Abstract
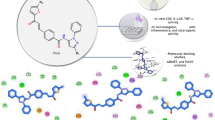
The present research was aimed at the synthesis and screening of 35 novel 2-(substituted phenyl imino)-5-benzylidene-4-thiazolidinones having different substitutions at imino phenyl and arylidene groups. The title compounds were synthesized by Knoevenagel condensation at the 5th position of the 4-thiazolidinone ring, in the presence of sodium acetate. The structures were assigned on the basis of spectral data. The compounds were screened for in vivo anti-inflammatory, antinociceptive and in vitro free-radical scavenging activities. The compounds exhibited significant activities when compared with standard drugs. The distinctive property of the derivatives was that none of them had an acidic group, like conventional NSAIDs, but exhibited significant in vivo activity in acute inflammation models. Further, the active compounds of each series were docked against cyclooxygeanase (COX)-2 enzyme using Glide module of Maestro 11.1 program. It was evident from the docking results that 3-chlorophenylimino and 2-chloro moiety on 5-benzylidene nucleus of the 4-thiazolidinone derivative (30) could easily fit into the COX-2-binding pocket, considered as critical interaction for COX-2 inhibition. Interestingly, some of the compounds exhibited the potential of becoming dual action or even triple action drug candidates, which could target degenerative disorders associated with excessive free-radical generation.





Similar content being viewed by others
References
Bhandari SV, Bothara KG, Patil AA, Chitre TS, Sarkate AP, Gore ST, Dangre SC, Khachane CV (2009) Design, synthesis and pharmacological screening of novel antihypertensive agents using hybrid approach. Bioorg Med Chem 17:390–400
Bhati SK, Kumar A (2008) Synthesis of new substituted azetidinoyl and thiazolidinoyl-1, 3, 4-thiadiazino (6, 5-b) indoles as promising anti-inflammatory agents. Eur J Med Chem 43:2323–2330
Bruno G, Costantino L, Curinga C, Maccari R, Monforte F, Nicolo F, Ottana R, Vigorita M (2002) Synthesis and aldose reductase inhibitory activity of 5-arylidene-2, 4-thiazolidinediones. Bioorg Med Chem 10:1077–1084
Chakraborti AK, Garg SK, Kumar R, Motiwala HF, Jadhavar PS (2010) Progress in COX-2 inhibitors: a journey so far. Curr Med Chem 17:1563–1593
Chawla P, Singh R, Saraf SK (2012a) Effect of chloro and fluoro groups on the antimicrobial activity of 2, 5-disubstituted 4-thiazolidinones: a comparative study. Med Chem Res 21:3263–3271
Chawla P, Singh R, Saraf SK (2012b) Syntheses and evaluation of 2, 5-disubstituted 4-thiazolidinone analogues as antimicrobial agents. Med Chem Res 21:2064–2071
Clarkson PM, Tremblay I (1988) Exercise-induced muscle damage, repair, and adaptation in humans. J Appl Physiol 65:1–6
Crofford LJ, Wilder RL, Ristimäki A, Sano H, Remmers EF, Epps HR, Hla T (1994) Cyclooxygenase-1 and-2 expression in rheumatoid synovial tissues. Effects of interleukin-1 beta, phorbol ester and corticosteroids. J Clin Investig 93:1095–1101
Dandiya P, Menon M (1963) Studies on central nervous system depressants.(III). Influence of some tranquillizing agents on morphine analgesia. Arch Int Pharmacodyn Ther 141:223
Davies O, Raventos J, Walpole A (1946) A method for the evaluation of analgesic activity using rats. Br J Pharmacol 1:255–264
Dixion W (1985) The up and down method or small samples. J Am Stat Assoc 60:967–978
Flohe L, Beckmann R, Giertz H, Loschen G (1985) Oxygen-centered free radicals as mediators of inflammation. In: Sies H (ed). Oxidative stress. Academic Press, London, p 403–436
Friesner RA, Murphy RB, Repasky MP, Frye LL, Greenwood JR, Halgren TA, Sanschagrin PC, Mainz DT (2006) Extra precision glide: Docking and scoring incorporating a model of hydrophobic enclosure for protein− ligand complexes. J Med Chem 49:6177–6196
George S, Parameswaran MK, Chakraborty R, Ravi TK (2008) Synthesis and evaluation of the biological activities of some 3-{[5-(6-methyl-4-aryl-2-oxo-1, 2, 3, 4-tetrahydropyrimidin-5-yl)-1, 3, 4-oxadiazol-2-yl]-imino}-1, 3-dihydro-2H-indol-2-one derivatives. Acta Pharm 58:119–129
Geronikaki AA, Lagunin AA, Hadjipavlou-Litina DI, Eleftheriou PT, Filimonov DA, Poroikov VV, Alam I, Saxena AK (2008) Computer-aided discovery of anti-inflammatory thiazolidinones with dual cyclooxygenase/lipoxygenase inhibition. J Med Chem 51:1601–1609
Glide (2018) Schrödinger Release 2018-1. Schrödinger, LLC, New York, NY
Halliwell B, Gutteridge JM (2015) Free radicals in biology and medicine, 5th edn. Oxford University Press, USA
Harder E, Damm W, Maple J, Wu C, Reboul M, Xiang JY, Wang L, Lupyan D, Dahlgren MK, Knight JL (2015) OPLS3: a force field providing broad coverage of drug-like small molecules and proteins. J Chem Theory Comput 12:281–296
Hossain SU, Bhattacharya S (2007) Synthesis of O-prenylated and O-geranylated derivatives of 5-benzylidene2, 4-thiazolidinediones and evaluation of their free radical scavenging activity as well as effect on some phase II antioxidant/detoxifying enzymes. Bioorg Med Chem Lett 17:1149–1154
Ishida T, In Y, Inoue M, Tanaka C, Hamanaka N (1990) Conformation of (Z)-3-carboxymethyl-[(2E)-2-methyl-3-phenylpropenylidene] rhodanine (epalrestat), a potent aldose reductase inhibitor: X-ray crystallographic, energy calculational, and nuclear magnetic resonance studies. J Chem Soc Perkin Trans 2:1085–1091
Ligprep (2018) Schrödinger Release 2018-1. Schrödinger, LLC, New York, NY
Maccari R, Del Corso A, Giglio M, Moschini R, Mura U, Ottanà R (2011) In vitro evaluation of 5-arylidene-2-thioxo-4-thiazolidinones active as aldose reductase inhibitors. Bioorg Med Chem Lett 21:200–203
Maestro (2018) Schrödinger Release 2018-1. Schrödinger, LLC, New York, NY
Mensor LL, Menezes FS, Leitão GG, Reis AS, Santos TCD, Coube CS, Leitão SG (2001) Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytother Res 15:127–130
Momose Y, Meguro K, Ikeda H, Hatanaka C, Oi S, Sohda T (1991) Studies on antidiabetic agents. X. Synthesis and biological activities of pioglitazone and related compounds. Chem Pharm Bull 39:1440–1445
Omar K, Geronikaki A, Zoumpoulakis P, Camoutsis C, Soković M, Ćirić A, Glamočlija J (2010) Novel 4-thiazolidinone derivatives as potential antifungal and antibacterial drugs. Bioorg Med Chem 18:426–432
Orhan H, Şahin G (2001) In vitro effects of NSAIDS and paracetamolon oxidative stress-related parameters of human erythrocytes. Exp Toxicol Pathol 53:133–140
Orlando BJ, Malkowski MG (2016) Crystal structure of rofecoxib bound to human cyclooxygenase-2. Sect Acta Crystallogr F Struct Biol Comm 72:772–776
Ottana R, Maccari R, Barreca ML, Bruno G, Rotondo A, Rossi A, Chiricosta G, Di Paola R, Sautebin L, Cuzzocrea S (2005) 5-Arylidene-2-imino-4-thiazolidinones: design and synthesis of novel anti-inflammatory agents. Bioorg Med Chem 13:4243–4252
Ottanà R, Maccari R, Ciurleo R, Vigorita MG, Panico AM, Cardile V, Garufi F, Ronsisvalle S (2007) Synthesis and in vitro evaluation of 5-arylidene-3-hydroxyalkyl-2-phenylimino-4-thiazolidinones with antidegenerative activity on human chondrocyte cultures. Bioorg Med Chem 15:7618–7625
Oyaizu M (1986) Studies on products of browning reaction, Jpn. J Nutr Diet 44:307–315
Panico AM, Vicini P, Geronikaki A, Incerti M, Cardile V, Crascì L, Messina R, Ronsisvalle S (2011) Heteroarylimino-4-thiazolidinones as inhibitors of cartilage degradation. Bioorg Chem 39:48–52
Saldanha L, Elias G, Rao M (1990) Oxygen radical scavenging activity of phenylbutenones and their correlation with anti-inflammatory activity. Arzneimittelforschung 40:89–91
Schrödinger (2018) Release 2018-4. QikProp, Schrödinger, LLC, New York, NY
Seth K, Garg SK, Kumar R, Purohit P, Meena VS, Goyal R, Banerjee UC, Chakraborti AK (2014) 2-(2-Arylphenyl) benzoxazole as a novel anti-inflammatory scaffold: synthesis and biological evaluation. ACS Med Chem Lett 5:512–516
Shih MH, Ke FY (2004) Syntheses and evaluation of antioxidant activity of sydnonyl substituted thiazolidinone and thiazoline derivatives. Bioorg Med Chem 12:4633–4643
Singh N, Tripathi AC, Tewari A, Kumar SarafSK (2015) Ulcerogenicity devoid novel non-steroidal anti-inflammatory agents (NSAIDS): syntheses, computational studies, and activity of 5-aryliden-2-imino-4-thiazolidinones. Med Chem Res 24:1927–1941
Soare JR, Dinis TC, Cunha AP, Almeida L (1997) Antioxidant activities of some extracts of Thymus zygis. Free Radic Res 26:469–478
Taranalli A, Bhat A, Srinivas S, Saravanan E (2008) anti-inflammatory, analgesic and antipyretic activity of certain thiazolidinones. Indian J Pharm Sci 70:159–164
Tominaga H, Kobayashi Y, Goto T, Kasemura K, Nomura M (2005) DPPH radical-scavenging effect of several phenylpropanoid compounds and their glycoside derivatives. Yakugaku Zasshi 125:371–375
Vicini P, Geronikaki A, Anastasia K, Incerti M, Zani F (2006) Synthesis and antimicrobial activity of novel 2-thiazolylimino-5-arylidene-4-thiazolidinones. Bioorg Med Chem 14:3859–3864
Vicini P, Geronikaki A, Incerti M, Zani F, Dearden J, Hewitt M (2008) 2-Heteroarylimino-5-benzylidene-4-thiazolidinones analogues of 2-thiazolylimino-5-benzylidene-4-thiazolidinones with antimicrobial activity: Synthesis and structure–activity relationship. Bioorg Med Chem 16:3714–3724
Winter CA, Risley EA, Nuss GW (1962) Carrageenin-induced edema in hind paw of the rat as an assay for anti-inflammatory drugs. Proc Soc Exp Biol Med 111:544–547
Acknowledgements
The authors are thankful to Central Drug Research Institute (CDRI) India for spectral characterization.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chawla, P., Kalra, S., Kumar, R. et al. Novel 2-(substituted phenyl Imino)-5-benzylidene-4-thiazolidinones as possible non-ulcerogenic tri-action drug candidates: synthesis, characterization, biological evaluation And docking studies. Med Chem Res 28, 340–359 (2019). https://doi.org/10.1007/s00044-018-02288-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-018-02288-z