Abstract
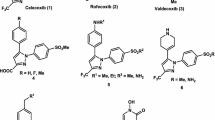
The ester derivatives of indomethacin were prepared by condensing indomethacin with an equimolar quantity of an appropriate alcoholic compound in anhydrous dichloromethane in the presence of DCC and DMAP. Spectral studies comprising of IR, 1H NMR, mass, and microanalysis were performed in order to confirm their structures. In vivo anti-inflammatory studies were carried out using carrageenan rat paw edema method and in vivo ulcerogenic studies by ulcer index method for the panel of synthesized compounds. Out of eleven compounds, the compound IIc displayed moderate anti-inflammatory activity with no observable ulcerogenic effect when compared to indomethacin. Furthermore, compound IIc, indomethacin and celecoxib were tested at a concentration of 20 μM against COX-1 and COX-2 enzymes by colorimetric COX inhibitor screening assay. Compound IIc was found to be active against COX-2 and COX-1 enzymes exhibiting 62.0 and 12.9% inhibition, respectively.

Similar content being viewed by others
References
Bayly CI, Black WC, Leger S, Quimet N, Quellet M, Percival MD (1999) Structure-based design of COX-2 selectivity into flurbiprofen. Bioorg Med Chem Lett 9:307–312
Bhandari KH, Newa M, Yoon SI, Kim JS, Jang KY, Kim JA, Yoo BK, Woo JS, Lee JH, Kim DD, Choi HG, Yong CS (2007) Evaluation of physicochemical properties, skin permeation and accumulation profiles of ketorolac fatty ester prodrugs. Biol Pharm Bull 30(11):2211–2216
Black WC, Bayly C, Belley M, Chan CC, Charleson S, Denis D, Gauthier JY, Gordon R, Guay D, Kargman S, Lau CK, Leblanc Y, Mancini J, Ouellet M, Percival D, Roy P, Skorey K, Tagari P, Vickers P, Wong E, Xu L, Prasit P (1996) From indomethacin to a selective COX-2 inhibitor: development of indolalkanoic acids as potent and selective cyclooxygenase-2 inhibitors. Bioorg Med Chem Lett 6(6):725–730
Kalgutkar AS, Marnett AB, Crews BC, Remmel RP, Marnett LJ (2000) Ester and amide derivatives of the nonsteroidal antiinflammatory drug, indomethacin, as selective cyclooxygenase-2 inhibitors. J Med Chem 43(15):2860–2870
Kalgutkar AS, Rowlinson SW, Crews BC, Marnett LJ (2002) Amide derivatives of meclofenamic acid as selective cyclooxygenase-2 inhibitors. Bioorg Med Chem Lett 12:521–524
Khanna S, Madan M, Vangoori A, Banerjee R, Thaimattam R, Jafar Sadik Basha SK, Ramesh M, Casturi SR, Pal M (2006) Evaluation of glycolamide esters of indomethacin as potential cyclooxygenase-2 (COX-2) inhibitors. Bioorg Med Chem 14(14):4820–4833
Laine L (2001) Approaches to nonsteroidal anti-inflammatory drug use in the high-risk patient. Gastroenterology 120:594–606
Leblanc Y, Black WC, Chan CC, Charleson S, Delorme D, Denis D, Gauthier JY, Grimm EL, Gordon R, Guay D, Hamel P, Kargman S, Lau CK, Mancini J, Ouellet M, Percival D, Roy P, Skorey K, Tagari P, Vickers P, Wong E, Xu Land Prasit P (1996) Synthesis and biological evaluation of both enantiomers of L-761, 000 as inhibitors of cyclooxygenase 1 and 2. Bioorg Med Chem Lett 6(6):731–736
Mishra A, Veerasamy R, Jain PK, Dixit VK, Agrawal RK (2008) Synthesis, characterization and pharmacological evaluation of amide prodrugs of ketorolac. Eur J Med Chem 43(11):2464–2472
Srivastava SK, Nath C, Gupta MB, Vrat S, Sinha NJ, Dhawan NK, Gupta GP (1991) Protection against gastric ulcer by verapamil. Pharmacol Res 23:81–86
Vane JR (1971) Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nature New Biology 231:232–235
Winter CA, Porter CC (1957) Effect of alterations in side chain. J Amer Pharm Ass Sci Ed 46:515–519
Wood KW, McCroskey RW, Michaelides MR, Wada CK, Hulkower KI, Bell L (2001) Thiazole analogues of the NSAID indomethacin as selective COX-2 inhibitors. Bioorg Med Chem 11:1325–1328
Acknowledgments
The author MAB is grateful to the Director, SGSITS, Indore and Director, CDRI, Lucknow for providing the necessary facilities for this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arockia Babu, M., Shukla, R., Nath, C. et al. Synthesis and biological evaluation of ester derivatives of indomethacin as selective COX-2 inhibitors. Med Chem Res 21, 2223–2228 (2012). https://doi.org/10.1007/s00044-011-9747-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-011-9747-5