Summary
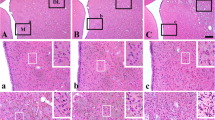
A topographical and cellular immunohistochemical analysis was performed on the striatonigral system of rats with unilateral, reversible middle cerebral artery (MCA) occlusion. Antibodies to calcineurin (CaN), parvalbumin (PV), choline acetyltransferase (ChAT) and glial fibrillary acidic protein (GFAP) were used in this study. Sixty days after the operation, the ipsilateral striatum showed a characteristic cell type-specific injury in the dorsolateral part of the nucleus (i.e., non-limbic striatum): a marked reduction in the number of medium-sized spinous neurons expressing CaN immunoreactivity and a selective sparing of PV-and ChAT-positive interneurons. There was also a marked depletion of striatonigral afferents visualized by CaN immunostaining in the lateral portion of the substantia nigra pars reticulata, which is considered to be implicated with motor function. In addition, it was noted that such striatonigral involvement was accompanied by marked gliosis showing strong GFAP immunolabeling. The present data suggest that rats with reversible MCA occlusion can be a useful animal model for studying cell type-specific ischemic injury and subdivisional involvement of the striatonigral pathway as a part of the cortico-subcortical loop subserving motor function.
Similar content being viewed by others
References
Beal MF, Kowall NW, Ellison DW, Mazurek MF, Schwartz KJ, Martin JB (1986) Replication of the neurochemical characteristics of Huntington's disease by quinolinic acid. Nature 321:168–171
Beal ME, Kowall NW, Schwartz KJ, Ferrante RJ, Martin JB (1988) Model of Huntington's disease. Science 241:475
Beal MF, Kowall NW, Schwartz KJ, Ferrante RJ, Martin JB (1989) Differential sparing of somatostatin-neuropeptide Y and cholinergic neurons following striatal excitotoxin lesions. Synapse 3:38–47
Bolam JP, Wainer BH, Smith AD (1984) Characterization of cholinergic neurons in the rat neostriatum: a combination of choline acetyltransferase immunocytochemistry, Golgi impregnation and electron microscopy. Neuroscience 12:711–718
Borit A, Rubinstein LJ, Urich H (1975) The striatonigral degenerations: putaminal pigments and nosology. Brain 98:101–112
Coyle JT, Schwarcz R (1976) Lesions of striatal neurons with kainic acid provides model for Huntington's chorea. Nature 263:244–246
Crain BJ, Westerkam WD, Harrison AH, Nadler JV (1988) Selective neuronal death after transient forebrain ischemia in the Mongorian gerbil: a silver-impregnation study. Neuroscience 27:387–402
DeLong MR, Georgopoulos AP, Crutcher MD, Mitchell SJ, Richardson RT, Alexander GE (1984) Functional organization of the basal ganglia: contributions of single-cell recording studies. Ciba Found Symp 107:64–78
Evarts EV, Kimura M, Wurtz RH (1984) Behavioral correlates of activity in the basal ganglia neurons. Trend Neurosci 7:447–453
Ferrante RJ, Kowall NW, Beal MF, Richardson EP Jr, Bird ED, Martin JB (1985) Selective sparing of a class of striatal neurons in Huntington's disease. Science 230:561–563
Ferrante RJ, Kowall NW, Beal MF, Martin JB, Bird ED, Richardson EP Jr (1987) Morphologic and histochemical characteristics of a spared set of striatal neurons in Huntington's disease. J Neuropathol Exp Neurol 46:12–27
Ferrante RJ, Beal MF, Kowall NW et al (1987) Sparing of acetylcholinesterase-containing striatal neurons in Huntington's disease. Brain Res 411:162–166
Freund F, Buzsaki G, Leon A, Baimbridge KG, Somogyi P (1990) Relationship for neuronal vulnerability and calcium binding protein immunoreactivity in ischemia. Exp Brain Res 83:55–66
Gerfen CR, Baimbridge KG, Miller JJ (1985) The neostriatal mosaic: complementary distribution of calcium-binding protein and parvalbumin in the basal ganglia of the rat and monkey. Proc Natl Acad Sci USA 82:8780–8784
Goto S, Matsukado Y, Miyamoto E, Yamada M (1987) Morphological characterization of the rat striatal neurons expressing calcineurin immunoreactivity. Neuroscience 22:189–201
Goto S, Hirano A, Rojas-Corona RR (1989) Calcineurin immunoreactivity in striatonigral degeneration. Acta Neuropathol 78:65–71
Goto S, Hirano A, Matsumoto S (1989) Subdivisional involvement of nigro-striatal loop in idiopathic Parkinson's disease and striatonigral degeneration. Ann Neurol 26:766–770
Goto S, Hirano A, Matsumoto S (1990) Immunohistochemical study of striatal efferents and nigral dopaminergic neurons in parkinsonism-dementia complex on Guam in comparison with those in Parkinson's and Alzheimer's diseases. Ann Neurol 27:520–527
Goto S, Hirano A, Matsumoto S (1990) Met-enkephalin immunoreactivity in the basal ganglia in Parkinson's disease and striatonigral degeneration. Neurology 40:1051–1056
Goto S, Nagahiro S, Ushio Y, Hofer W (1992) A simple enhancement method for the silver-gold-intensified diaminobenzidine reaction in the light microscopic immunoperoxidase technique. J Histochem Cytochem 40:1423–1425
Ito H, Goto S, Sakamoto S, Hirano A (1992) Calbindin-D28K in the basal ganglia of patients with parkinsonism. Ann Neurol 32:543–550
Kirino T, Tamura A, Sano K (1984) Delayed neuronal death in the rat hippocampus following transient forebrain ischemia. Acta Neuropathol (Berl) 64:139–147
Koizumi J, Yoshida Y, Nakazawa T, Ooneda G (1986) Experimental studies of ischemic brain edema. I. A new experimental model of cerebral embolism in rats in which recirculation can be introduced in the ischemic area. Jpn J Stroke 8:1–7
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84–91
McGeer PL, McGeer EG (1976) Duplication of biochemical changes of Huntington's chorea by intrastriatal injections of glutamic and kainic acids. Nature 263:517–519
Meldrum BS (1985) Excitatory amino acids and anoxic/ischemic brain damage. Trend Neurosci 8:47–48
Nakano S, Kogure K, Fujikura H (1990) Ischemia-induced slowly progressive neuronal damage in the rat brain. Neuroscience 38:115–124
Nauta WJH, Domesick VB (1984) Afferent and efferent relationships of the basal ganglia. Ciba Found Symp 107:3–23
Petito CK, Pulsinelli WA (1984) Sequential development of reversible and irreversible neuronal damage following cerebral ischemia. J Neuropathol Exp Neurol 43:141–153
Pulsinelli WA, Brierley JB, Plum F (1982) Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann Neurol 11:491–498
Rothman SM, Olney JW (1986) Glutamate and pathophysiology of hypoxic-ischemic brain damage. Ann Neurol 19:105–111
Smith M-L, Auer RN, Siejo BK (1984) The density and distribution of ischemic brain injury in the rat following 2–10 min of forebrain ischemia. Acta Neuropathol (Berl) 64:319–332
Author information
Authors and Affiliations
Additional information
Supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education and Culture of Japan
Rights and permissions
About this article
Cite this article
Goto, S., Nagahiro, S., Korematsu, K. et al. Striatonigral involvement following transient focal cerebral ischemia in the rats: an immunohistochemical study on a reversible ischemia model. Acta Neuropathol 85, 515–520 (1993). https://doi.org/10.1007/BF00230491
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00230491