Abstract
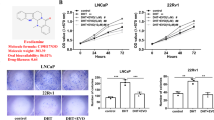
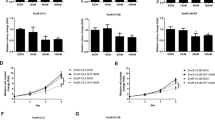
The androgen receptor (AR) plays an important role in the pathogenesis and development of prostate cancer (PCa). Mostly, PCa progresses to androgen-independent PCa, which has activated AR signaling from androgen-dependent PCa. Thus, inhibition of AR signaling may be an important therapeutic target in androgen-dependent and castration-resistant PCa. In this study, we determined the anticancer effect of a newly found natural compound, sakurasosaponin (S-saponin), using androgen-dependent and castration-resistant PCa cell lines. S-saponin induces mitochondrial-mediated cell death in both androgen-dependent (LNCaP) and castration-resistant (22Rv1 and C4-2) PCa cells, via AR expression. S-saponin treatment induces a decrease in AR expression in a time- and dose-dependent manner and a potent decrease in the expression of its target genes, including prostate-specific antigen (PSA), transmembrane protease, serin 2 (TMPRSS2), and NK3 homeobox 1 (NKX3.1). Furthermore, S-saponin treatment decreases B-cell lymphoma-extra large (Bcl-xL) and mitochondrial membrane potential, thereby increasing the release of cytochrome c into the cytosol. Moreover, Bcl-xL inhibition and subsequent mitochondria-mediated cell death caused by S-saponin were reversed by Bcl-xL or AR overexpression. Interestingly, S-saponin-mediated cell death was significantly reduced by a reactive oxygen species (ROS) scavenger, N-acetylcystein. Animal xenograft experiments showed that S-saponin treatment significantly reduced tumor growth of AR-positive 22Rv1 xenografts but not AR-negative PC-3 xenografts. Taken together, for the first time, our results revealed that S-saponin induces mitochondrial-mediated cell death in androgen-dependent and castration-resistant cells through regulation of AR mechanisms, including downregulation of Bcl-xL expression and induction of ROS stress by decreasing mitochondrial membrane potential.







Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136(5):E359–386
Taitt HE (2018) Global trends and prostate cancer: a review of incidence, detection, and mortality as influenced by race, ethnicity, and geographic location. Am J Mens Health 12:(6):1807–1823
Fujita K, Nonomura N (2018) Role of androgen receptor in prostate cancer: a review. World J Mens Health 10(36):180040
Massard C, Fizazi K Targeting continued androgen receptor signaling in prostate cancer. Clin Cancer Res 17(12):3876–3883
Tan MH, Li J, Xu HE, Melcher K, Yong EL (2015) Androgen receptor: structure, role in prostate cancer and drug discovery. Acta Pharmacol Sin 361:(1):3–23. https://doi.org/10.1371/journal.pgen.1006946
Isbarn H, Boccon-Gibod L, Carroll PR, Montorsi F, Schulman C, Smith MR, Sternberg CN, Studer UE (2009) Androgen deprivation therapy for the treatment of prostate cancer: consider both benefits and risks. Eur Urol 55(1):62–75
Cai Z, Chen W, Zhang J, Li H (2018) Androgen receptor: what we know and what we expect in castration-resistant prostate cancer. Int Urol Nephrol 50(10):1753–1764
Dong L, Zieren RC, Xue W, de Reijke TM, Pienta KJ (2019) Metastatic prostate cancer remains incurable, why? Asian J Urol 6(1):26–41
Watson PA, Arora VK, Sawyers CL (2015) Emerging mechanisms of resistance to androgen receptor inhibitors in prostate cancer. Nat Rev Cancer 15(12):701–711
Bluemn EG, Nelson PS (2012) The androgen/androgen receptor axis in prostate cancer. Curr Opin Oncol 24(3):251–257
Culig Z (2014) Targeting the androgen receptor in prostate cancer. Expert Opin Pharmacother 15(10):1427–1437
Nevedomskaya E, Baumgart SJ, Haendler B (2018) Recent advances in prostate cancer treatment and drug discovery. Int J Mol Sci 19(5):E1359
Ahmed A, Ali S, Sarkar FH (2014) Advances in androgen receptor targeted therapy for prostate cancer. J Cell Physiol 229(3):271–276
Chao DT, Korsmeyer SJ (1998) BCL-2 family: regulators of cell death. Annu Rev Immunol 16:395–419
Chan SL, Yu VC (2004) Proteins of the bcl-2 family in apoptosis signalling: from mechanistic insights to therapeutic opportunities. Clin Exp Pharmacol Physiol 31(3):119–128
Yamaguchi H, Wang HG (2002) Bcl-XL protects BimEL-induced Bax conformational change and cytochrome C release independent of interacting with Bax or BimEL. J Biol Chem 277(44):41604–41612
Sasazawa Y, Futamura Y, Tashiro E, Imoto M (2009) Vacuolar H+-ATPase inhibitors overcome Bcl-xL-mediated chemoresistance through restoration of a caspase-independent apoptotic pathway. Cancer Sci 100(8):1460–1467
Grad JM, Zeng XR, Boise LH (2000) Regulation of Bcl-xL: a little bit of this and a little bit of STAT. Curr Opin Oncol 12(6):543–549
Lebedeva I, Rando R, Ojwang J, Cossum P, Stein CA (2000) Bcl-xL in prostate cancer cells: effects of overexpression and down-regulation on chemosensitivity. Cancer Res 60(21):6052–6060
Lin Y, Fukuchi J, Hiipakka RA, Kokontis JM, Xiang J (2007) Up-regulation of Bcl-2 is required for the progression of prostate cancer cells from an androgen-dependent to an androgen-independent growth stage. Cell Res 17(6):531–536
Castilla C, Congregado B, Chinchon D, Torrubia FJ, Japon MA, Saez C (2006) Bcl-xL is overexpressed in hormone-resistant prostate cancer and promotes survival of LNCaP cells via interaction with proapoptotic Bak. Endocrinology 147(10):4960–4967
Sun A, Tang J, Hong Y, Song J, Terranova PF, Thrasher JB, Svojanovsky S, Wang HG, Li B (2008) Androgen receptor-dependent regulation of Bcl-xL expression: Implication in prostate cancer progression. Prostate 68(4):453–461
Liao X, Tang S, Thrasher JB, Griebling TL, Li B (2005) Small-interfering RNA-induced androgen receptor silencing leads to apoptotic cell death in prostate cancer. Mol Cancer Ther 4(4):505–515
Vinh LB, Nguyet NTM, Yang SY, Kim JH, Thanh NV, Cuong NX, Nam NH, Minh CV, Hwang I, Kim YH (2019) Cytotoxic triterpene saponins from the mangrove Aegiceras corniculatum. Nat Prod Res 33(5):628–634
Garcia-Sosa K, Sanchez-Medina A, Alvarez SL, Zacchino S, Veitch NC, Sima-Polanco P, Pena-Rodriguez LM (2011) Antifungal activity of sakurasosaponin from the root extract of Jacquinia flammea. Nat Prod Res 25(12):1185–1189
Sanchez-Medina A, Pena-Rodriguez LM, May-Pat F, Karagianis G, Waterman PG, Mallet AI, Habtemariam S (2010) Identification of sakurasosaponin as a cytotoxic principle from Jacquinia flammea. Nat Prod Commun 5(3):365–368
Merseburger AS, Hupe MC (2016) An update on triptorelin: current thinking on androgen deprivation therapy for prostate cancer. Adv Ther 33(7):1072–1093
Banerjee PP, Banerjee S, Brown TR, Zirkin BR (2018) Androgen action in prostate function and disease. Am J Clin Exp Urol 6(2):62–77
Newman DJ, Cragg GM (2016) Natural products as sources of new drugs from 1981 to 2014. J Nat Prod 79(3):629–661
Cragg GM (1830) Newman DJ (2013) Natural products: a continuing source of novel drug leads. Biochim Biophys Acta 6:3670–3695
Kim R (2005) Unknotting the roles of Bcl-2 and Bcl-xL in cell death. Biochem Biophys Res Commun 333(2):336–343
Antignani A, Youle RJ (2006) How do Bax and Bak lead to permeabilization of the outer mitochondrial membrane? Curr Opin Cell Biol 18(6):685–689
Kim R, Emi M, Tanabe K (2005) Caspase-dependent and -independent cell death pathways after DNA damage (Review). Oncol Rep 14(3):595–599
Kim R, Emi M, Tanabe K (2006) Role of mitochondria as the gardens of cell death. Cancer Chemother Pharmacol 57(5):545–553
Kuwana T, Newmeyer DD (2003) Bcl-2-family proteins and the role of mitochondria in apoptosis. Curr Opin Cell Biol 15(6):691–699
Kim E, Jung Y, Choi H, Yang J, Suh JS, Huh YM, Kim K, Haam S (2010) Prostate cancer cell death produced by the co-delivery of Bcl-xL shRNA and doxorubicin using an aptamer-conjugated polyplex. Biomaterials 31(16):4592–4599
Yamanaka K, Rocchi P, Miyake H, Fazli L, So A, Zangemeister-Wittke U, Gleave ME (2006) Induction of apoptosis and enhancement of chemosensitivity in human prostate cancer LNCaP cells using bispecific antisense oligonucleotide targeting Bcl-2 and Bcl-xL genes. BJU Int 97(6):1300–1308
Meng Y, Tang W, Dai Y, Wu X, Liu M, Ji Q, Ji M, Pienta K, Lawrence T, Xu L (2008) Natural BH3 mimetic (-)-gossypol chemosensitizes human prostate cancer via Bcl-xL inhibition accompanied by increase of Puma and Noxa. Mol Cancer Ther 7(7):2192–2202
Pilling AB, Hwang C (2019) Targeting prosurvival BCL2 signaling through Akt blockade sensitizes castration-resistant prostate cancer cells to enzalutamide. Prostate 79(11):1347–1359
Funding
This work was supported by the “Procurement and development of foreign biological resources” funded by the Ministry of Science ICT and Future Planning of the Korean government (NRF-2016K1A1A8A01938718) and the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2018R1D1A1B07047406 and NRF-2018R1D1A1B07044392).
Author information
Authors and Affiliations
Contributions
I-SS and S-WJ designed the research; K-HS and N-IB isolated and identified S-saponin; I-SS, YJJ, JEK, and JS performed the research; I-SS, YJJ, JEK, JS, and S-WJ analyzed the data; and I-SS and S-WJ wrote the paper.
Corresponding authors
Ethics declarations
Conflict of interest
No potential conflicts of interest are disclosed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Song, IS., Jeong, Y.J., Kim, J. et al. Pharmacological inhibition of androgen receptor expression induces cell death in prostate cancer cells. Cell. Mol. Life Sci. 77, 4663–4673 (2020). https://doi.org/10.1007/s00018-019-03429-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-019-03429-2