Abstract
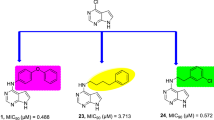
Rimonabant and its analogues have been synthesized in moderate to good yields using a simple synthetic route. All the newly synthesized compounds were subjected to in vitro screening against M. tuberculosis and M. smegmatis. The most potent analogue JMG-14 exhibits MIC value of 3.13 compared to 3.25 and 50 µg/ml for ethambutol and pyrazinamide, respectively. The molecular docking reveals that pyrazole ring, number and position of halogen atoms play a crucial role in deciding interactions with MTCYP-121. These findings open up a new avenue in the search of potent anti-TB drugs with rimonabant and its novel analogue JMG-14 as lead molecules.
Graphical Abstract









Similar content being viewed by others
References
Abreu RM, Froufe HJ, Queiroz MJ, Ferreira IC (2012) Selective flexibility of side-chain residues improves VEGFR-2 docking score using AutoDock Vina. Chem Biol Drug Des 79(4):530–534
Belin P, Le Du MH, Fielding A, Lequin O, Jacquet M, Charbonnier JB, Lecoq A, Thai R, Courcon M, Masson C, Dugavet R, Pernodet JL, Gondry M (2009) Identification and structural basis of the reaction catalyzed by CYP121, an essential cytochrome P450 in Mycobacterium tuberculosis. Proc Natl Acad Sci USA 106(18):7426–7431
Chiang CY, Schaaf HS (2010) Management of drug-resistant tuberculosis. Int J Tuberc LungDis 14:672–682
Corbett EL, Watt CJ, Walker N, Maher D, Williams BG, Raviglione MC, Dye C (2003) The growing burden of tuberculosis: global trends and interactions with the HIV epidemic. Arch Intern Med 163(9):1009–1021
Dumas VG, Defelipe LA, Petruk AA, Turjanski AG, Marti MA (2013) QM/MM study of the C–C coupling reaction mechanism of CYP121, an essential cytochrome p450 of mycobacterium tuberculosis. Proteins 82(6):1004–1021
Elzinga G, Raviglione MC, Maher D (2004) Scale up: meeting targets in global tuberculosis control. Lancet 363(9411):814–819
Handoko SD, Ouyang X, Su CT, Kwoh CK, Ong YS (2012) QuickVina: accelerating AutoDock Vina using gradient-based heuristics for global optimization. IEEE/ACM Trans Comput Biol Bioinform. 9(5):1266–1272
Hudson SA, McLean KJ, Surade S, Yang Y-Q, Leys D, Ciulli A, Munro AW, Abell C (2012) Application of fragment screening and merging to the discovery of inhibitors of the mycobacterium tuberculosis cytochrome P450 CYP121. Angew Chem Int Ed 51(37):9311–9316
Jagerovic N, Fernandez-Fernandez C, Goya P (2008) CB1 cannabinoid antagonists: structure-activity relationships and potential therapeutic applications. Curr Top Med Chem 8(3):205–230
Kang JG, Park CY (2012) Anti-obesity drugs: a review about their effects and safety. Diabetes Metab J 36(1):13–25
Kotagiri VK, Suthrapu S, Mukunda Reddy J, Prasad Rao C, Bollugoddu V, Bhattacharya A, Bandichor R (2007) An improved synthesis of rimonabant: anti-obesity drug. Org Proc Res Dev 11:910–912
Kumar V, Kaur K, Gupta GK, Sharma AK (2013) Pyrazole containing natural products: synthetic preview and biological significance. Eur J Med Chem 69:735–753
Lan R, Liu Q, Fan P, Lin S, Fernando SR, McCallion D, Pertwee R, Makriyannis A (1999) Structure-activity relationships of pyrazole derivatives as cannabinoid receptor antagonists. J Med Chem 42(4):769–776
Lange JH, Kruse CG (2005) Keynote review: medicinal chemistry strategies to CB1 cannabinoid receptor antagonists. Drug Discov Today 10(10):693–702
Masand VH, Jawarkar RD, Mahajan DT, Hadda TB, Sheikh J, Patil KN (2012) QSAR and CoMFA studies of biphenyl analogs of the anti-tuberculosis drug (6S)-2-nitro-6-{[4-(trifluoromethoxy) benzyl]oxy}-6,7-dihydro-5H-imidazo[2,1-b][1,3]oxazine(PA-824). Med Chem Res 21:2624–2629
Masand VH, Mahajan DT, Hadda TB, Jawarkar RD, Chavan H, Bandgar BP, Chauhan H (2013) Molecular docking and quantitative structure–activity relationship (QSAR) analyses of indolylarylsulfones as HIV-1 non-nucleoside reverse transcriptase inhibitors. Med Chem Res 23(1):417–425
McLean KJ, Marshall KR, Richmond A, Hunter IS, Fowler K, Kieser T, Gurcha SS, Besra GS, Munro AW (2002) Azole antifungals are potent inhibitors of cytochrome P450 mono-oxygenases and bacterial growth in mycobacteria and streptomycetes. Microbiology 148:2937–2949
Menozzi G, Merello L, Fossa P, Schenone S, Ranise A, Mosti L, Bondavalli F, Loddo R, Murgioni C, Mascia V, La Colla P, Tamburini E (2004) Synthesis, antimicrobial activity and molecular modeling studies of halogenated 4-[1H-imidazol-1-yl(phenyl)methyl]-1,5-diphenyl-1H-pyrazoles. Bioorg Med Chem 12(20):5465–5483
Moore AV, Kirk SM, Callister SM, Mazurek GH, Schell RF (1999) Safe determination of susceptibility of Mycobacterium tuberculosis to antimycobacterial agents by flow cytometry. J. Clin Microbio 37(3):479–483
NCCLS-National Committee for Clinical Laboratory Standards (1995) Antimycobacterial susceptibility testing for Mycobacterium tuberculosis proposed standard M24-T. Villanova, PA
Nunn P, Williams B, Floyd K, Dye C, Elzinga G, Raviglione M (2005) Tuberculosis control in the era of HIV. Nature Rev Immunol 5(10):819–826
Shukla D, Mesfin YM, Hailemariam D, Biadglign S, Kibret KT (2014) Association between HIV/AIDS and multi-drug resistance tuberculosis: a systematic review and meta-analysis. PLoS ONE 9(1):e82235
Stigliani JL, Bernardes-Genisson V, Bernadou J, Pratviel G (2012) Cross-docking study on InhA inhibitors: a combination of Autodock Vina and PM6-DH2 simulations to retrieve bio-active conformations. Org Bio-Mol Chem 10(31):6341–6349
Sundaramurthi JC, Kumar S, Silambuchelvi K, Hanna LE (2011) Molecular docking of azole drugs and their analogs on CYP121 of mycobacterium tuberculosis. Bioinformation 7(3):130–133
Tiwari A, Saxena S, Pant AB, Srivastava P (2012) Protein-ligand interaction studies of retinol-binding protein 3 with herbal molecules using AutoDock for the management of Eales’ disease. J Ocul Biol Dis Infor 5(2):40–43
www.who.int/topics/tuberculosis/en/, Global Tuberculosis Report 2013, in, 2013
www.who.int/topics/tuberculosis/en/, Tuberculosis prevalence surveys: a handbook, in, 2011
Acknowledgments
JMG gratefully acknowledges CSIR-OSDD, CSIR-ORIGIN and DST-SERB, for the financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gajbhiye, J.M., More, N.A., Patil, M.D. et al. Discovery of Rimonabant and its potential analogues as anti-TB drug candidates. Med Chem Res 24, 2960–2971 (2015). https://doi.org/10.1007/s00044-015-1346-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-015-1346-4