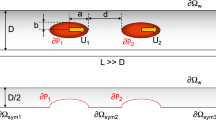
Abstract
Parallel flows of two fluids in microfluidic devices are used for miniaturized chemistry, physics, biology and bioengineering studies, and the streams are often considered to remain parallel. However, as the two fluids do not always have the same density, interface reorientation induced by density stratification is unavoidable. In this paper, flow characteristics of an aqueous polystyrene nanofluid and a sucrose-densified aqueous solution flowing parallel in microchannels are examined. Nanoparticles 100 nm in diameter are used in the study. The motion of the nanoparticles is simulated using the Lagrangian description and directly observed by a confocal microscope. Matched results are obtained from computational and empirical analysis. Although solution density homogenizes rapidly resulting from a fast diffusion of sucrose in water, the nanofluid is observed to rotate for an extended period. Angular displacement of the nanofluid depends on the ratio of gravitational force to viscous force, Re/Fr 2, where Re is the Reynolds number and Fr is the Froude number. In the developing region at the steady state, the angular displacement is related to y/D h, the ratio between distance from the inlet and the hydraulic diameter of the microfluidic channel. The development of nanofluid flow feature also depends on h/w, the ratio of microfluidic channel’s height to width. The quantitative description of the angular displacement of nanofluid will aid rational designs of microfluidic devices utilizing multistream, multiphase flows.









Similar content being viewed by others
Abbreviations
- x, y, z (μm):
-
Spatial coordinates
- h (μm):
-
Microfluidic channel height
- w (μm):
-
Microfluidic channel width
- l (μm):
-
Microfluidic channel length
- D h (μm):
-
Microfluidic channel hydraulic diameter
- L p (μm):
-
Developing length
- ρ (kg m−3):
-
Density of the fluid
- μ (Pa s):
-
Viscosity of the fluid
- p (Pa):
-
Static pressure
- \( \overline{\overline{\tau }} \) (Pa):
-
Stress tensor
- \( \vec{g} \) (m s−2):
-
Gravitational acceleration
- \( \vec{u} \) (m s−1):
-
Fluid phase velocity
- Y i :
-
Local mass fraction of each species
- D i,m (m2 s−1):
-
Diffusion coefficient of species i in the mixture
- \( \rho_{\text{P}} \) (kg m−3):
-
Density of the particles
- d p (μm):
-
Particle diameter
- \( \vec{u}_{\text{p}} \) (m s−1):
-
Particle velocity
- \( \dot{m}_{\text{p}} \) (kg s−1):
-
Mass flow rate of the particles
- \( \Updelta t \) (s):
-
Time step
- \( C_{\text{D}} \) :
-
Drag coefficient
- \( F_{\text{D}} \) (kg m s−2):
-
Drag force
- T (K):
-
Absolute temperature of the fluid
- k B :
-
Boltzmann constant
- d i,j (Pa):
-
Deformation tensor
- Fr :
-
Froude number
- Re :
-
Reynolds number
- \( Re_{\text{p}} \) :
-
Relative Reynolds number
References
Abecassis B, Cottin-Bizonne C, Ybert C, Ajdari A, Bocquet L (2008) Boosting migration of large particles by solute contrasts. Nat Mater 7(10):785–789. doi:10.1038/nmat2254
Bazylak A, Sinton D, Djilali N (2005) Improved fuel utilization in microfluidic fuel cells: a computational study. J Power Sources 143(1–2):57–66. doi:10.1016/j.jpowsour.2004.11.029
Bresme F, Oettel M (2007) Nanoparticles at fluid interfaces. J Phys: Condens Matter 19(41):413101. doi:10.1088/0953-8984/19/41/413101
Chen H, Meiners J-C (2004) Topologic mixing on a microfluidic chip. Appl Phys Lett 84(12):2193. doi:10.1063/1.1686895
Choban ER, Markoski LJ, Wieckowski A, Kenis PJA (2004) Microfluidic fuel cell based on laminar flow. J Power Sources 128(1):54–60. doi:10.1016/j.jpowsour.2003.11.052
Cohen JL, Westly DA, Pechenik A, Abruña HD (2005) Fabrication and preliminary testing of a planar membraneless microchannel fuel cell. J Power Sources 139(1–2):96–105. doi:10.1016/j.jpowsour.2004.06.072
Drew DA (1983) Mathematical-modeling of 2-phase flow. Annu Rev Fluid Mech 15:261–291. doi:10.1146/annurev.fl.15.010183.001401
Ferrigno R, Stroock AD, Clark TD, Mayer M, Whitesides GM (2002) Membraneless vanadium redox fuel cell using laminar flow. J Am Chem Soc 124(44):12930–12931. doi:10.1021/ja020812q
Grzelczak M, Vermant J, Furst EM, Liz-Marzan LM (2010) Directed self-assembly of nanoparticles. ACS Nano 4(7):3591–3605. doi:10.1021/nn100869j
Hatch A, Kamholz AE, Hawkins KR, Munson MS, Schilling EA, Weigl BH, Yager P (2001) A rapid diffusion immunoassay in a T-sensor. Nat Biotechnol 19(5):461–465. doi:10.1038/88135
Hessel V, Hardt S, Lowe H, Schonfeld F (2003) Laminar mixing in different interdigital micromixers: I. Experimental characterization. Aiche J 49(3):566–577. doi:10.1002/aic.690490304
Huh D, Bahng JH, Ling YB, Wei HH, Kripfgans OD, Fowlkes JB, Grotberg JB, Takayama S (2007) Gravity-driven microfluidic particle sorting device with hydrodynamic separation amplification. Anal Chem 79(4):1369–1376. doi:10.1021/ac061542n
Ismagilov RF, Stroock AD, Kenis PJA, Whitesides G, Stone HA (2000) Experimental and theoretical scaling laws for transverse diffusive broadening in two-phase laminar flows in microchannels. Appl Phys Lett 76(17):2376. doi:10.1063/1.126351
Jeong W, Kim J, Kim S, Lee S, Mensing G, Beebe DJ (2004) Hydrodynamic microfabrication via “on the fly’’ photopolymerization of microscale fibers and tubes. Lab Chip 4(6):576–580. doi:10.1039/b411249k
Kamholz AE, Yager P (2001) Theoretical analysis of molecular diffusion in pressure-driven laminar flow in microfluidic channels. Biophys J 80(1):155–160
Kamholz AE, Weigl BH, Finlayson BA, Yager P (1999) Quantitative analysis of molecular interaction in a microfluidic channel: the T-sensor. Anal Chem 71(23):5340–5347. doi:10.1021/ac990504j
Kamholz AE, Schilling EA, Yager P (2001) Optical measurement of transverse molecular diffusion in a microchannel. Biophys J 80(4):1967–1972
Kane RS, Takayama S, Ostuni E, Ingber DE, Whitesides GM (1999) Patterning proteins and cells using soft lithography. Biomaterials 20(23–24):2363–2376. doi:10.1016/s0142-9612(99)00165-9
Kang YJ, Yoon SY, Lee KH, Yang S (2010) A highly accurate and consistent microfluidic viscometer for continuous blood viscosity measurement. Artif Organs 34(11):944–949. doi:10.1111/j.1525-1594.2010.01078.x
Kenis PJA, Ismagilov RF, Whitesides GM (1999) Microfabrication inside capillaries using multiphase laminar flow patterning. Science 285(5424):83–85. doi:10.1126/science.285.5424.83
Kohnen G, Sommerfeld M (1998) Numerische berechnung verdampfender sprühnebel. Chem Tech (Leipzig) 50:225–234
Li A, Ahmadi G (1992) Dispersion and deposition of spherical-particles from point sources in a turbulent channel flow. Aerosol Sci Tech 16(4):209–226. doi:10.1080/02786829208959550
Lin Y, Skaff H, Emrick T, Dinsmore AD, Russell TP (2003) Nanoparticle assembly and transport at liquid–liquid interfaces. Science 299(5604):226–229. doi:10.1126/science.1078616
Masoumi N, Sohrabi N, Behzadmehr A (2009) A new model for calculating the effective viscosity of nanofluids. J Phys D Appl Phys 42(5):055501. doi:10.1088/0022-3727/42/5/055501
Paek SH, Choi YK, Kim DS (2010) Selective microfabrication of silver electrodes inside a microchannel by multiphase laminar flow with density difference. Microelectron Eng 87(5–8):1375–1378. doi:10.1016/j.mee.2009.11.176
Paek SH, Yang W, Kim JS, Hwang CJ, Choi YK, Kim DS (2011) Microfabrication of dual-opposite electrodes inside a microchannel by means of multiphase laminar flow with density difference. Micro Nano Lett 6(8):678. doi:10.1049/mnl.2011.0277
Pollack L, Tate MW, Darnton NC, Knight JB, Gruner SM, Eaton WA, Austin RH (1999) Compactness of the denatured state of a fast-folding protein measured by submillisecond small-angle x-ray scattering. P Natl Acad Sci USA 96(18):10115–10117. doi:10.1073/pnas.96.18.10115
Pollack L, Tate M, Finnefrock A, Kalidas C, Trotter S, Darnton N, Lurio L, Austin R, Batt C, Gruner S, Mochrie S (2001) Time resolved collapse of a folding protein observed with small angle x-ray scattering. Phys Rev Lett 86(21):4962–4965. doi:10.1103/PhysRevLett.86.4962
Saffman PG (1965) Lift on a small sphere in a slow shear flow. J Fluid Mech 22(2):385–400. doi:10.1017/s0022112065000824
Stiles PJ, Fletcher DF (2003) Effects of gravity on the steady state of a reaction in a liquid-state microreactor—deviations from Poiseuille flow. Phys Chem Chem Phys 5(6):1219–1224. doi:10.1039/b211686c
Stroock AD (2002) Chaotic mixer for microchannels. Science 295(5555):647–651. doi:10.1126/science.1066238
Takayama S, McDonald JC, Ostuni E, Liang MN, Kenis PJA, Ismagilov RF, Whitesides GM (1999) Patterning cells and their environments using multiple laminar fluid flows in capillary networks. P Natl Acad Sci USA 96(10):5545–5548. doi:10.1073/pnas.96.10.5545
Takayama S, Ostuni E, LeDuc P, Naruse K, Ingber DE, Whitesides GM (2001a) Laminar flows—Subcellular positioning of small molecules. Nature 411(6841):1016–1016. doi:10.1038/35082637
Takayama S, Ostuni E, Qian XP, McDonald JC, Jiang XY, LeDuc P, Wu MH, Ingber DE, Whitesides GM (2001b) Topographical micropatterning of poly(dimethylsiloxane) using laminar flows of liquids in capillaries. Adv Mater 13(8):570–574. doi:10.1002/1521-4095(200104)13:8<570::aid-adma570>3.0.co;2-b
Takayama S, Ostuni E, LeDuc P, Naruse K, Ingber DE, Whitesides GM (2003) Selective chemical treatment of cellular microdomains using multiple laminar streams. Chem Biol 10(2):123–130. doi:10.1016/s1074-5521(03)00019-x
Tokeshi M, Minagawa T, Uchiyama K, Hibara A, Sato K, Hisamoto H, Kitamori T (2002) Continuous-flow chemical processing on a microchip by combining microunit operations and a multiphase flow network. Anal Chem 74(7):1565–1571. doi:10.1021/ac011111z
Weigl BH, Yager P (1999) Microfluidics—microfluidic diffusion-based separation and detection. Science 283(5400):346–347. doi:10.1126/science.283.5400.346
Xuan J, Leung MKH, Leung DYC, Ni M (2011) Density-induced asymmetric pair of Dean vortices and its effects on mass transfer in a curved microchannel with two-layer laminar stream. Chem Eng J 171(1):216–223. doi:10.1016/j.cej.2011.01.011
Yamada M, Nakashima M, Seki M (2004) Pinched flow fractionation: continuous size separation of particles utilizing a laminar flow profile in a pinched microchannel. Anal Chem 76(18):5465–5471. doi:10.1021/ac049863r
Yamaguchi Y, Takagi F, Watari T, Yamashita K, Nakamura H, Shimizu H, Maeda H (2004) Interface configuration of the two layered laminar flow in a curved microchannel. Chem Eng J 101(1–3):367–372. doi:10.1016/j.cej.2003.10.018
Yamaguchi Y, Honda T, Briones MP, Yamashita K, Miyazaki M, Nakamura H, Maeda H (2006) Influence of gravity on a laminar flow in a microbioanalysis system. Meas Sci Technol 17(12):3162–3166. doi:10.1088/0957-0233/17/12/s07
Yamaguchi Y, Honda T, Briones MP, Yamashita K, Miyazaki M, Nakamura H, Maeda H (2007) Influence of gravity on two-layer laminar flow in a microchannel. Chem Eng Technol 30(3):379–382. doi:10.1002/ceat.200600326
Yamashita K, Yamaguchi Y, Miyazaki M, Nakamura H, Shimizu H, Maeda H (2004) Sequence-selective DNA detection using multiple laminar streams: a novel microfluidic analysis method. Lab Chip 4(1):1–3. doi:10.1039/b311571b
Yang Y, Oztekin A, Neti S, Mohapatra S (2012) Particle agglomeration and properties of nanofluids. J Nanopart Res 14(5):852. doi:10.1007/s11051-012-0852-2
Yoon SK, Mitchell M, Choban ER, Kenis PJ (2005) Gravity-induced reorientation of the interface between two liquids of different densities flowing laminarly through a microchannel. Lab Chip 5(11):1259–1263. doi:10.1039/b508680a
Zhang DZ, Prosperetti A (1997) Momentum and energy equations for disperse two-phase flows and their closure for dilute suspensions. Int J Multiph Flow 23(3):425–453. doi:10.1016/s0301-9322(96)00080-8
Zhao C, Cheng XH (2011) Microfluidic separation of viruses from blood cells based on intrinsic transport processes. Biomicrofluidics 5(3):032004. doi:10.1063/1.3609262
Zhao B, Moore JS, Beebe DJ (2001) Surface-directed liquid flow inside microchannels. Science 291(5506):1023–1026. doi:10.1126/science.291.5506.1023
Zhao JJ, Duan YY, Wang XD, Wang BX (2011) Effect of nanofluids on thin film evaporation in microchannels. J Nanopart Res 13(10):5033–5047. doi:10.1007/s11051-011-0484-y
Zhao JJ, Duan YY, Wang XD, Wang BX (2012) Effects of solid–gas coupling and pore and particle microstructures on the effective gaseous thermal conductivity in aerogels. J Nanopart Res 14(8):1024. doi:10.1007/s11051-012-1024-0
Zheng B, Roach LS, Ismagilov RF (2003) Screening of protein crystallization conditions on a microfluidic chip using nanoliter-size droplets. J Am Chem Soc 125(37):11170–11171. doi:10.1021/ja037166v
Zheng B, Tice JD, Ismagilov RF (2004) Formation of droplets of in microfluidic channels alternating composition and applications to indexing of concentrations in droplet-based assays. Anal Chem 76(17):4977–4982. doi:10.1021/ac0495743
Acknowledgments
We are grateful for the helpful discussions about confocal microscopy with Prof. H. Daniel Ou-Yang, Yi Hu, and Ming-Tzo Wei, about FLUENT with Yan Xu. We also thank Krissada Surawathanawises for Scanning Electron Microscope imaging. Funding for the research is provided by National Institute of Health under Grant No NIAID-1R21AI081638.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, C., Oztekin, A. & Cheng, X. Gravity-induced swirl of nanoparticles in microfluidics. J Nanopart Res 15, 1611 (2013). https://doi.org/10.1007/s11051-013-1611-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-013-1611-8