.S.: Nature’s Swiss ar my knife: the diverse protective roles of anthocyanins in leaves. — J. Biomed. Biotechnol. 5: 314–320, 2004.
Gould, K.S., Dudle, D.A., Neufeld, H.S.: Why some stems are red: cauline anthocyanins shield photosystem II against high light stress. — J. exp. Bot. 61: 2707–2717, 2010.
Grotewold, E.: The genetics and biochemistry of floral pigments. — Annu. Rev. Plant Biol. 57: 761–780, 2006.
Guo, J., Hu, X., Duan, R.: Interactive effects of cytokinins, light, and sucrose on the phenotypes and the syntheses of anthocyanins and lignins in cytokinin overproducing transgenic Arabidopsis. — J. Plant Growth Regul. 24: 93–101, 2005.
Hatier, J.H.B., Clearwater, M.J., Gould, K.S.: The Functional significance of black-pigmented leaves: photosynthesis, photoprotection and productivity in Ophiopogon planiscapus ‘Nigrescens’. — PLoS ONE 8: e67850, 2013.
Hatier, J.H.B., Gould, K.S.: Foliar anthocyanins as modulators of stress signals. — J. theor. Biol. 253: 625–627, 2008.
Holton, T.A., Cornish, E.C.: Genetics and biochemistry of anthocyanin biosynthesis. — Plant Cell 7: 1071–1083, 1995.
Huang, Y., Han, C., Peng, W., Peng, Z., Xiong, X., Zhu, Q., Gao, B., Xie, D., Ren, C.: Brassinosteroid negatively regulates jasmonate inhibition of root growth in Arabidopsis. — Plant Signal. Behav. 5: 140–142, 2010.
Jenkins, G.I., Long, J.C., Wade, H.K., Shenton, M.R., Bibikova, T.N.: UV and blue light signalling: pathways regulating chalcone synthase gene expression in Arabidopsis. — New Phytol. 151: 121–131, 2001.
Jeong, S.W., Das, P.K., Jeoung, S.C., Song, J.Y., Lee, H.K., Kim, Y.K., Kim, W.J., Park, Y.I., Yoo, S.D., Choi, S.B., Choi, G., Park, Y.I.: Ethylene suppression of sugar-induced anthocyanin pigmentation in Arabidopsis. — Plant Physiol. 154: 1514–1531, 2010.
Koes, R.E., Quattrocchio, F., Mol, J.N.M.: The flavonoid biosynthetic pathway in plants: function and evolution. — Bioessays 16: 123–132, 1994.
Krishna, P.: Brassinosteroid-mediated stress responses. — J. Plant Growth Regul. 22: 289–297, 2003.
Kwon, Y., Oh, J.E., Noh, H., Hong, S.W., Bhoo, S.H., Lee, H.: The ethylene signaling pathway has a negative impact on sucrose-induced anthocyanin accumulation in Arabidopsis. — J. Plant Res. 124: 193–200, 2011.
Laxmi, A., Paul, L.K., Peters, J.L., Khurana, J.P.: Arabidopsis constitutive photomorphogenic mutant, bls1, displays altered brassinosteroid response and sugar sensitivity. — Plant mol. Biol. 56: 185–201, 2004.
Loreti, E., Povero, G., Novi, G., Solfanelli, C., Alpi, A., Perata, P.: Gibberellins, jasmonate and abscisic acid modulate the sucrose-induced expression of anthocyanin biosynthetic genes in Arabidopsis. — New Phytol. 179: 1004–1016, 2008.
Luan, L.Y., Zhang, Z.W., Xi, Z.M., Huo, S.-S., Ma, L.N.: Brassinosteroids regulate anthocyanin biosynthesis in the ripening of grape berries. — S. Afr. J. Enol. Viticult. 34: 196–203, 2013.
Luccioni, L.G., Oliverio, K.A., Yanovsky, M.J., Boccalandro, H.E., Casal, J.J.: Brassinosteroid mutants uncover fine tuning of phytochrome signaling. — Plant Physiol. 128: 173–181, 2002.
Mehdy, M.C., Lamb, C.J.: Chalcone isomerase cDNA cloning and mRNA induction by fungal elicitor, wounding and infection. — EMBO J. 6: 1527–1533, 1987.
Nagira, Y., Ikegami, K., Koshiba, T., Ozeki, Y.: Effect of ABA upon anthocyanin synthesis in regenerated Torenia shoots. — J Plant Res. 119: 137–144, 2006.
Nakamoto, D., Ikeura, A., Asami, T., Yamamoto, K.T.: Inhibition of brassinosteroid biosynthesis by either a dwarf4 mutation or a brassinosteroid biosynthesis inhibitor rescues defects in tropic responses of hypocotyls in the Arabidopsis mutant nonphototropic hypocotyl 4. — Plant Physiol. 141: 456–464, 2006.
Noguchi, T., Fujioka, S., Choe, S., Takatsuto, S., Yoshida, S., Yuan, H., Feldmann, K.A., Tax, F.E.: Brassinosteroid insensitive dwarf mutants of Arabidopsis accumulate brassinosteroids. — Plant Physiol. 121: 743–752, 1999.
Pelletier, M.K., Murrell, J.R., Shirley, B.W.: Characterization of flavonol synthase and leucoanthocyanidin dioxygenase genes in Arabidopsis (further evidence for differential regulation of “early” and “late” genes). — Plant Physiol. 113: 1437–1445, 1997.
Peng, Z., Han, C., Yuan, L., Zhang, K., Huang, H., Ren, C.: Brassinosteroid enhances jasmonate-induced anthocyanin accumulation in Arabidopsis seedlings. — J. Integr. Plant Biol. 53: 632–640, 2011.
Qi, T., Song, S., Ren, Q., Wu, D., Huang, H., Chen, Y., Fan, M., Peng, W., Ren, C., Xie, D.: The jasmonate-ZIM-domain proteins interact with the WD-repeat/bHLH/MYB complexes to regulate jasmonate-mediated anthocyanin accumulation and trichome initiation in Arabidopsis thaliana. — Plant Cell 23: 1795–1814, 2011.
Ren, C., Han, C., Peng, W., Huang, Y., Peng, Z., Xiong, X., Zhu, Q., Gao, B., Xie, D.: A leaky mutation in DWARF4 reveals an antagonistic role of brassinosteroid in the inhibition of root growth by jasmonate in Arabidopsis. — Plant Physiol. 151: 1412–1420, 2009.
Sasse, J.M.: Physiological actions of brassinosteroids: an update. — J. Plant Growth Regul. 22: 276–288, 2003.
Shan, X., Zhang, Y., Peng, W., Wang, Z., Xie, D.: Molecular mechanism for jasmonate-induction of anthocyanin accumulation in Arabidopsis. — J. exp. Bot. 60: 3849–3860, 2009.
Shin, D.H., Choi, M., Kim, K., Bang, G., Cho, M., Choi, S.B., Choi, G., Park, Y.I.: HY5 regulates anthocyanin biosynthesis by inducing the transcriptional activation of the MYB75/PAP1 transcription factor in Arabidopsis, — FEBS Lett. 587: 1543–1547, 2013.
Solfanelli, C., Poggi, A., Loreti, E., Alpi, A., Perata, P.: Sucrose-specific induction of the anthocyanin biosynthetic pathway in Arabidopsis. — Plant Physiol. 140: 637–646, 2006.
Springob, K., Nakajima, J., Yamazaki, M., Saito, K.: Recent advances in the biosynthesis and accumulation of anthocyanins. — Natur. Prod. Rep. 20: 288–303, 2003.
Symons, G.M., Davies, C., Shavrukov, Y., Dry, I.B., Reid, J.B., Thomas, M.R.: Grapes on steroids. Brassinosteroids are involved in grape berry ripening. — Plant Physiol. 140: 150–158, 2006.
Szekeres, M., Németh, K., Koncz-Kálmán, Z., Mathur, J., Kauschmann, A., Altmann, T., Rédei, G.P., Nagy, F., Schell, J., Koncz, C.: Brassinosteroids rescue the deficiency of CYP90, a cytochrome P450, controlling cell elongation and de-etiolation in Arabidopsis. — Cell 85: 171–182, 1996.
Teng, S., Keurentjes, J., Bentsink, L., Koornneef, M., Smeekens, S.: Sucrose-specific induction of anthocyanin biosynthesis in Arabidopsis requires the MYB75/PAP1 gene. — Plant Physiol. 139: 1840–1852, 2005.
Treutter, D.: Significance of flavonoids in plant resistance and enhancement of their biosynthesis. — Plant Biol. 7: 581–591, 2005.
Winkel-Shirley, B.: Biosynthesis of flavonoids and effects of stress. — Curr. Opin. Plant Biol. 5: 218–223, 2002.
Yokota, T.: The structure, biosynthesis and function of brassinosteroids. — Trends Plant Sci. 2: 137–143, 1997.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements: This work was supported by grants from the National Science Foundation of China (30770195), the Hunan Provincial Natural Science Foundation of China (12JJ2021), the Scientific Research Fund of Hunan Provincial Education Department (12C0166), the Hunan Provincial Graduate Student’s Innovative Research (CX2013B301), and the Program for Innovative Research Team in University (IRT1239). The authors L.B. YUAN, Z.H. PENG, and T.T. ZHI contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Yuan, L.B., Peng, Z.H., Zhi, T.T. et al. Brassinosteroid enhances cytokinin-induced anthocyanin biosynthesis in Arabidopsis seedlings. Biol Plant 59, 99–105 (2015). https://doi.org/10.1007/s10535-014-0472-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-014-0472-z