Summary
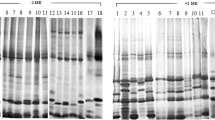
Several high molecular weight endosperm glutenin subunits, coded by genes located on chromosomes 1A, 1B and 1D of common wheat, Triticum aestivum L. em. Thell., were isolated from excised gel segments and subjected to amino acid analysis and peptide mapping; the latter was carried out following a limited digestion with trypsin, chymotrypsin or Staphylococcus aureus — V8 protease. Generally, all high molecular weight glutenins had a similar amino acid composition but several significant differences were observed in some of them. Both analyses revealed that the structural similarity among the various subunits was related to the homology of the genes coding them: subunits coded by homoalleles, i.e., different alleles of the same gene, were most similar; those coded by homoeoalleles, i.e., alleles of homoeologous genes, were less similar; whereas subunits coded either by alleles of different genes of the same gene cluster, or by nonhomoeoalleles of homoeologous clusters, were the least similar. Several small peptides derived from protease digestion of various subunits had a higher than expected staining intensity indicating that small peptide repeats may be interspersed within the glutenin subunits. The evolutionary course of the high molecular weight glutenins is discussed.
Similar content being viewed by others
References
Argos P, Pedersen K, Marks MD, Larkins BA (1982) A structural model for maize zein proteins. J Biol Chem 257:9984–9990
Bartels D, Thompson RD (1983) The characterization of cDNA clones coding for wheat storage proteins. Nucleic Acids Res 11:2961–2977
Bietz JA, Shepherd KW, Wall JS (1975) Single-kernel analysis of glutenin: use in wheat genetics and breeding. Cereal Chem 52:513–532
Brown JWS, Kemble RJ, Law CN, Flavell RB (1979) Control of endosperm protein in Triticum aestivum (var. ‘Chinese Spring’) and Aegilops umbellulata by homoeologous group 1 chromosomes. Genetics 93:189–200
Drapeau GR (1977) Cleavage at glutamic acid with Staphylococcal protease. In: Hirs CHW, Timascheff SN (eds) Methods in enzymology, vol 47. Academic Press, New York, pp 189–191
Fitch WM (1973) Aspects of molecular evolution. Annu Rev Genet 7:343–380
Forde J, Forde BG, Fry RP, Kreis M, Shewry PR, Miflin BJ (1983) Identification of barley and wheat cDNA clones related to the high-MW polypeptides of wheat glutenin. FEBS Lett 162:360–366
Galili G, Feldman M (1983a) Genetic control of endosperm proteins in wheat. 1. The use of high resolution one-dimensional gel electrophoresis for the allocation of genes coding for endosperm protein subunits in the common wheat cultivar ‘Chinese Spring’. Theor Appl Genet 64:97–101
Galili G, Feldman M (1983b) Genetic control of endosperm proteins in wheat. 2. Variation in high molecular weight glutenin and gliadin subunits of Triticum aestivum. Theor Appl Genet 66:77–86
Galili G, Feldman M (1983c) Diploidization of endosperm protein genes in polyploid wheats. In: Proc 6th Int Wheat Genet Symp, Kyoto, Japan, pp 1119–1123
Geraghty D, Peifer MA, Rubenstein I, Messing J (1981) The primary structure of a plant storage protein: zein. Nucleic Acids Res 9:5163–5174
Kasarda DD (1980) Structure and properties of α-gliadins. Ann Technol Agric 29:151–173
Lawrence GJ, Shepherd KW (1980) Variation in glutenin protein subunits of wheat. Aust J Biol Sci 33:221–233
Payne PI, Holt LM, Law CN (1981) Structural and genetical studies on the high-molecular-weight subunits of wheat glutenin. 1. Allelic variation in subunits amongst varieties of wheat (Triticum aestivum). Theor Appl Genet 60: 229–236
Payne PI, Holt LM, Lawrence GJ, Law CN (1982) The genetics of gliadin and glutenin, the major storage proteins of the wheat endosperm. Qual Plant Foods Hum Nutr 31: 229–241
Pedersen K, Devereux J, Wilson DR, Sheldon E, Larkins BA (1982) Cloning and sequence analysis reveal structural variation among related zein genes in maize. Cell 29: 1015–1026
Shewry PR, Field JM, Faulks AJ, Parmar S, Miflin BJ, Dieter MD, Lew EJ-L, Kasarda DD (1984) The purification and N-terminal amino acid sequence analysis of the high molecular weight gluten polypeptides of wheat. Biochim Biophys Acta 788:23–34
Smythe DG (1967) Techniques in enzymic hydrolysis. In: Hirs CHW (ed) Methods in enzymology, vol 11. Academic Press, New York, pp 214–231
Thompson RD, Bartels D, Harberd NP, Flavell RB (1983) Characterization of the multigene family coding for HMW glutenin subunits in wheat using cDNA clones. Theor Appl Genet 67:87–96
Wall JS (1979) The role of wheat proteins in determining baking quality. In: Laidman DL, Wyn Jones RG (eds) Recent advances in the biochemistry of cereals. Academic Press, New York, pp 275–311
Author information
Authors and Affiliations
Additional information
Communicated by J. MacKey
Rights and permissions
About this article
Cite this article
Galili, G., Feldman, M. Structural homology of endosperm high molecular weight glutenin subunits of common wheat (Triticum aestivum L.). Theoret. Appl. Genetics 70, 634–642 (1985). https://doi.org/10.1007/BF00252289
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00252289