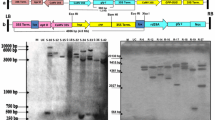
Abstract
Main conclusion
Heterologous expression of a fungal NADP(H)-GDH gene ( MgGDH ) from Magnaporthe grisea can improve dehydration stress tolerance in rice by preventing toxic accumulation of ammonium.
Glutamate dehydrogenase (GDH; EC 1.4.1.2 and EC 1.4.1.4) may act as a stress-responsive enzyme in detoxification of high intracellular ammonia and production of glutamate for proline synthesis under stress conditions. In present study, a fungal NADP(H)-GDH gene (MgGDH) from Magnaporthe grisea was over-expressed in rice (Oryza sativa L. cv. ‘kitaake’), and the transgenic plants showed the improvement of tolerance to dehydration stress. The kinetic analysis showed that His-TF-MgGDH preferentially utilizes ammonium to produce l-glutamate. Moreover, the affinity of His-TF-MgGDH for ammonium was dramatically higher than that of His-TF-OsGDH for ammonium. Over-expressing MgGDH transgenic rice plants showed lower water-loss rate and higher completely close stomata than the wild-type plants under dehydration stress conditions. In transgenic plants, the NADP(H)-GDH activities were markedly higher than those in wild-type plants and the amination activity was significantly higher than the deamination activity. Compared with wild-type plants, the transgenic plants accumulated much less NH4 + but higher amounts of glutamate, proline and soluble sugar under dehydration stress conditions. These results indicate that heterologous expression of MgGDH can prevent toxic accumulation of ammonium and in return improve dehydration stress tolerance in rice.





Similar content being viewed by others
Abbreviations
- Mg:
-
Magnaporthe grisea
- GDH:
-
Glutamate dehydrogenase
- GOGAT:
-
Glutamate synthase
- GS:
-
Glutamine synthetase
- 2-OG:
-
2-oxoglutarate
- TF:
-
Trigger factor
- P5CS:
-
△1-Pyrroline-5-carboxylate synthetase
- P5CR:
-
△1-Pyrroline-5-carboxylic acid reductase
- GSA:
-
Glutamic γ-semialdehyde
- PCR:
-
Polymerase chain reaction
References
Abiko T, Obara M, Ushioda A, Hayakawa T, Hodges M, Yamaya T (2005) Localization of NAD-isocitrate dehydrogenase and glutamate dehydrogenase in rice roots: candidates for providing carbon skeletons to NADH-glutamate synthase. Plant Cell Physiol 46:1724–1734
Abiko T, Wakayama M, Kawakami A, Obara M, Kisaka H, Miwa T, Aoki N, Ohsugi R (2010) Changes in nitrogen assimilation, metabolism, and growth in transgenic rice plants expressing a fungal NADP(H)-dependent glutamate dehydrogenase (gdhA). Planta 232:299–311
Abraham E, Rigo G, Szekely G, Nagy R, Koncz C, Szabados L (2003) Light-dependent induction of proline biosynthesis by abscisic acid and salt stress is inhibited by brassinosteroid in Arabidopsis. Plant Mol Biol 51:363–372
Ameziane R, Bernhard K, Lightfoot D (2000) Expression of the bacterial gdhA gene encoding a NADPH glutamate dehydrogenase in tobacco affects plant growth and development. Plant Soil 221:47–57
Armengaud P, Thiery L, Buhot N, Grenier-De March G, Savoure A (2004) Transcriptional regulation of proline biosynthesis in Medicago truncatula reveals developmental and environmental specific features. Physiol Plant 120:442–450
Boyer JS (1982) Plant productivity and environment. Science 218:443–448
Britto DT, Kronzucker HJ (2002) NH4 + toxicity in higher plants: a critical review. J Plant Physiol 159:567–584
Calle F, Martin M, Sabater B (1986) Cytoplasmic and mitochondrial localization of the glutamate dehydrogenase induced by senescence in barley (Hordeum vulgare). Physiol Plant 66:451–456
Cao WH, Liu J, He XJ, Mu RL, Zhou HL, Chen SY, Zhang JS (2007) Modulation of ethylene responses affects plant salt-stress responses. Plant Physiol 143:707–719
Chen QJ, Zhou HM, Chen J, Wang XC (2006) Using a modified TA cloning method to create entry clones. Anal Biochem 358:120–125
Chyzhykova OA, Palladina TO (2006) The role of amino acids and sugars in supporting of osmotic homeostasis in maize seedlings under salinization conditions and treatment with synthetic growth regulators. Ukr Biokhim Zh 78:124–129
De Ronde JA, Spreeth MH, Cress WA (2000) Effect of antisense l-△1-pyrroline-5-carboxylate reductase transgenic soybean plants subjected to osmotic and drought stress. Plant Growth Regul 32:13–26
Delauney AJ, Verma DPS (1993) Proline biosynthesis and osmoregulation in plants. Plant J 4:215–223
Delauney AJ, Hu C-AA, Kavi Kishor PB, Verma DPS (1993) Cloning of ornithined δ-aminotransferase cDNA from Vigna aconitifolia by trans-complementation in E. coli and regulation of proline biosynthesis. J Biol Chem 268:18673–18678
Du C, Lin J, Yang Y, Liu H, Li C, Zhou Y, Li Y, Tang D, Zhao X, Zhu Y, Liu X (2014) Molecular cloning, characterization and function analysis of a GDH gene from Sclerotinia sclerotiorum in rice. Mol Biol Rep 41:3683–3693
Fukao T, Yeung E, Bailey-Serres J (2011) The submergence tolerance regulator SUB1A mediates crosstalk between submergence and drought tolerance in rice. Plant Cell 23:412–427
Garg AK, Kim JK, Owens TG, Ranwala AP, Choi YD, Kochian LV, Wu RJ (2002) Trehalose accumulation in rice plants confers high tolerance levels to different abiotic stresses. Proc Natl Acad Sci USA 99:15898–15903
Gilmour SJ, Sebolt AM, Salazar MP, Everard JD, Thomashow MF (2000) Overexpression of the Arabidopsis CBF3 transcriptional activator mimics multiple biochemical changes associated with cold acclimation. Plant Physiol 124:1854–1865
Gulati A, Jaiwal PK (1996) Effect of NaCl on nitrate reductase, glutamate dehydrogenase and glutamate in Vigna radiata calli. Bio. Plant 38:177–183
Gupta AK, Kaur N (2005) Sugar signalling and gene expression in relation to carbohydrate metabolism under abiotic stresses in plants. J Biosci 30:761–776
Hetherington AM, Woodward FI (2003) The role of stomata in sensing and driving environmental change. Nature 424:901–908
Hoai NTT, Shim IS, Kobayashi K, Usui K (2003) Accumulation of some nitrogen compounds in response to salt stress and their relationships with salt tolerance in rice (Oryza sativa) seedlings. Plant Growth Regul 41:159–164
Ito Y, Katsura K, Maruyama K, Taji T, Kobayashi M, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2006) Functional analysis of rice DREB1/CBF-type transcription factors involved in cold-responsive gene expression in transgenic rice. Plant Cell Physiol 47:141–153
Kanamori K, Weiss RL, Roberts JD (1987) Role of glutamate dehydrogenase in ammonia assimilation in nitrogen-fixing Bacillus macerans. J Bacteriol 169:4692–4695
Kinghorn JR, Pateman JA (1973) NAD and NADP l-glutamate dehydrogenase activity and ammonium regulation in Aspergillus nidulans. J Gen Microbiol 78:39–46
Kishor P, Hong Z, Miao GH, Hu C, Verma D (1995) Overexpression of △1-pyrroline-5-carboxylate synthetase increases proline production and confers osmotolerance in transgenic plants. Plant Physiol 108:1387–1394
Krug FJ, Ru˚zˇic ˇka J, Hansen EH (1979) Determination of ammonia in low concentrations with Nessler’s reagent by flow injection analysis. Analyst 104:47–54
Kumar RG, Shah K, Dubey RS (2000) Salinity induced behavioral changes in malate dehydrogenase and glutamate dehydrogenase activities in rice seedlings of differing salt tolerance. Plant Sci 156:23–34
Lasa B, Frechilla S, Aparicio-Tejo PM, Lamsfus C (2002) Role of glutamate dehydrogenase and phosphoenolpyruvate carboxylase activity in ammonium nutrition tolerance in roots. Plant Physiol Biochem 40:969–976
Lauriere C, Daussant J (1983) Identification of the ammonia-dependent-isoenzyme of glutamate dehydrogenase as the form induced by senescence or darkness stress in the first leaf of wheat. Physiol Plant 48:89–92
Lea PJ, Miflin BJ (1974) Alternative route for nitrogen assimilation in higher plants. Nature 18:614–616
Li C, Yu L, Liu Z, Zhu L, Hu Y, Zhu M, Zhu X, Shi Y, Meng S (2006) Schistosoma japonicum: the design and experimental evaluation of a multivalent DNA vaccine. Cell Mol Biol Lett 11:449–460
Lightfoot DA, Mungur R, Ameziane R, Nolte S, Long L, Bernhard K, Colter A, Jones K, Iqbal MJ, Varsa E, Yong B (2007) Improved drought tolerance of transgenic Zea mays plants that express the glutamate dehydrogenase gene (gdhA) of E. coli. Euphytica 156:103–116
Lin JZ, Zhou B, Yang YZ, Mei J, Guo XH, Huang XQ, Tang DY, Liu XM (2009) Piercing and vacuum infiltration of the mature embryo: a simplified Method for Agrobacterium mediated transformation of Indica rice. Plant Cell Rep 28:1065–1074
Liu J, Zhu JK (1997) Proline accumulation and salt-stress-induced gene expression in a salt-hypersensitive mutant of Arabidopsis. Plant Physiol 114:591–596
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2[−△ △C(T)] method. Methods 25:402–408
Loulakakis KA, Primikirios NI, Nikolantonakis MA, Roubelakis-Angelakis KA (2002) Immunocharacterization of Vitis vinifera L. ferredoxin-dependent glutamate synthase and its spatial and temporal changes during leaf development. Planta 215:630–638
Lutts S, Majerus V, Kinet JM (1999) NaCl effects on proline metabolism in rice (Oryza sativa) seedlings. Physiol Plant 105:450–458
Masclaux C, Valadier MH, Brugie`re N, Morot-Gaudry JF, Hirel B (2000) Characterization of the sink/source transition in tobacco (Nicotiana tabacum L.) shoots in relation to nitrogen management and leaf senescence. Planta 211:510–518
Mattioni C, Gabbrielli R, Vongronsveld J, Clijsters H (1997) Nickel and cadmium toxicity and enzymatic activity in Ni tolerant and non-tolerant populations of Silene italica pers. J Plant Physiol 150:173–177
Miflin BJ, Lea PJ (1980) Ammonia assimilation. In: Miflin BJ (ed) The biochemistry of plants, vol 5. Academic Press, New York, pp 169–202
Mungur R (2002) Metabolic profiles of GDH transgenic crops. MS thesis, SIUC Carbondale
Mungur R, Wood AJ, Lightfoot DA (2006) Water potential is maintained during water deficit in Nicotiana tabacum expressing the E. coli glutamate dehydrogenase gene. Plant Growth Regul 50:231–238
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Nanjo T, Kobayashi M, Yoshiba Y, Kakubari Y, Yamaguchi-Shinozaki K, Shinozaki K (1999a) Antisense suppression of proline degradation improves tolerance to freezing and salinity in Arabidopsis thaliana. FEBS Lett 461:205–210
Nanjo T, Kobayashi M, Yoshiba Y, Sanada Y, Wada K, Tsukaya H, Kakubari Y, Yamaguchi-Shinozaki K, Shinozaki K (1999b) Biological functions of proline in morphogenesis and osmotolerance revealed in antisense transgenic Arabidopsis thaliana. Plant J 18:185–193
Nolte SA, Young BG, Mungur R, Lightfoot DA (2004) The glutamate dehydrogenase gene gdhA increased the resistance of tobacco to glufosinate. Weed Res 44:335–339
Noor S, Punekar NS (2005) Allosteric NADP-glutamate dehydrogenase from aspergilli: purification, characterization and implications for metabolic regulation at the carbon–nitrogen interface. Microbiology 151:1409–1419
Ohta S, Mita S, Hattori T, Nakamura K (1990) Construction and expression in tobacco of a β-glucuronidase (GUS) reporter gene containing an intron within coding sequence. Plant Cell Physiol 31:805–813
Robinson SA, Slade AP, Fore GG, Phillips R, Radcliffe RG, Stewart GR (1991) The role of GDH in plant nitrogen metabolism. Plant Physiol 95:509–516
Skopelitis DS, Paranychianakis NV, Paschalidis KA, Pliakonis ED, Delis ID, Yakoumakis DI, Kouvarakis A, Papadakis AK, Stephanou EG, Roubelakis-Angelakis KA (2006) Abiotic stress generates ROS that signal expression of anionic glutamate dehydrogenases to form glutamate for proline synthesis in tobacco and grapevine. Plant Cell 18:2767–2781
Srivastava HS, Singh RP (1987) Role and regulation of l-glutamate dehydrogenase activity in higher plants. Phytochemistry 26:597–610
Sweetlove LJ, Mowday B, Hebestreit HF, Leaver CJ, Millar AH (2001) Nucleoside diphosphate kinase III is localized to the intermembrane space in plant mitochondria. FEBS Lett 508:272–276
Tabuchi A, Kikui S, Matsumoto H (2004) Differential effects of aluminium on osmotic potential and sugar accumulation in the root cells of Al-resistant and Al-sensitive wheat. Physiol Plant 120:106–112
Terce´-Laforgue T, Dubois F, Ferrario-Mery S, Crecenzo MAP, Sangwan R, Hirel B (2004a) Glutamate dehydrogenase of tobacco is mainly induced in the cytosol of phloem companion cells when ammonia is provided either externally or released during photorespiration. Plant Physiol 136:4308–4317
Terce´-Laforgue T, Mack G, Hirel B (2004b) New insights towards the function of glutamate dehydrogenase revealed during source-sink transition of tobacco (Nicotiana tabacum) plants grown under different nitrogen regimes. Physiol Plant 120:220–228
Toki S, Hara N, Ono K, Onodera H, Tagiri A, Oka S, Tanaka H (2006) Early infection of scutellum tissue with Agrobacterium allows high-speed transformation of rice. Plant J 47:969–976
Valliyodan B, Nguyen HT (2006) Understanding regulatory networks and engineering for enhanced drought tolerance in plants. Curr Opin Plant Biol 9:189–195
Vines HM, Wedding RT (1960) Some effects of ammonia on plant metabolism and a possible mechanism for ammonia toxicity. Plant Physiol 35:820–825
Wang F, Tian B (2001) Neurospora NADP glutamate dehydrogenases and its expression in E. coli and transgenic plant. Chin Sci Bull 46:137–140
Wootton JC (1983) Re-assessment of ammonium-ion affinities of NADP-specific glutamate dehydrogenases. Biochem J 209:527–531
Xiang Y, Huang YM, Xiong LZ (2007) Characterization of stress-responsive CIPK genes in rice for stress tolerance improvement. Plant Physiol 144:1416–1428
Xiao B, Huang Y, Tang N, Xiong L (2007) Over-expression of a LEA gene in rice improves drought resistance under the field conditions. Theor Appl Genet 115:35–46
Yamaya T, Oaks A (1987) Synthesis of glutamate by mitochondria—an anaplerotic function for glutamate dehydrogenase. Physiol Plant 70:749–756
Ye H, Du H, Tang N, Li X, Xiong L (2009) Identification and expression profiling analysis of TIFY family genes involved in stress and phytohormone responses in rice. Plant Mol Biol 71:291–305
Yoo SD, Cho YH, Sheen J (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2:1565–1572
Yoshiba Y, Kiyosue T, Nakashima K, Yamaguchi-Shinozaki K, Shinozaki K (1997) Regulation of levels of proline as an osmolyte in plants under water stress. Plant Cell Physiol 38:1095–1102
Zhang HN, Wang ZQ, Cui GJ, Lin TB (2009) Difference in seedlings ammonium assimilation of wheat cultivars with different drought resistance under osmotic stress. Ying Yong Sheng Tai Xue Bao 20:2406–2410
Zhang SX, Haider I, Kohlen W, Jiang L, Bouwmeester H, Meijer AH, Schluepmann H, Liu CM, Ouwerkerk PBF (2012) Function of the HD-Zip I gene Oshox22 in ABA-mediated drought and salt tolerances in rice. Plant Mol Biol 80:571–585
Zhou XC, Lin JZ, Zhou YB, Yang YZ, Liu H, Zhang CS, Tang DY, Zhao XY, Zhu YH, Liu XM (2014a) Changes in nitrogen utilization and growth in transgenic rice over-expressing a fungal NADP(H)-dependent glutamate dehydrogenase (CeGDH). Crop Sci, pp 1–22
Zhou YB, Liu H, Zhou XC, Yan YZ, Du CQ, Li YX, Liu DR, Zhang CS, Deng XL, Tang DY, Zhao XY, Zhu YH, Lin JZ, Liu XM (2014b) Over-expression of a fungal NADP(H)-dependent glutamate dehydrogenase PcGDH improves nitrogen assimilation and growth quality in rice. Mol Breed 34:335–349
Acknowledgments
The authors thank Dr. Hong Liu for providing the fungi Magnaporthe grisea and assistance of cloning MgGDH. This research was supported by Important National Science and Technology Specific Projects (No. 2009ZX08001-030B), National Science Foundation of China (No. 31170172), Hunan Provincial Natural Science Foundation of China (No.12JJ3024), Cooperative Innovation Center of Engineering and New Products for Developmental Biology of Hunan Province (No. 20134486), Planned Science and Technology Project of Hunan Province (No. 2014WK2005 and No. 2014NK3001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Y. Zhou, C. Zhang and J. Lin contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhou, Y., Zhang, C., Lin, J. et al. Over-expression of a glutamate dehydrogenase gene, MgGDH, from Magnaporthe grisea confers tolerance to dehydration stress in transgenic rice. Planta 241, 727–740 (2015). https://doi.org/10.1007/s00425-014-2214-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-014-2214-z