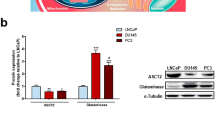
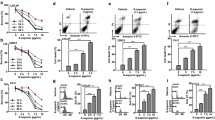
Abstract
Epibrassinolide (EBR) is a biologically active compound of the brassinosteroids, steroid-derived plant growth regulator family. Generally, brassinosteroids are known for their cell expansion and cell division-promoting roles. Recently, EBR was shown as a potential apoptotic inducer in various cancer cells without affecting the non-tumor cell growth. Androgen signaling controls cell proliferation through the interaction with the androgen receptor (AR) in the prostate gland. Initially, the development of prostate cancer is driven by androgens. However, in later stages, a progress to the androgen-independent stage is observed, resulting in metastatic prostate cancer. The androgen-responsive or -irresponsive cells are responsible for tumor heterogeneity, which is an obstacle to effective anti-cancer therapy. Polyamines are amine-derived organic compounds, known for their role in abnormal cell proliferation as well as during malignant transformation. Polyamine catabolism-targeting agents are being investigated against human cancers. Many chemotherapeutic agents including polyamine analogs have been demonstrated to induce polyamine catabolism that depletes polyamine levels and causes apoptosis in tumor models. In our study, we aimed to investigate the mechanism of apoptotic cell death induced by EBR, related with polyamine biosynthetic and catabolic pathways in LNCaP (AR+), DU145 (AR−) prostate cancer cell lines and PNT1a normal prostate epithelial cell line. Induction of apoptotic cell death was observed in prostate cancer cell lines after EBR treatment. In addition, EBR induced the decrease of intracellular polyamine levels, accompanied by a significant ornithine decarboxylase (ODC) down-regulation in each prostate cancer cell and also modulated ODC antizyme and antizyme inhibitor expression levels only in LNCaP cells. Catabolic enzymes SSAT and PAO expression levels were up-regulated in both cell lines; however, the specific SSAT and PAO siRNA treatments prevented the EBR-induced apoptosis only in LNCaP (AR+) cells. In a similar way, MDL 72,527, the specific PAO and SMO inhibitor, co-treatment with EBR during 24 h, reduced the formation of cleaved fragments of PARP in LNCaP (AR+) cells.








Similar content being viewed by others
Abbreviations
- AR:
-
Androgen receptor
- AZI:
-
Antizyme inhibitor
- BAK1:
-
BRI1-associated receptor kinase
- BRI1:
-
Brassinosteroid insensitive 1
- BR:
-
Brassinosteroid
- DCFH-DA:
-
2′,7′-Dichlorofluorescein diacetate
- DiOC6:
-
3,3′-Dihexyloxacarbocyanine iodide
- DMSO:
-
Dimethyl sulfoxide
- EBR:
-
Epibrassinolide
- HPLC:
-
High-pressure liquid chromatography
- HRP:
-
Horseradish peroxidase
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide
- OAZ:
-
ODC antizyme
- ODC:
-
Ornithine decarboxylase
- PA:
-
Polyamine
- PAO:
-
Polyamine oxidase
- PBS:
-
Phosphate-buffered saline
- PVDF:
-
Polyvinyldifluoride
- ROS:
-
Reactive oxygen species
- SDS-PAGE:
-
Sodium dodecyl sulfate polyacrylamide gel electrophoresis
- SSAT:
-
Spermidine/spermine N 1-acetyltransferase
- TBS-T:
-
Tris-buffered saline with Tween-20
- Δψm:
-
Mitochondrial membrane potential
References
Agostinelli E, Seiler N (2007) Lysosomotropic compounds and spermine enzymatic oxidation products in cancer therapy (review). Int J Oncol 31(3):473–484
Agostinelli E, Arancia G, Vedova LD, Belli F, Marra M, Salvi M, Toninello A (2004) The biological functions of polyamine oxidation products by amine oxidases: perspectives of clinical applications. Amino Acids 27(3–4):347–358
Agostinelli E, Condello M, Molinari A, Tempera G, Viceconte N, Arancia G (2009) Cytotoxicity of spermine oxidation products to multidrug resistant melanoma M14 ADR2 cells: sensitization by the MDL 72527 lysosomotropic compound. Int J Oncol 35(3):485–498
Agostinelli E, Tempera G, Viceconte N, Saccoccio S, Battaglia V, Grancara S, Toninello A, Stevanato R (2010) Potential anticancer application of polyamine oxidation products formed by amine oxidase: a new therapeutic approach. Amino Acids 38(2):353–368
Ali B, Hayat S, Fariduddin Q, Ahmad A (2008) 24-epibrassinolide protects against the stress generated by salinity and nickel in Brassica juncea. Chemosphere 72(9):1387–1392
Allen WL, McLean EG, Boyer J, McCulla A, Wilson PM, Coyle V, Longley DB, Casero RA Jr, Johnston PG (2007) The role of spermidine/spermine N 1-acetyltransferase in determining response to chemotherapeutic agents in colorectal cancer cells. Mol Cancer Ther 6(1):128–137
Arisan ED, Obakan P, Coker A, Palavan-Unsal N (2012) Inhibition of ornithine decarboxylase alters the roscovitine-induced mitochondrial-mediated apoptosis in MCF-7 breast cancer cells. Mol Med Rep 5(5):1323–1329
Basu HS, Thompson TA, Church DR, Clower CC, Mehraein-Ghomi F, Amlong CA, Martin CT, Woster PM, Lindstrom MJ, Wilding G (2009) A small molecule polyamine oxidase inhibitor blocks androgen-induced oxidative stress and delays prostate cancer progression in the transgenic adenocarcinoma of the mouse prostate model. Cancer Res 69(19):7689–7695
Bishop GJ, Koncz C (2002) Brassinosteroids and plant steroid hormone signaling. Plant Cell 14(Suppl):S97–S110
Carange J, Longpre F, Daoust B, Martinoli MG (2011) 24-epibrassinolide, a phytosterol from the brassinosteroid family, protects dopaminergic cells against MPP-induced oxidative stress and apoptosis. J Toxicol 2011:392859
Coker A, Arisan ED, Palavan-Unsal N (2012) Silencing of the polyamine catabolic key enzyme SSAT prevents CDK inhibitor-induced apoptosis in Caco-2 colon cancer cells. Mol Med Rep 5(4):1037–1042
Devereux W, Wang Y, Stewart TM, Hacker A, Smith R, Frydman B, Valasinas AL, Reddy VK, Marton LJ, Ward TD, Woster PM, Casero RA (2003) Induction of the PAOh1/SMO polyamine oxidase by polyamine analogues in human lung carcinoma cells. Cancer Chemother Pharmacol 52(5):383–390
Gerner EW, Meyskens FL Jr (2004) Polyamines and cancer: old molecules, new understanding. Nat Rev Cancer 4(10):781–792
Hector S, Tummala R, Kisiel ND, Diegelman P, Vujcic S, Clark K, Fakih M, Kramer DL, Porter CW, Pendyala L (2008) Polyamine catabolism in colorectal cancer cells following treatment with oxaliplatin, 5-fluorouracil and N 1, N 11 diethylnorspermine. Cancer Chemother Pharmacol 62(3):517–527
Hu Y, Bao F, Li J (2000) Promotive effect of brassinosteroids on cell division involves a distinct CycD3-induction pathway in Arabidopsis. Plant J 24(5):693–701
Kahana C (2009) Antizyme and antizyme inhibitor, a regulatory tango. Cell Mol Life Sci 66(15):2479–2488
Krauspe R, Scheer A (1986) Coomassie brilliant blue G-250 dye-binding technique for determination of autolytic protein breakdown in Euglena gracilis and comparison to other methods of autolysis measurement. Anal Biochem 153(2):242–250
Kumi-Diaka J, Sanderson NA, Hall A (2000) The mediating role of caspase-3 protease in the intracellular mechanism of genistein-induced apoptosis in human prostatic carcinoma cell lines, DU145 and LNCaP. Biol Cell 92(8–9):595–604
Lawson KR, Marek S, Linehan JA, Woster PM, Casero RA Jr, Payne CM, Gerner EW (2002) Detoxification of the polyamine analogue N 1-ethyl-N 11-[(cycloheptyl)methy]-4,8-diazaundecane (CHENSpm) by polyamine oxidase. Clin Cancer Res 8(5):1241–1247
Li J, Wen J, Lease KA, Doke JT, Tax FE, Walker JC (2002) BAK1, an Arabidopsis LRR receptor-like protein kinase, interacts with BRI1 and modulates brassinosteroid signaling. Cell 110(2):213–222
Malikova J, Swaczynova J, Kolar Z, Strnad M (2008) Anticancer and antiproliferative activity of natural brassinosteroids. Phytochemistry 69(2):418–426
Nam KH, Li J (2002) BRI1/BAK1, a receptor kinase pair mediating brassinosteroid signaling. Cell 110(2):203–212
Nowotarski SL, Woster PM, Casero RA (2013) Polyamines and cancer: implications for chemotherapy and chemoprevention. Expert Rev Mol Med 15:e3
Olsen RR, Chung I, Zetter BR (2012) Knockdown of antizyme inhibitor decreases prostate tumor growth in vivo. Amino Acids 42(2–3):549–558
Romanutti C, Castilla V, Coto CE, Wachsman MB (2007) Antiviral effect of a synthetic brassinosteroid on the replication of vesicular stomatitis virus in Vero cells. Int J Antimicrob Agents 29(3):311–316
Schipper RG, Romijn JC, Cuijpers VM, Verhofstad AA (2003) Polyamines and prostatic cancer. Biochem Soc Trans 31(2):375–380
Seiler N, Delcros JG, Moulinoux JP (1996) Polyamine transport in mammalian cells. An update. Int J Biochem Cell Biol 28(8):843–861
Singh AB, Thomas TJ, Thomas T, Singh M, Mann RA (1992) Differential effects of polyamine homologues on the prevention of DL-alpha-difluoromethylornithine-mediated inhibition of malignant cell growth and normal immune response. Cancer Res 52(7):1840–1847
Steigerova J, Oklestkova J, Levkova M, Rarova L, Kolar Z, Strnad M (2010) Brassinosteroids cause cell cycle arrest and apoptosis of human breast cancer cells. Chem Biol Interact 188(3):487–496
Steigerova J, Rarova L, Oklest’kova J, Krizova K, Levkova M, Svachova M, Kolar Z, Strnad M (2012) Mechanisms of natural brassinosteroid-induced apoptosis of prostate cancer cells. Food Chem Toxicol 50(11):4068–4076
Su KL, Liao YF, Hung HC, Liu GY (2009) Critical factors determining dimerization of human antizyme inhibitor. J Biol Chem 284(39):26768–26777
Taplin ME, Ho SM (2001) Clinical review 134: the endocrinology of prostate cancer. J Clin Endocrinol Metab 86(8):3467–3477
Wang X, Feith DJ, Welsh P, Coleman CS, Lopez C, Woster PM, O’Brien TG, Pegg AE (2007) Studies of the mechanism by which increased spermidine/spermine N 1-acetyltransferase activity increases susceptibility to skin carcinogenesis. Carcinogenesis 28(11):2404–2411
Wu YD, Lou YJ (2007) Brassinolide, a plant sterol from pollen of Brassica napus L., induces apoptosis in human prostate cancer PC-3 cells. Pharmazie 62(5):392–395
Acknowledgments
This work was partially supported by Istanbul University Research Projects Center (Project no: 20958), Istanbul Kultur University Scientific Projects Support Center; Italian MIUR (Ministero dell’Istruzione, dell’Università e della Ricerca) (EA); Istituto Superiore di Sanità “Project Italy–USA” (EA); funds MIUR-PRIN (Cofin) (EA) and Istituto Pasteur Fondazione Cenci Bolognetti (EA).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
726_2013_1574_MOESM1_ESM.pdf
EBR did not modulate cell cycle distribution and induce apoptosis in PNT1a prostate epithelial cells (a) PNT1a cells were seeded into a 6-well plate and treated with EBR (25 μM) for 24 h. After ethanol fixation and PI (propidium iodide) staining, samples were analyzed by flow cytometry. Cell cycle distribution of was determined by CellQuest software. Data shown are obtained from a representative experiment (b) Total proteins were isolated from PNT1a cells; the expression profiles of Apaf-1 and cleaved caspase-9 were determined by immunoblotting following 24 h EBR treatment while PARP expression was analyzed at 24, 48 and 72 h. β-actin was used as a loading control. (PDF 102 kb)
726_2013_1574_MOESM2_ESM.pdf
SSAT and PAO silencing determination with qRT-PCR. Cells were seeded in 6 well plates and treated with EBR for 24 h. Total RNA was isolated and cDNA synthesis was perfomed. To determine the efficiency of selected concentration of siRNA to silence (a) SSAT or (b) PAO, qRT-PCR with appropriate primers was performed. (PDF 62 kb)
Rights and permissions
About this article
Cite this article
Obakan, P., Arisan, E.D., Calcabrini, A. et al. Activation of polyamine catabolic enzymes involved in diverse responses against epibrassinolide-induced apoptosis in LNCaP and DU145 prostate cancer cell lines. Amino Acids 46, 553–564 (2014). https://doi.org/10.1007/s00726-013-1574-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-013-1574-1