Abstract
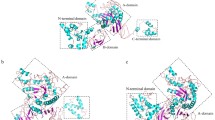
The importance of 17 glutamate residues of a truncated Bacillus sp. strain TS-23 α-amylase (BACΔNC) was investigated by site-directed mutagenesis. The Ala- and Asp-substituted variants were overexpressed in the recombinant E. coli cells and the 54-kDa proteins were purified to nearly homologous by nickel-chelate chromatography. Glu-295, which locates in the conserved region III of amylolytic enzymes, mutations resulted in a complete loss of enzyme activity. The specific activity for E151A was decreased by more than 30%, while other variants showed activity comparable to that of BACΔNC. A decreased half-life at 70°C was observed for Glu-219 variants with respective to the wild-type enzyme, suggesting that replacement of Glu-219 by either Ala or Asp might have a significant destabilizing effect on the protein structure.





Similar content being viewed by others
Abbreviations
- BACΔNC:
-
the N- and C-terminally truncated Bacillus sp. strain TS-23 α-amylase
- SDS:
-
sodium dodecyl sulfate
- SDS-PAGE:
-
SDS-polyacrylamide gel electrophoresis
- TEMED:
-
N,N,N′,N′-tetramethylethylenediamine
- IPTG:
-
isopropyl-β-d-thiogalactopyranoside
- Ni2+-NTA:
-
nickel nitrilotriacetate
- BLA:
-
Bacillus licheniformis α-amylase
References
Boel E., Brady L., Brzozowski A. M., Derewenda Z., Dodson G. G., Jensen V. J., Petersen S. B., Swift H., Thim L., Woldike H. F. (1990). Biochemistry 29:6244–6249
Bradford, M. M. (1976). Anal. Chem. 72:248–254
Buisson G., Duee E., Haser R., and Payan, F. (1987). EMBO J. 6:3909–3916
Chang, C. T., Lo, H. F., Chi, M. C., Yao, C. Y., Hsu, W. H., and Lin, L. L. (2003). Extremophiles 7:505–509
Declerck, N., Machius, M., Wiegand, G., Huber, R., and Gaillardin, C. (2000). J. Mol. Biol. 301:1041–1057
Guzman-Maldonado, H., and Paredes-Lopez, O. (1995). Crit. Rev. Food Sci. Nutr. 35:373–403
Henrissat, B. (1991). Biochem. J. 280:309–316
Ito, S., Kobayashi, T., Ara, K., Ozaki, K., Kawai, S., and Hatada, Y. (1998). Extremophiles 2:185–190
Kang, Y.N., Adachi, M., Utsumi, S., and Mikami, B. (2004). J. Mol. Biol. 339:1129–1140
Karn, R. C. (1978). Adv. Comp. Physiol. Biochem. 7:1–103
Katsuya, Y., Mezaki, Y., Kubota, M., and Matsuura, Y. (1998). J. Mol. Biol. 281:885–897
Kim, J. S., Chan, S. S., Kim, H. J., Kim, T. J., Ha, N. C., Oh, S. T., Cho, H. S., Cho, M. J., Kim, M. J., Lee, H. S., Kim, J. W., Choi, K. Y., Park, K. H., and Oh, B. H. (1999). J. Biol. Chem. 274: 26279–26286
Kossmann, J., and Lloyd, J. (2000). Crit. Rev. Biochem. Mol. Biol. 35:141–196
Kuriki, T., and Imanaka, T. (1999). J. Biosci. Bioeng. 87:557–565
Laemmli, U. K. (1970). Nature (Lodon) 227:680–685
Lawson, C. L., van Montfort, R., Strokopytov, B., Rozeboom, H. J., Kalk, K. H., de Vries, G., Penninga, D., Dijkhuizen, L., and Dijkstra, B. W. (1994). J. Mol. Biol. 236:590–600
Lehmann, M., Pasamontes, L., Lassen, S. F., Wyss, M. (2000). Biochim. Biophys. Acta 1543:408–415
Li, F., and Mullins, J. I. (2002). Methods Mol. Biol. 182:19–27
Lin, L. L., Hsu, W. H., and Chu, W. S. (1997). J. Appl. Microbiol. 82:325–334
Lin, L. L., Tsau, M. R., and Chu, W. S. (1994). Appl. Microbial. Biotechnol. 42:51–56
Lo, H. F., Lin, L. L., Li, C. C., Hsu, W. H., and Chang, C. T. (2001). Curr. Microbiol. 43:170–175
Lo, H. F., Lin, L. L., Chiang, W. Y., Chi, M. C., Hsu, W. H., and Chang, C. T. (2002). Arch. Microbiol. 178:115–123
Machius, M., Wiegand, G., and Huber, R. (1995). J. Mol. Biol. 246:545–559
Matsuura, Y., Kusunoki, M., Harada, W., and Kakudo, M. (1984). J. Biochem (Tokyo) 95:697–702
Miller, G. L. (1959). Anal. Chem. 31:426–428
Morishita, Y., Hasegawa, K., Matsuura, Y., Katsuya, Y., Kubato, M., and Sakai, S. (1997). J. Mol. Biol. 267: 661–672
Ohdan, K., Kuriki, T., Kaneko, H., Shimada, J., Takada, T., Fujimoto, Z., Mizuno, H., and Okada, S. (1999). Appl. Environ. Microbiol. 65:4652–4658
Qian, M., Haser, R., and Payan, F. (1993). J. Mol. Biol. 231:758–799
Sambrook, J., and Russell, D. W. (2001) In: Molecular Cloning: A Laboratory Manual, Cold spring harbor laboratory Press, New York, pp. 1.31–1.131
Sarcabal, P., Remaud-Simeon, M., Willemot, R., Potocki de Montalk, G., Svensson, B., and Monsan, P. (2000). FEBS Lett. 474:33–37
Strokopytov, B., Knegtel, R. M. A., Penninga, D., Roozeboom, H. J., Kalk, K. H., Dijkhuizen, L., and Dijkstra, B. W. (1996). Biochemistry 35:4241–4249
Uitdehaag, J. C. M., Mosi, R., Kalk, K. H., van der Veen, B. A., Dijkhuizen, L., Withers, S. G., and Dijkstra, B. W. (1999). Nat. Struct. Biol. 6:432–436
Vihinen, M., and Mäntsälä, P. (1989). Crit. Rev. Biochem. Mol. Biol. 24:329–418
Watanabe, K., Miyake, K., and Suzuki, Y. (2001). Biosci. Biotechnol. Biochem. 65:2058–2064
Acknowledgments
This work was supported by Grants NSC 93-2313-B-241-005 and NSC 94-2313-B-241-005 from National Science Council of Taiwan, Republic of China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lin, LL., Chen, PJ., Liu, JS. et al. Identification of Glutamate Residues Important for Catalytic Activity or Thermostability of a Truncated Bacillus sp. Strain TS-23 α-amylase by Site-directed Mutagenesis. Protein J 25, 232–239 (2006). https://doi.org/10.1007/s10930-006-9006-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-006-9006-7