Abstract
In poultry slaughtering, cross-contamination with Salmonella Enteritidis is a constant ongoing challenge. Interaction between food contact surfaces can potentially transfer pathogenic material like feces from carcasses to another one. One approach to break this chain is to modify surfaces that frequently come into contact with the animal during the slaughtering process. Surface alterations like nanoscale coatings have already been successfully applied in various fields to lower the bacterial load. The aim of the study was to compare bacterial attachment, proliferation and detachment of Salmonella Enteritidis and Escherichia coli on uncoated and on nanoscale silica coated rubber picker fingers at laboratory scale. It was shown that both target organisms did not adhere less to coated surface than to uncoated picker fingers, whereas no difference in bacterial growth or detachment was detected. It can be concluded that the coating used in this study did not contribute to a reduction of the bacterial load on this surface in the specific experimental setups employed. Further studies should focus on whether nanoscale surface modifications achieve improved results under more practical conditions and whether other factors such as surface durability can be influenced by a coating.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Salmonella (S.) enterica cause the second most common bacterial enteritidis in humans with approximately 60,000 cases in 2021 in Europe [1]. Due to the past control programs in Europe, the number of infections caused by S. enterica in primary production of poultry and pigs could be reduced by accompanying steps such as logistical slaughtering. This have an effect on cross-contamination by decreasing the bacterial load at the various stations of the process, but it appears that these measures are not yet sufficient [2]. Even carcasses with a negative Salmonella status can be contaminated with Salmonella during slaughter due to soiled equipment [3]. Despite this, cases of salmonellosis are still on a high level, most recently with about 9000 cases in 2022, in Germany [4]. Salmonella Infantis is the most common serovar recovered in the slaughtering process environment, and in terms of zoonotic potential, serovars Typhimurium and Enteritidis appear to be the most clinically important ones [5, 6].
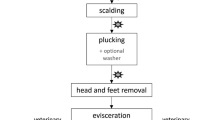
One of the main causes for salmonellosis is still the consumption of improper heat treatment contaminated food, besides of eggs or egg products, in particular pork and poultry meat [7]. Slaughtering process risk such as carcass contamination and cross-contamination are in the focus [8], where potentially pathogenic organic material is passed from carcass to carcass via equipment such as conveyer belts, shackles or other food contact surfaces [9, 10]. This occurs particularly during poultry defeathering, as large amounts of organic material accumulate in this highly mechanized process, and the exchange rate between carcasses is correspondingly high [11]. Pacholewicz et al. [12] showed that the defeathering step should still be given special attention with respect to microbial cross-contamination. In a study, in which swabs and water samples were collected from the slaughter line before slaughter, it was found that the area of plucking had the highest Salmonella prevalence of 10.4% (for the defeathering machine: 17.0%) [6]. Additionally, in the subsequent highly mechanized evisceration, feces can leak from the organs due to the removal of the intestinal convulsions and contaminate subsequent carcasses or the machines themselves [13, 14].
It is already known that different Salmonella serovars such as Sofia, Typhimurium, Virchow and Infantis have the ability to adhere to different substrates commonly used in the slaughtering process, for example rubber and stainless steel [15]. Most serovars are also able to form biofilms on stainless steel depending on prevalent conditions [16]. Therefore, it is of interest to influence the interaction between surface and carcass in a way that adapting the surface and makes it unfavorable to bacteria. In the slaughtering process itself, various materials with an increased risk of cross-contamination have already been identified, such as generally stainless-steel surfaces, conveyor belts, picker fingers [10] and cutting boards [17]
On natural surfaces like plants or insects, topographical structures in nanometer range could be detected, which protect the corresponding living being against environmental influences [18, 19]. Modification of contact surfaces by direct material changes or coatings has long been known in other fields such as in human medicine like implants in dentistry [20] or active packaging with nanocoating in food industry [21, 22]. There are various considerations about the exact functional principle of these modified surfaces. Many factors play a role in the bacteria-surface interaction under consideration: first, the surface itself with its properties such as chemical composition, surface tension, surface free energy, as well as topographic changes such as roughness and the associated wettability [23, 24]. The bacterium or bacteria [18] and the environment affecting the interaction must also be taken into account [24], which is why the effect of a coating can vary in each case. The surface modifications change the wettability of the surface to hydrophobic, so that the self-cleaning effect of the coating can occur [25, 26].Other studies report a rupturing of the bacterial cell wall and thus a bactericidal effect of such surface modifications [27, 28]. The exact principle of such complex surface modifications and the interaction of the different parameters of surface, bacterium and environment have not been completely understood and explained until today [23].
In past studies, different materials were directly modified or coated. Nanostructured gold surfaces, for example, were shown to be bactericidal against Staphylococcus (St.) aureus showing cell deformation and cell rupture after two hours of incubation [29]. The textured surface of other metals such as aluminum allows Escherichia (E.) coli and Listeria (L.) innocua to attach better when they have a nanoporous topography compared to nanosmooth surfaces [30]. It was also discovered that different nanometer scale topographies of TiO2 grown on titanium, commonly used in human medicine, affected the attachment of bacteria such as St. epidermidis by killing 99% of bacteria during two hours of contact time with nanostructured surface [27]. PMMA (poly(methyl methacrylate)) as an example of plastics or synthetic materials is capable of forming nanopillar structures, which was found to result in higher dead fraction of adherent E. coli (16–141%) and thus reduced viability compared to flat controls [31]. However, also coating of surfaces seems promising, which is realized in some cases by the sol–gel technology, which allows to coat the surface by dipping or spraying [32]. Furthermore, coating of diamond-like carbon nanocomposites with embedded silver nanoparticles was the most effective in terms of reducing L. monocytogenes and Campylobacter jejuni compared to uncoated silicon, stainless steel and HDPE (high density polyethylene) [33]. Titanium discs coated with a thin tantalum film led to reduction in adhesion and growth of Streptococcus mutans and Porphyromonas gingivalis [20].
The application of nanoscale coating on surfaces exposed in the scalding room and defeathering area like picker fingers could influence the bacterial behavior and decreases the prevalence of Salmonella [6]. It has already been shown that nanoscale silica coating of stainless steel tends to reduce the attachment in Salmonella Enteritidis and E. coli, and with some tendency to increase detachment of E. coli from coated stainless steel [34]. In addition, a study revealed decreased adhesion for Pseudomonas aeruginosa (57%) and St. aureus (20%) on nanoscale gold coated polystyrene surfaces compared to microscale surfaces [35]. Ivanova et al. [36] showed that synthesized black silicon as a surface material exhibits fine needle-like structures, which is bactericidal to Gram-negative as well as Gram-positive bacteria. In another study, it was shown that the concentration of 4% of a coating with silica-titania nano core-shells showed the best antibacterial effect against E. coli and Bacillus [37]. Di Cerbo et al. [38] showed that nanotechnological coating of variously rough stainless steel may be due to the synergistic effect of reduced bacterial adhesion and increased hydrophobicity.
Therefore, the aim of this study is to investigate the bacterial attachment, growth and detachment of Salmonella Enteritidis and E. coli on nanoscale silica coated and uncoated picker fingers. E. coli is chosen as an indicator bacterium for fecal contamination of surfaces. The presence of the pathogen suggests a possible contamination of surfaces with Salmonella spp. [39]. When applied as a marker organism, E. coli shows a similar distribution pattern on poultry in defeathering machines as naturally occurring microorganisms on carcasses [40].
Materials and methods
Bacterial material
The bacterial strains employed, Salmonella isolate 19-SA00115 (S. E.) and E. coli isolate (20-AB00467) were obtained from the German Federal Institute for Risk Assessment and were both isolated from chicken meat samples. The cryopreserved isolates (− 80 °C, Cryobank, Mast Group Ltd., Germany) were grown on tryptone glucose yeast extract agar (Plate Count Agar; PC; TN1189; sifin diagnostics GmbH, Germany) and were used for experiments for a maximum of four weeks. Bacterial suspension including 0.3% bovine serum albumin (BSA; A6588; VWR International GmbH, Germany) concentration was prepared as described previously [34].
Surface preparation
Commercial picker fingers (20 mm bore diameter, material thickness: 17.5 mm front, 19 mm rear; total length: 97.5 mm, effective length: 90 mm; 7.5 mm protrusion to the outside (EAN: 04032966060909; Westfalia Werkzeug company GmbH & Co KG, Germany)) were used.
They were cleaned according to DIN EN 13697:2019–10 by setting them 60 min inside a 5% Decon90 (Decon Laboratories Ltd., United Kingdom) solution for decontamination. After rinsing with distilled water, they were immersed in 95% propanol (Carl Roth GmbH, Germany) solution for 15 min for sterilization and washed with distilled water, air dried in a biosafety cabinet, and subsequently autoclaved at 121 °C. Picker fingers were either stored in closed plastic bags at room temperature to be used directly for the experimental setup or were coated with nanoscale silicon dioxide. This coating was performed by Nanopool GmbH, Germany, which used its commercial product Liquid Glass Metal to apply a nanoscale layer of silicon dioxide to the picker fingers. New picker fingers were used for each experiment.
Study design
Bacterial attachment, growth and detachment of target organisms were investigated for picker fingers in regard to coating. One experiment with three uncoated and three coated picker fingers was carried out three times per approach.
Attachment
Two types of attachment were simulated at ambient temperature (25 ± 5 °C). 1) Picker fingers were immersed for one minute in 20 ml of bacterial suspension with an approximate concentration of 1.0 × 108 cfu/ml in a centrifuge tube (50 ml, TPP Techno Plastic Products AG, Switzerland) and briefly vortexed to allow complete wetting of the surface to simulate the spilling of potentially contaminated fluids in the slaughterhouse. Picker fingers were tapped three times per third on the rim of a glass dish to remove excess fluid. 2) The forces acting on the picker fingers during defeathering of the poultry were simulated with a silicon brush immersed in bacterial suspension of a concentration of 1.0 × 108 cfu/ml by passing it three times lengthwise over the third of the picker finger with constant pressure. After each third, the brush was immersed again and the process repeated until the picker finger was completely covered.
Proliferation
Growth of the target organisms on picker fingers were observed for eight hours at 30 °C. The eight-hour period was chosen to represent a work shift in slaughterhouses. Picker fingers were immersed for one minute in a gently vortexed 20 ml bacterial suspension with approx. 1.0 × 105 cfu/ml. Excess liquid was removed by tapping the picker fingers three times per third on the rim of a glass dish. All picker fingers were each placed individually upright in centrifuge tubes so that they almost completely sealed the tube and guaranteed a relative humidity of min. 70%. Tubes with picker fingers were placed upright into a polypropylene container (6.0 l; Sunware B.V., Netherland) with an attached but not closed lid. One such container comprising each three coated and three uncoated picker fingers was examined every hour.
Detachment
Detachment of bacteria was examined for water and detergent foam. Picker fingers were immersed in a 20 ml bacterial suspension (approx. 1.0 × 108 cfu/ml) for one minute. After removal of excess liquid by tapping picker fingers were immersed for three seconds in a centrifuge tube containing 30 ml of 79–82 °C distilled water. Afterwards, excess liquid was removed again by tapping the edge of a glass dish one time per third of picker finger.
A 0.5% detergent solution of a sodium hydroxide-based protein and fat solving detergent (Eiweiß-Fettlöser flüssig, Ernst GmbH & Co. KG, Germany) was prepared to test the detachment of bacteria with foam on the different surfaces. A volume of 7.5 ml was transferred into a centrifuge tube and shaken manually for 10 s to produce a stable foam. The picker fingers were immersed for one minute in 20 ml bacterial suspension (approx. 1.0 × 108 cfu/ml) in a centrifuge tube, then excess liquid was removed as described above and picker fingers were transferred to the centrifuge tube containing the freshly prepared foam. After five minutes exposure time, picker fingers were transferred for three seconds into a centrifuge tube filled with 30 ml of 79–82 °C distilled water finally freed from excess water by tapping each third of the picker finger on the rim of a glass dish.
Recovery and enumeration of bacteria
The bacteria were recovered following the described treatment, based on the method of Arnold et al. [41], as follows: the picker finger was cut off after the third rib from the terminal end with sterile scissors (see Fig. 1), transferred to centrifuge tube filled with 10 ml sodium chloride peptone solution and vortexed for 15 s at 2500 rpm (shaker RS-OS 5, Phoenix Instrument GmbH, Germany). Subsequently, decimal dilutions were spread on PC agar plates for enumeration and incubated at 37 °C for 24 h. Grown colonies were counted.
Data analysis
All bacterial counts were log10 transformed for statistical analysis. For every experimental day, the initial bacterial concentration was determined for further calculation of attached, multiplied, and detached bacteria because of slight day-by-day differences. The average values of three replicates were taken for bacterial attachment. From this, the mean differences between the numbers of bacteria attached and recovered were calculated from three picker fingers per replicate. Since the value represents the number of bacteria that were unable to attach to the surface the results are expressed as reduced attachment. Bacterial growth was calculated as the average of three replicates from the differences of three picker fingers at a given time minus the bacterial count at time point 0. The bacteria detached from the surfaces were calculated as the average of three replicates from the mean differences of the applied and recovered bacterial counts of three picker fingers per replicate according to the respective cleaning method. Differences between uncoated and coated discs per treatment (attachment/detachment) or per time point (growth) were statistically analyzed by unpaired t test at an alpha level of significance of 0.05. Data were mainly distributed normally as revealed by Shapiro–Wilk test. All statistical analyses were executed by Prism 9 (GraphPad Software, LLC, USA).
Results
Attachment
The immersion of picker fingers resulted in mean reduced attachment of 1.047 log10 cfu on uncoated and 0.963 log10 cfu on coated surface for S. E. (P = 0.130). For E. coli, a similar slight but statistically significant difference was detected (P = 0.047) with an average of 1.172 log10 cfu on uncoated and 1.029 log10 cfu on coated picker fingers (see Fig. 2A).
When the picker fingers were brushed with S. E. suspension, a similar number of bacteria were unable to attach on uncoated (0.979 log10 cfu) and coated (0.988 log10 cfu) picker fingers (P = 0.961). The same effect was observed for E. coli (P = 0.562) with an average of 0.892 log10 cfu unattached bacteria on uncoated picker fingers compared to 0.871 log10 cfu on coated ones (see Fig. 2B).
Proliferation
Similar growth was observed for both target organisms irrespective of surface modification. Starting with a concentration of 1.0 × 105 cfu/ml, bacteria grew more than four log within the observed eight hours. Neither bacterial count of S. E. nor of E. coli differed at any hour regarding the coating to a statistically significant extent (see Fig. 3, Table 1).
Detachment
By cleaning the surfaces inoculated with S. E. using hot water, an average of 3.652 log10 cfu detached from uncoated picker fingers and a similar amount of 3.578 log10 cfu could be removed from the coated ones (P = 0.876). A similar result was observed for E. coli, with an average detachment of 3.893 log10 cfu from uncoated and 3.684 log10 cfu from coated picker fingers (P = 0.620) (Fig. 4A).
Using detergent foam, 2.904 log10 cfu S. E. were removed from uncoated and 3.046 log10 cfu from coated picker fingers (P = 0.260). Comparable results were also observed for E. coli: an average of 2.970 log10 cfu detached from the uncoated surface and 2.635 log10 cfu from the coated surface (P = 0.272) (Fig. 4B).
Discussion
Attachment
In this study, S. E. and E. coli tend to attach less to uncoated surfaces of rubber picker fingers than to silicon dioxide coated ones. It was found that this slight difference was statistically significant for E. coli after simulated contamination by immersion. This was unexpected from previous results for stainless steel [34] but may be explained by the characteristic of the rubber picker finger material itself. Arnold et al. [42] reported that compared to other material like stainless steel, rubber picker fingers exhibited a reduced bacterial adherence when they were brand-new and superficially unchanged. Due to the accompanying surface’ structure alteration induced by the applied silicon dioxide, the coating might reduce this immanent material effect and, thus, lead to the observed results. However, not only the picker finger itself, but also the defeathering in the plucking machine can have an influence on bacterial attachment. This was tried to be imitated in the present study by brushing. It should be noted that the distribution pattern of microorganisms during the plucking is influenced by the movement of the picker fingers and thus depends on the type of machine, which has implications for the extent of cross-contamination of the defeathering process. Allen et al. [40] found that on poultry inoculated with E. coli, fewer marker organisms were found on carcasses defeathered by a contrarotating machine (mean log10 5.23 ± 0.09 cfu/ml carcass rinse for single pass) than on the animals plucked by the disc machine (mean log10 2.43 ± 0.41 cfu/ml carcass rinse). In the present study, this aspect was neglected because only single picker fingers were used in the experiments. No difference was found for S. E. and only a tendency for increased attachment on uncoated picker fingers for E. coli which in turn also indicates good general resistance of the rubber picker fingers against bacterial attachment.
Proliferation
Growth of different Salmonella serovars on various food contact surfaces such as fiberglass reinforced plastic wall paneling, stainless steel, and acetal resin could be observed for several days [43]. Consequently, it was assumed that growth would be possible on both surfaces during the estimated period.
A reason for the observed similar growth of the two target organisms on uncoated and coated picker finger could be the bacterial cell extensions, which explore the surface microstructures like appendages and can thus attach better and multiply accordingly. Friedlander et al. [44] discovered that on polydimethylsiloxane surfaces with certain topographical surface the adhesion of E. coli is significantly reduced in the first two hours compared to one of the not structured, flat controls. This behavior abruptly reverses to a significantly increased adhesion at longer exposure. That can be justified on the basis of the trapped air within the nanostructured surface, which are displaced after a period, four hours, by the bacteria-containing medium allowing the bacteria to also penetrate into the crevices with the cell appendages and thus have the opportunity to form a network. The solid–liquid-air state, which is described by Cassie-Baxter [45], as on porous, heterogeneous surfaces resulting in the hydrophobicity of the surface, could have been disturbed by bacterial suspension right at the beginning in the experimental setup presented. This would have allowed the bacteria to adhere to the pits of the nanostructured surface through the appendages. This could explain why there is no difference in growth between the surfaces in both bacteria in the present study.
Detachment
Hardly any difference was found between uncoated and coated picker fingers for S. E. and E. coli by washing the inoculated picker fingers with water or foam to observe their detachment from the surface. Rather, it seems that both isolates tend to detach better with water and also with foam from uncoated picker fingers than from coated ones.
As already mentioned, the organic material produced during defeathering plays a role in cross-contamination. Organic soiling was imitated in the present study by adding BSA to the bacterial suspension. Based on DIN EN 13697 the added BSA can be considered as a high protein concentration, which has also an influence on the interaction between bacteria and surface [46]. Singh et al. [47] described two effects on nanostructured surfaces like titania films. On the one hand, there is the passivation effect, which states that the accumulation of proteins reduces bacterial adhesion and biofilm formation on nanostructured surfaces by forming a thick layer that reduces the interaction of bacteria with the nanostructured surface and inhibits bacterial adhesion. On the other hand, the surface can be significantly flattened by the protein layer, which can suppress possible effects of nanoscale morphology on bacterial adhesion and detachment. Both effects may contribute to a similar surface structure, i.e. a protein layer on which bacteria will attach finally. Therefore, detachment would rather be from the protein layer than from the material surface itself and will, thus, result in the observed similar detachment rates. Supplementary, not only proteins themselves, but also organic material, as it is massively produced also in the mechanically operated plucking plants, influences the bacteria-surface interaction [11].
The laboratory scale experiments mimicked conditions and potential contamination routes at slaughterhouses to test the effectiveness of the coating in context of this environment. Two types of contamination routes that take place in real slaughtering processes and, therefore, impact the bacterial numbers in general were neglected in our study: the direct contact of carcasses with each other and the contamination of carcasses by other factors such as air, which seems to play a role for a contaminated following carcass and also for the surrounding environment of the respective defeathering machines and also the preceding carcasses [40, 48].
Conclusion
In the present study, the response of Salmonella Enteritidis and Escherichia coli was comparatively investigated on uncoated and nanoscale coated rubber picker fingers in different tests mimicking the slaughterhouse environment.
The applied coating was only able to affect the bacterial load marginally and should be improved to gain a higher impact. Future studies should aim to ensure that the modification of food contact surfaces is precisely matched to the substrate, application area and its environment, as surfaces are subject to considerable stress and extreme conditions. In a practical environment, where several bacterial species get onto the surface during the slaughter process, the potential for biofilm formation is high, which can be challenging for a structurally identical surface modification. The limitation of the coating can also be wear and abrasion. Substantial loss can lead to a reduction of the effect. A surface that has multiple levels of roughness or several structures at the micro- and nanoscale could be challenging for various bacterial species and end up reducing cross-contamination.
Data availability and materials
All data included in this manuscript are available upon request by contacting with the corresponding authors.
References
European Food Safety Authority (2022) The European Union One Health 2021 Zoonoses Report 20:1-273. https://doi.org/10.2903/j.efsa.2022.7666
Berghaus RD, Thayer SG, Law BF et al (2013) Enumeration of Salmonella and Campylobacter spp. in environmental farm samples and processing plant carcass rinses from commercial broiler chicken flocks. Appl Environ Microbiol 79:4106–4114. https://doi.org/10.1128/AEM.00836-13
Olsen JE, Brown DJ, Madsen M et al (2003) Cross-contamination with Salmonella on a broiler slaughterhouse line demonstrated by use of epidemiological markers. J Appl Microbiol 94:826–835. https://doi.org/10.1046/j.1365-2672.2003.01911.x
Robert Koch-Institut (2023) Epidemiologisches Bulletin 1/2023. https://www.rki.de/DE/Content/Infekt/EpidBull/Archiv/2023/Ausgaben/01_23.pdf?__blob=publicationFile
Chanamé Pinedo L, Mughini-Gras L, Franz E et al (2022) Sources and trends of human salmonellosis in Europe, 2015–2019: An analysis of outbreak data. Int J Food Microbiol 379:109850. https://doi.org/10.1016/j.ijfoodmicro.2022.109850
Zeng H, de Reu K, Gabriël S et al (2021) Salmonella prevalence and persistence in industrialized poultry slaughterhouses. Poult Sci 100:100991. https://doi.org/10.1016/j.psj.2021.01.014
World Health Organization (2020) Salmonella (non-typhoidal). https://www.who.int/news-room/fact-sheets/detail/salmonella-(non-typhoidal). Accessed 01 Jul 2020
QS Qualität und Sicherheit GmbH (2020) Leitfaden Salmonellenmonitoring und -reduzierungsprogramm für die Geflügelfleischerzeugung. https://www.q-s.de/services/files/downloadcenter/h-salmonellenmonitoring/2023/leitfaeden/deutsch/Leitfaden_Salmonellenmonitoring_Gefl%C3%BCgel_01.01.2023.pdf
Shackelford AD (1988) Modifications of processing methods to control Salmonella in poultry. Poult Sci 67:933–935. https://doi.org/10.3382/ps.0670933
Rasschaert G, Houf K, de Zutter L (2007) Impact of the slaughter line contamination on the presence of Salmonella on broiler carcasses. J Appl Microbiol 103:333–341. https://doi.org/10.1111/j.1365-2672.2006.03248.x
Rouger A, Tresse O, Zagorec M (2017) Bacterial contaminants of poultry meat: sources, species, and dynamics. Microorganisms 5:50. https://doi.org/10.3390/microorganisms5030050
Pacholewicz E, Swart A, Wagenaar JA et al (2016) Explanatory variables associated with Campylobacter and Escherichia coli concentrations on broiler chicken carcasses during processing in two slaughterhouses. J Food Prot 79:2038–2047. https://doi.org/10.4315/0362-028X.JFP-16-064
Peyrat MB, Soumet C, Maris P et al (2008) Phenotypes and genotypes of campylobacter strains isolated after cleaning and disinfection in poultry slaughterhouses. Vet Microbiol 128:313–326. https://doi.org/10.1016/j.vetmic.2007.10.021
Berrang ME, Buhr RJ, Cason JA et al (2001) Broiler carcass contamination with Campylobacter from feces during defeathering. J Food Prot 64:2063–2066. https://doi.org/10.4315/0362-028x-64.12.2063
Chia TWR, Goulter RM, McMeekin T et al (2009) Attachment of different Salmonella serovars to materials commonly used in a poultry processing plant. Food Microbiol 26:853–859. https://doi.org/10.1016/j.fm.2009.05.012
Wang H, Ding S, Dong Y et al (2013) Biofilm formation of Salmonella serotypes in simulated meat processing environments and its relationship to cell characteristics. J Food Prot 76:1784–1789. https://doi.org/10.4315/0362-028X.JFP-13-093
Dantas STA, Rossi BF, Bonsaglia ECR et al (2018) Cross-contamination and biofilm formation by Salmonella enterica serovar enteritidis on various cutting boards. Foodborne Pathog Dis 15:81–85. https://doi.org/10.1089/fpd.2017.2341
Tripathy A, Sen P, Su B et al (2017) Natural and bioinspired nanostructured bactericidal surfaces. Adv Colloid Interface Sci 248:85–104. https://doi.org/10.1016/j.cis.2017.07.030
Ivanova EP, Hasan J, Webb HK et al (2012) Natural bactericidal surfaces: mechanical rupture of Pseudomonas aeruginosa cells by Cicada wings. Small 8:2489–2494. https://doi.org/10.1002/smll.201200528
Zhu Y, Gu Y, Qiao S et al (2017) Bacterial and mammalian cells adhesion to tantalum-decorated micro-/nano-structured titanium. J Biomed Mater Res A 105:871–878. https://doi.org/10.1002/jbm.a.35953
Ahmed J, Arfat YA, Bher A et al (2018) Active chicken meat packaging based on polylactide films and bimetallic Ag–Cu nanoparticles and essential oil. J Food Sci 83:1299–1310. https://doi.org/10.1111/1750-3841.14121
Gallocchio F, Cibin V, Biancotto G et al (2016) Testing nano-silver food packaging to evaluate silver migration and food spoilage bacteria on chicken meat. Food Addit Contam Part A Chem Anal Control Expo Risk Asses 33:1063–1071. https://doi.org/10.1080/19440049.2016.1179794
Bazaka K, Crawford RJ, Ivanova EP (2011) Do bacteria differentiate between degrees of nanoscale surface roughness? Biotechnol J 6:1103–1114. https://doi.org/10.1002/biot.201100027
van Houdt R, Michiels CW (2010) Biofilm formation and the food industry, a focus on the bacterial outer surface. J Appl Microbiol 109:1117–1131. https://doi.org/10.1111/j.1365-2672.2010.04756.x
Wenzel RN (1936) Resistance of solid surfaces to wetting by water. Ind Eng Chem 28:988–994. https://doi.org/10.1021/ie50320a024
Barthlott W, Neinhuis C (1997) Purity of the sacred lotus, or escape from contamination in biological surfaces. Planta 202:1–8. https://doi.org/10.1007/s004250050096
Cao Y, Su B, Chinnaraj S et al (2018) Nanostructured titanium surfaces exhibit recalcitrance towards Staphylococcus epidermidis biofilm formation. Sci Rep 8:1071. https://doi.org/10.1038/s41598-018-19484-x
Elbourne A, Chapman J, Gelmi A et al (2019) Bacterial-nanostructure interactions: the role of cell elasticity and adhesion forces. J Colloid Interface Sci 546:192–210
Songmei Wu, Zuber F, Brugger J et al (2016) Antibacterial Au nanostructured surfaces. Nanoscale 8:2620–2625. https://doi.org/10.1039/C5NR06157A
Feng G, Cheng Y, Wang S-Y et al (2014) Alumina surfaces with nanoscale topography reduce attachment and biofilm formation by Escherichia coli and Listeria spp. Biofouling 30:1253–1268. https://doi.org/10.1080/08927014.2014.976561
Dickson MN, Liang EI, Rodriguez LA et al (2015) Nanopatterned polymer surfaces with bactericidal properties. Biointerphases. https://doi.org/10.1116/14922157
Aegerter MA, Almeida R, Soutar A et al (2008) Coatings made by sol–gel and chemical nanotechnology. J Sol-Gel Sci Technol 47:203–236. https://doi.org/10.1007/s10971-008-1761-9
Zakarienė G, Novoslavskij A, Meškinis Š et al (2018) Diamond like carbon Ag nanocomposites as a control measure against Campylobacter jejuni and Listeria monocytogenes on food preparation surfaces. Diam Relat Mater 81:118–126. https://doi.org/10.1016/j.diamond.2017.12.007
Schumann-Muck FM, Hillig N, Braun PG et al (2023) Impact of nanoscale coating of stainless steel on Salmonella Enteritidis and Escherichia coli. J Food Safety. https://doi.org/10.1111/jfs.13075
Nguyen DHK, Pham VTH, Truong VK et al (2018) Role of topological scale in the differential fouling of Pseudomonas aeruginosa and Staphylococcus aureus bacterial cells on wrinkled gold-coated polystyrene surfaces. Nanoscale 10:5089–5096. https://doi.org/10.1039/c7nr08178b
Ivanova EP, Hasan J, Webb HK et al (2013) Bactericidal activity of black silicon. Nat Commun. https://doi.org/10.1038/ncomms3838
Verma J, Khanna AS, Sahney R et al (2020) Super protective anti-bacterial coating development with silica-titania nano core-shells. Nanoscale Adv 2:4093–4105. https://doi.org/10.1039/d0na00387e
Di Cerbo A, Mescola A, Rosace G et al (2021) Antibacterial effect of stainless steel surfaces treated with a nanotechnological coating approved for food contact. Microorganisms. https://doi.org/10.3390/microorganisms9020248
Foley SL, Lynne AM, Nayak R (2008) Salmonella challenges: prevalence in swine and poultry and potential pathogenicity of such isolates. J Anim Sci 86:E149–E162. https://doi.org/10.2527/jas.2007-0464
Allen VM, Tinker DB, Hinton MH et al (2003) Dispersal of micro-organisms in commercial defeathering systems. Br Poult Sci 44:53–59. https://doi.org/10.1080/0007166031000085436
Arnold JW (2007) Bacterial contamination on rubber picker fingers before, during, and after processing. Poult Sci 86:2671–2675. https://doi.org/10.3382/ps.2007-00187
Arnold JW, Silvers S (2000) Comparison of poultry processing equipment surfaces for susceptibility to bacterial attachment and biofilm formation. Poult Sci 79:1215–1221. https://doi.org/10.1093/ps/79.8.1215
Allan JT, Yan Z, Kornacki JL (2004) Surface material, temperature, and soil effects on the survival of selected foodborne pathogens in the presence of condensate. J Food Prot 67:2666–2670. https://doi.org/10.4315/0362-028X-67.12.2666
Friedlander RS, Vlamakis H, Kim P et al (2013) Bacterial flagella explore microscale hummocks and hollows to increase adhesion. Proc Natl Acad Sci USA 110:5624–5629. https://doi.org/10.1073/pnas.1219662110
Cassie ABD, Baxter S (1944) Wettability of porous surfaces. Trans Faraday Soc 40:546. https://doi.org/10.1039/TF9444000546
Campoccia D, Montanaro L, Agheli H et al (2006) Study of Staphylococcus Aureus adhesion on a novel nanostructured surface by chemiluminometry. Int J Artif Organs 29:622–629. https://doi.org/10.1177/039139880602900612
Singh AV, Vyas V, Patil R et al (2011) Quantitative characterization of the influence of the nanoscale morphology of nanostructured surfaces on bacterial adhesion and biofilm formation. PLoS ONE 6:e25029. https://doi.org/10.1371/journal.pone.0025029
Allen VM, Hinton MH, Tinker DB et al (2003) Microbial cross-contamination by airborne dispersion and contagion during defeathering of poultry. Br Poult Sci 44:567–576. https://doi.org/10.1080/00071660310001616183
Acknowledgements
The study was supported by funds of the Federal Ministry of Food and Agriculture (BMEL) based on a decision of the Parliament of the Federal Republic of Germany via the Federal Office for Agriculture and Food (BLE) under the innovation support programme (grant number: 281C104D18). Open Access funding enabled and organized by Projekt DEAL.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Compliance with ethics requirements
This article does not contain any studies with human or animal subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schumann-Muck, F.M., Blaeske, V., Braun, P.G. et al. Effectiveness of nanoscale silicon dioxide-coated picker fingers on Salmonella Enteritidis and Escherichia coli. Eur Food Res Technol 250, 203–211 (2024). https://doi.org/10.1007/s00217-023-04378-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-023-04378-8