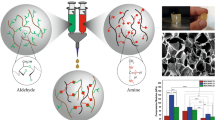
Abstract
Microfiber assemblies prepared from ellagic acid (EA) were functionalized with histidine (His) and dispersed in hyaluronic acid (HA) hydrogel microstructures. Swelling studies indicated that the hybrids had a relatively lower water uptake compared to HA and was pH dependent. The percentage swelling ratio for EA–His–HA hybrids was 48 % when 0.04 mg/mL of HA was incorporated and increased to 70 % when 1.2 mg/mL HA was integrated. Release studies using the dye crystal violet (CV) as a model drug showed that the rates were concentration-dependent. Further the hybrids were found to be thermally stable compared to HA. Cellular toxicity assays performed with normal rat kidney (NRK) cells indicated biocompatibility and adherence of the hybrids to the cells. Thus, we have developed a new family of hybrid hydrogels which readily formed on the EA–His functionalized microfibers and may have potential applications in drug delivery or tissue regeneration applications.









Similar content being viewed by others
References
Langer RS, Peppas NA (1981) Present and future applications of biomaterials in controlled drug delivery. Biomaterials 2:201–204
Nair LS, Laurencin CT (2006) Polymers as biomaterials for tissue engineering and controlled drug delivery. Adv Biochem Eng Biotechnol 102:47–90
Eastoe JE, Leach AA (1977) In: Ward AG, Courts A (eds) The science and technology of gelatin. Academic, New York
Tabata Y, Ikda Y (1993) Targeting of muramyl dipeptide to macrophages by gelatin conjugation to enhance their in vivo antitumor activity. J Control Release 27:79–88
Dash M, Chiellini F, Ottenbrite RM, Chiellini E (2011) Chitosan—a versatile semi-synthetic polymer in biomedical applications. Prog Polym Sci 36:981–1014
Quinoñones JP, Szopko R, Schmidt C, Covas CP (2011) Novel drug delivery systems: chitosan conjugates covalently attached to steroids with potential anticancer and agrochemical activity. Carbohydr Polym 84:858–864
Hu SG, Jou CH, Yang MC (2004) Biocompatibility and antibacterial activity of chitosan and collagen immobilized poly (3-hydroxybutryic acid-co-3-hydroxyvaleric acid). Carbohydr Polym 58:173–179
Thanou M, Duncan R (2003) Polymer-protein and polymer-drug conjugates in cancer therapy. Curr Opin Investig Drugs 4:701–709
Katre NV (1993) The conjugation of proteins with polyethylene glycol and other polymers: altering properties of proteins to enhance their therapeutic potential. Adv Drug Deliv Rev 10:91–114
Oh EJ, Park K, Kim KS, Kim J, Yang JA, Kong JH, Lee MY, Hoffman AS, Hahn SK (2010) Target specific and long-acting delivery of protein, peptide and nucleotide therapeutics using hyaluronic acid derivatives. J Control Release 141:2–12
Xie Y, Upton Z, Richards S, Rizzi SC, Leavesley DL (2011) Hyaluronic acid: evaluation as a potential delivery vehicle for vitronectin: growth factor complexes in wound healing applications. J Control Release 153:225–232
Shu XZ, Liu Y, Luo Y, Roberts MC, Prestwich GD (2002) Disulfide cross-linked hyaluronan hydrogels. Biomacromolecules 3:1304–1311
Eckici S, Iigin P, Butun S, Sahiner N (2011) Hyaluronic acid hydrogel particles with tunable charges as potential drug delivery devices. Carbohydr Polym 84:1306–1313
Saravanakumar G, Choi KY, Yoon HY, Kim K, Park JH, Kwon IC, Park K (2010) Hydrotropic hyaluronic acid conjugates: synthesis, characterization, and implications as a carrier of paclitaxel. Int J Pharm 394:154–161
Dong X, Liu C (2010) Preparation and characterization of self-assembled nanoparticles of hyaluornic acid-deoxycholic acid conjugates. J Nanomater. doi:10.1155/2010/906936
Manju S, Sreenivasan K (2011) Conjugation of curcumin onto hyaluronic acid enhances its aqueous solubility and stability. J Colloid Interface Sci 359:318–325
Li F, Bae BC, Na K (2010) Acetylated hyaluronic acid/photosensitizer conjugate for the preparation of nanogels with controllable phototoxicity: synthesis, characterization, autophotoquenching properties, and in vitro phototoxicity against HeLa cells. Bioconjug Chem 21:1312–1320
Liu CC, Liu KW, Chang YJ, Wang YJ (2010) A novel biodegradable amphiphilic diblock copolymers based on poly(lactic acid) and hyaluronic acid biomaterials for drug delivery. J Polym Res 17:459–469
Daniel EM, Krupnick AS, Heur YH, Blinzler JA, Nims RW, Stoner GD (1989) Extraction, stability and quantitation of ellagic acid in various fruits and nuts. J Food Compos Anal 2:338–349
Sharma M, Li L, Celver J, Killian C, Kovoor A, Seeram NP (2010) Effects of fruit ellagitanin extracts, ellagic acid and their colonic metabolite, urolithin A, on Wnt signaling. J Agric Food Chem 58:3965–3969
Makena PS, Chung KT (2007) Effects of various plant polyphenols on bladder carcinogen benzidine-induced mutagenicity. Food Chem Toxicol 45:1899–1909
Kim S, Nishimoto SK, Bumgardner JD, Haggard WO, Gaber MW, Yang YA (2010) Chitosan/β-glycerophosphate thermo-sensitive gel for the delivery of ellagic acid for the treatment of brain cancer. Biomaterials 31:4157–4166
Kim S, Gaber MW, Zawaski JA, Zhang F, Richardson M, Zhang XA, Yang Y (2009) The inhibition of glioma growth in vitro and in vivo by a chitosan/ellagic acid composite biomaterial. Biomaterials 30:4743–4751
Sharma G, Italia JL, Sonaje K, Tikoo K, Ravi Kumar MNV (2007) Biodegradable in situ gelling system for subcutaneous administration of ellagic acid and ellagic acid loaded nanoparticles: evaluation of their antioxidant potential against cyclosporine induced nephrotoxicity in rats. J Control Release 118:27–37
Yamanlar S, Sant S, Boudou T, Picart C, Khademhosseini A (2011) Surface functionalization of hyaluronic acid hydrogels by polyelectrolyte multilayer films. Biomaterials 32:5590–5599
Lao UL, Sun M, Matsumoto M, Mulchandani A, Chen W (2007) Genetic engineering of self-assembled protein hydrogel based on elastin-like sequences with metal binding functionality. Biomacromolecules 8:3736–3739
Bhattacharya S, Guillot S, DAbboue H, Tranchant JF, Salvetat JP (2008) Carbon nanotubes as structural nanofibers for hyaluronic acid hydrogel scaffolds. Biomacromolecules 9:505–509
Li H, Wang DQ, Liu BL, Gao LZ (2004) Synthesis of a novel gelatin-carbon nanotubes hybrid hydrogel. Colloids Surf B 33:85–88
Wang ZM, Chen YM (2007) Supramolecular hydrogels hybridized with single-walled carbon nanotubes. Macromolecules 40:3402–3407
Kawaguchi M, Fukushima T, Hayakawa T, Nakashima N, Inoue Y, Takeda S, Okamura K, Taniguchi K (2006) Preparation of carbon nanotube-alginate nanocomposite gel for tissue engineering. Dent Mater J 25:719–725
Jiang ZY, Xu SW, Lu Y, Yuan WK, Wu H, Lv C (2006) Nanotube-doped alginate gel as a novel carrier for BSA immobilization. J Biomater Sci Polym Ed 17:21–35
Thompson BC, Moulton SE, Gilmore KJ, Higgins MJ, Whitten PG, Wallace GG (2009) Carbon nanotube biogels. Carbon 47:1282–1291
Lyman C, Moulton SE, Wallace GG (2007) Carbon nanotube biofibers. Adv Mater 19:1244–1248
Kumar S (2006) Self-organization of disc-like molecules: chemical aspects. Chem Soc Rev 35:83–109
Barnaby SN, Yu SM, Tsiola A, Fath KR, Banerjee IA (2011) pH dependent spontaneous growth of ellagic acid assemblies for targeting Hela cells. J Nanosci Nanotechnol. In Press
Pace CN, Grimsley GR, Scholtz JM (2009) Protein ionizable groups pK values and their contribution to protein stability and solubility. J Biol Chem 284:13285–13289
Grimsley GR, Scholtz JM, Pace CN (2009) A summary of the measured pk values of ionizable groups in folded proteins. Protein Sci 18:247–251
Hasegawa M, Terauchi M, Kikuchi Y, Nakao A, Okubo J, Yoshinaga T, Hiratsuka H, Kobayashi M, Hoshi T (2003) Deprotonation processes of ellagic acid in solution and solid states. Monatsh Chem 134:811–821
Mignon P, Loverix S, Geerlings P (2005) Interplay between Π-Π interactions and the H-bonding ability of aromatic nitrogen bases. Chem Phys Lett 401:40–46
Churchill CD, Wetmore SD (2009) Noncovalent interactions involving histidine: the effect of charge on pi-pi stacking and T-shaped interactions with the DNA nucleobases. J Phys Chem B 113:16046–16058
De S, Drew MGB, Aliaga-Alcalde N, Datta D (2009) Imidazole-imidazole stacking in some inorganic complexes. Inorg Chim Acta 362:2879–2883
Yang SP, Chem XM, Ji LN (2000) Influence of the counter ions and ligands on structure of silver (I) helicates with di-Schiff bases containing imidazole groups. J Chem Soc Dalton Trans 2337–2344
Kim EJ, Shim G, Kim K, Kwon IC, Oh YK, Shim CK (2009) Hyaluronic acid complexed to biodegradable poly-L-arginine for targeted delivery of siRNAs. J Gene Med 11:791–803
Malay Ö, Yalçun D, Batigün A, Bayraktar O (2008) Characterization of silk fibroin/hyaluronic acid polyelectrolyte complex (PEC) films. J Therm Anal Calorim 94:749–755
Malay Ö, Bayraktar O, Batigun A (2007) Complex coacervation of silk fibroin and hyaluronic acid. Int J Biol Macromol 40:387–393
Suh KY, Yang JM, Khademhosseini A, Berry D, Tran TNT, Park H, Langer R (2005) Characterization of chemisorbed hyaluronic acid directly immobilized on solid substrates. J Biomed Mater Res B 72B:292–298
Kopecek J, Yang J (2009) Peptide directed self-assembly of hydrogels. Acta Biomater 5:805–816
Kumar S, Rai AK, Rai SB, Rai DK (2010) Infrared and Raman spectra of histidine: an ab initio DFT calculations of histidine molecule and its protonated forms. Indian J Physiol 84:563–573
Haswega K, Ono TA, Noguchi T (2002) Ab Initio density functional theory calculations and vibrational analysis of zinc-bound 4-methylimidazole as a model of a histidine ligand in metalloenzymes. J Phys Chem A 106:3377–3390
Manna U, Bharan S, Patil S (2009) Layer-by-layer self-assembly of modified hyaluronic acid/chitosan based on hydrogen bonding. Biomacromolecules 10:2632–2639
Wu CS, Liao HT (2005) A new biodegradable blends prepared from polylactide and hyaluronic acid. Polymer 46:10017–10026
Parker FS (1983) Application of infrared, Raman and resonance Raman spectroscopy. In: Biochemistry. Plenum, New York
Calderon L, Collin E, Velasco-Bayon D, Murphy M, O'Halloran D, Pandit A (2010) Type II collagen-hyaluronan hydrogel—a step towards a scaffold for intervertebral disc tissue engineering. Eur Cells Mater 20:134–148
Burke SE, Barrett CJ (2003) pH responsive properties of multilayered poly-L-lysine/hyaluronic acid surfaces. Biomacromolecules 4:1773–1783
Karlson L, Joabsson F, Thuresson K (2000) Phase behavior and rheology in water and in model paint formulations thickened with HM-EHEC: influence of the chemical structure and the distribution of hydrophobic tails. Carbohydr Polym 41:25–35
Surini S, Akiyama H, Morishita M, Nagai T, Takayama K (2003) Release phenomena of insulin from an implantable device composed of a polyion complex of chitosan and sodium hyaluronte. J Control Release 90:291–301
Yariv S, Müller-Vonmos M, Kahr G, Rub A (1989) Thermal analytic study of the adsorption of crystal violet by montmoillonite. Thermochim Acta 148:457–466
Bhasikuttan AC, Shastri LV, Sapre AV (2001) Interaction of triplet state of crystal violet with fullerene C60. J Photochem Photobiol A 143:17–21
Parak WJ, Domke J, George M, Karinal A, Radmacher M, Gaub HE, de Roos AD, Theuvenet AP, Wiegand G, Sackmann E, Behrends JC (1999) Electrically excitable normal rat kidney fibroblasts: a new model system for cell-semiconductor hybrids. Biophys J 76:1659–1667
Acknowledgments
This work was conducted, in part, using equipment from the Queens College Facilities for Imaging, Cell and Molecular Biology. The authors thank Dr. Areti Tsiola for the use of the NanoDrop Spectrometer, as well as Dr. Patrick Brock and Dr. Barbara Balestra from the Geology Department at Queens College for the use of the scanning electron microscope. These studies were funded, in part, by the Fordham Dean’s Office (S.B., N.N., and S.F.), Fordham University Faculty research grant (I.B.), and grants from the PSC-CUNY Research Award Program (K.F.). S.B. also thanks the Campion Institute/Office of Prestigious Fellowships at Fordham University for their support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barnaby, S.N., Nakatsuka, N., Frayne, S.H. et al. Formation of hyaluronic acid–ellagic acid microfiber hybrid hydrogels and their applications. Colloid Polym Sci 291, 515–525 (2013). https://doi.org/10.1007/s00396-012-2739-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-012-2739-2