Abstract
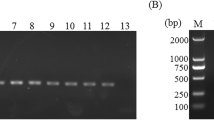
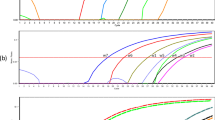
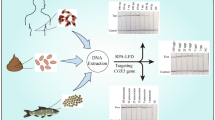
A real-time PCR assay targeting the cytochrome c oxidase gene of Clonorchis sinensis was developed and its diagnostic value was investigated in experimentally infected rats. The PCR assay amplified as little as 0.1 pg of C. sinensis genomic DNA and a single egg from experimentally spiked negative feces. The assay could detect C. sinensis DNA in feces 2 weeks post-infection (PI), whereas the Kato–Katz method and formalin–ether concentration techniques could detect the eggs in the feces from 3 weeks PI. Inverse correlations between eggs per gram of feces (EPG) and Ct values were observed 4, 6, and 8 weeks PI. Worm burden was positively correlated with EPG but negatively correlated with Ct values in different groups at 8 weeks PI. Fecal real-time PCR provides a potential tool for the diagnosis of clonorchiosis before eggs can be detected by conventional methods, and may be used to estimate the worm burden in the host.




Similar content being viewed by others
References
Bouvard V, Baan R, Straif K, Grosse Y, Secretan B, El Ghissassi F, Benbrahim-Tallaa L, Guha N, Freeman C, Galichet L, Cogliano V (2009) A review of human carcinogens—part B: biological agents. Lancet Oncol 10(4):321–322. doi:10.1016/S1470-2045(09)70096-8
Choi MH, Park IC, Li S, Hong ST (2003) Excretory–secretory antigen is better than crude antigen for the serodiagnosis of clonorchiasis by ELISA. Korean J Parasitol 41(1):35–39. doi:10.3347/kjp.2003.41.1.35
Choi D, Lim JH, Lee KT, Lee JK, Choi SH, Heo JS, Jang KT, Lee NY, Kim S, Hong ST (2006) Cholangiocarcinoma and Clonorchis sinensis infection: a case–control study in Korea. J Hepatol 44(6):1066–1073. doi:10.1016/j.jhep.2005.11.040
Chung DI, Choi DW (1988) Intensity of infection and development of adult Clonorchis sinensis in hamsters. Korean J Parasitol 26(1):9–14
Guy RA, Payment P, Krull UJ, Horgen PA (2003) Real-time PCR for quantification of Giardia and Cryptosporidium in environmental water samples and sewage. Appl Environ Microbiol 69(9):5178–5185. doi:10.1128/AEM.69.9.5178-5185.2003
Hanahan D (1983) Studies on transformation of Escherichia coli with plasmids. J Mol Biol 166(4):557–580. doi:10.1016/S0022-2836(83)80284-8
Hong ST (2003) Clonorchis sinensis. In: Miliotis MD, Bier JW (eds) International handbook of foodborne pathogens. Marcel Dekker, New York, pp 581–592
Hong ST, Rim HJ, Min DY, Li X, Xu J, Feng Z, Lee SH (2001) Control of clonorchiasis by repeated treatments with praziquantel. Korean J Parasitol 39(4):285–292. doi:10.3347/kjp.2001.39.4.285
Hong ST, Choi MH, Kim CH, Chung BS, Ji Z (2003) The Kato–Katz method is reliable for diagnosis of Clonorchis sinensis infection. Diagn Microbiol Infect Dis 47(1):345–347
Kim EM, Verweij JJ, Jalili A, van Lieshout L, Choi MH, Bae YM, Lim MK, Hong ST (2009) Detection of Clonorchis sinensis in stool samples using real-time PCR. Ann Trop Med Parasitol 103(6):513–518. doi:10.1179/136485909X451834
Korea Association of Health Promotion (2004) Prevalence of intestinal parasitic infections in Korea—the seventh report. Korea Association of Health Promotion, Korea
Le TH, Van De N, Blair D, Sithithaworn P, McManus DP (2006) Clonorchis sinensis and Opisthorchis viverrini: development of a mitochondrial-based multiplex PCR for their identification and discrimination. Exp Parasitol 112(2):109–114. doi:10.1016/j.exppara.2005.09.012
Li S, Kang HW, Choi MH, Hong ST (2006) Long-term storage of Clonorchis sinensis metacercariae in vitro. Parasitol Res 100(1):25–29. doi:10.1007/s00436-006-0242-8
Lim MK, Ju YH, Franceschi S, Oh JK, Kong HJ, Hwang SS, Park SK, Cho SI, Sohn WM, Kim DI, Yoo KY, Hong ST, Shin HR (2006) Clonorchis sinensis infection and increasing risk of cholangiocarcinoma in the Republic of Korea. Am J Trop Med Hyg 75(1):93–96
McManus DP, Le TH, Blair D (2004) Genomics of parasitic flatworms. Int J Parasitol 34(2):153–158. doi:10.1016/j.ijpara.2003.11.003
Monteiro L, Bonnemaison D, Vekris A, Petry KG, Bonnet J, Vidal R, Cabrita J, Megraud F (1997) Complex polysaccharides as PCR inhibitors in feces: Helicobacter pylori model. J Clin Microbiol 35(4):995–998
Müller B, Schmidt J, Mehlhorn H (2007) PCR diagnosis of infections with different species of Opisthorchiidae using a rapid clean-up procedure for stool samples and specific primers. Parasitol Res 100(4):905–909. doi:10.1007/s00436-006-0321-x
Park GM (2007) Genetic comparison of liver flukes, Clonorchis sinensis and Opisthorchis viverrini, based on rDNA and mtDNA gene sequences. Parasitol Res 100(2):351–357. doi:10.1007/s00436-006-0269-x
Rim HJ (2005) Clonorchiasis: an update. J Helminthol 79(3):269–281
Sandoval N, Siles-Lucas M, Lopez Aban J, Perez-Arellano JL, Garate T, Muro A (2006) Schistosoma mansoni: a diagnostic approach to detect acute schistosomiasis infection in a murine model by PCR. Exp Parasitol 114(2):84–88. doi:10.1016/j.exppara.2006.02.012
Sato M, Thaenkham U, Dekumyoy P, Waikagul J (2009) Discrimination of O. viverrini, C. sinensis, H. pumilio and H. taichui using nuclear DNA-based PCR targeting ribosomal DNA ITS regions. Acta Trop 109(1):81–83. doi:10.1016/j.actatropica.2008.09.015
Shen C, Kim J, Lee JK, Bae YM, Choi MH, Oh JK, Lim MK, Shin HR, Hong ST (2007) Collection of Clonorchis sinensis adult worms from infected humans after praziquantel treatment. Korean J Parasitol 45(2):149–152
ten Hove RJ, Verweij JJ, Vereecken K, Polman K, Dieye L, van Lieshout L (2008) Multiplex real-time PCR for the detection and quantification of Schistosoma mansoni and S. haematobium infection in stool samples collected in northern Senegal. Trans R Soc Trop Med Hyg 102(2):179–185. doi:10.1016/j.trstmh.2007.10.011
Thaenkham U, Visetsuk K, do Dung T, Waikagul J (2007) Discrimination of Opisthorchis viverrini from Haplorchis taichui using COI sequence marker. Acta Trop 103(1):26–32. doi:10.1016/j.actatropica.2007.05.006
Traub RJ, Macaranas J, Mungthin M, Leelayoova S, Cribb T, Murrell KD, Thompson RC (2009) A new PCR-based approach indicates the range of Clonorchis sinensis now extends to Central Thailand. PLoS Negl Trop Dis 3(1):e367. doi:10.1371/journal.pntd.0000367
Verweij JJ, Pit DS, van Lieshout L, Baeta SM, Dery GD, Gasser RB, Polderman AM (2001) Determining the prevalence of Oesophagostomum bifurcum and Necator americanus infections using specific PCR amplification of DNA from faecal samples. Trop Med Int Health 6(9):726–731
Verweij JJ, Brienen EA, Ziem J, Yelifari L, Polderman AM, Van Lieshout L (2007) Simultaneous detection and quantification of Ancylostoma duodenale, Necator americanus, and Oesophagostomum bifurcum in fecal samples using multiplex real-time PCR. Am J Trop Med Hyg 77(4):685–690
Wykoff DE (1959) Studies on Clonorchis sinensis. IV. Production of eggs in experimentally infected rabbits. J Parasitol 45(1):91–94
Xia CM, Rong R, Lu ZX, Shi CJ, Xu J, Zhang HQ, Gong W, Luo W (2009) Schistosoma japonicum: a PCR assay for the early detection and evaluation of treatment in a rabbit model. Exp Parasitol 121(2):175–179. doi:10.1016/j.exppara.2008.10.017
Acknowledgements
This study was supported by grant from the Seoul National University College of Medicine Research Fund (800-20050250).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rahman, S.M.M., Bae, Y.M., Hong, ST. et al. Early detection and estimation of infection burden by real-time PCR in rats experimentally infected with Clonorchis sinensis . Parasitol Res 109, 297–303 (2011). https://doi.org/10.1007/s00436-011-2253-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-011-2253-3