Abstract
Background
Mucositis, a common side effect of chemotherapy, is characterized by compromised digestive function, barrier integrity and immune competence.
Aims
Our aim was to evaluate the impact of a specifically designed diet Clinutren Protect® (CP), which contains whey proteins, TGFβ-rich casein, and free glutamine, on mucositis in rats.
Methods
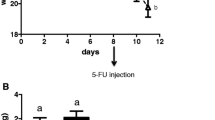
Mucositis was induced by three consecutive injections (day 0, day 1, day 2) of methotrexate (2.5 mg/kg). Rats had free access to CP or placebo diets from days −7 to 9. In the placebo diet, whey proteins and TGFβ-rich casein were replaced by TGFβ-free casein and glutamine by alanine. Intestinal parameters were assessed at day 3 and 9. Values, expressed as mean ± SEM, were compared using two-way ANOVA.
Results
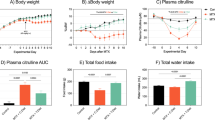
At day 3, villus height was markedly decreased in the placebo (296 ± 11 μm) and CP groups (360 ± 10 μm) compared with controls (464 ± 27 μm), but more markedly in the placebo as compared to CP group. The intestinal damage score was also reduced in the CP compared with the placebo group. Glutathione content increased in the CP compared with the placebo group (2.2 ± 0.2 vs. 1.7 ± 0.2 μmol/g tissue). Gut protein metabolism was more affected in the placebo than in the CP group. The fractional synthesis rate was decreased in the placebo group (93.8 ± 4.9%/day) compared with controls (121.5 ± 12.1, P < 0.05), but not in the CP group (106.0 ± 13.1). In addition, at day 9, rats exhibited improved body weight and food intake recovery in the CP compared to the placebo group.
Conclusions
Clinutren Protect® feeding reduces intestinal injury in the acute phase of methotrexate-induced mucositis in rats and improves recovery.







Similar content being viewed by others
Abbreviations
- BCAA:
-
branched chain amino acids
- CP:
-
Clinutren Protect®
- FSR:
-
fractional synthesis rate
- GSH:
-
glutathione
- IL:
-
interleukin
- MTX:
-
methotrexate
- PGPH:
-
peptidyl glutamyl peptide hydrolysis
- TGFβ:
-
transforming growth factor β
References
Keefe DM, Gibson RJ. Mucosal injury from targeted anti-cancer therapy. Support Care Cancer. 2007;15:483–490.
Logan RM, Stringer AM, Bowen JM, Gibson RJ, Sonis ST, Keefe DM. Serum levels of nf- kappab and pro-inflammatory cytokines following administration of mucotoxic drugs. Cancer Biol Ther. 2008;7.
Leblond J, Le Pessot F, Hubert-Buron A, et al. Chemotherapy-induced mucositis is associated with changes in proteolytic pathways. Exp Biol Med (Maywood). 2008;233:219–228.
Boukhettala N, Leblond J, Claeyssens S, et al. Methotrexate induces intestinal mucositis and alters gut protein metabolism independently of reduced food intake. Am J Physiol Endocrinol Metab. 2009;296:E182–E190.
Yoshizawa F. Regulation of protein synthesis by branched-chain amino acids in vivo. Biochem Biophys Res Commun. 2004;313:417–422.
Meister A. Glutathione, ascorbate, and cellular protection. Cancer Res. 1994;54:1969s–1975s.
Sido B, Seel C, Hochlehnert A, Breitkreutz R, Droge W. Low intestinal glutamine level and low glutaminase activity in crohn’s disease. A rational for glutamine supplementation? Dig Dis Sci. 2006;51:2170–2179.
Coeffier M, Claeyssens S, Hecketsweiler B, Lavoinne A, Ducrotte P, Dechelotte P. Enteral glutamine stimulates protein synthesis and decreases ubiquitin mrna level in human gut mucosa. Am J Physiol Gastrointest Liver Physiol. 2003;285:G266–G273.
Sukhotnik I, Khateeb K, Mogilner JG, et al. Dietary glutamine supplementation prevents mucosal injury and modulates intestinal epithelial restitution following ischemia-reperfusion injury in the rat. Dig Dis Sci. 2007;52:1497–1504.
Coeffier M, Marion R, Ducrotte P, Dechelotte P. Modulating effect of glutamine on il-1beta-induced cytokine production by human gut. Clin Nutr. 2003;22:407–413.
Xue H, Sawyer MB, Field CJ, Dieleman LA, Murray D, Baracos VE. Bolus oral glutamine protects rats against cpt-11-induced diarrhea and differentially activates cytoprotective mechanisms in host intestine but not tumour. J Nutr. 2008;138:740–746.
Daniele B, Perrone F, Gallo C, et al. Oral glutamine in the prevention of fluorouracil induced intestinal toxicity: a double blind, placebo controlled, randomised trial. Gut. 2001;48:28–33.
Braga-Neto MB, Warren CA, Oria RB, et al. Alanyl-glutamine and glutamine supplementation improves 5-fluorouracil-induced intestinal epithelium damage in vitro. Dig Dis Sci. 2008;53:2687–2696.
Li MO, Wan YY, Sanjabi S, Robertson AK, Flavell RA. Transforming growth factor-beta regulation of immune responses. Annu Rev Immunol. 2006;24:99–146.
Sukhotnik I, Mogilner JG, Shaoul R, et al. Responsiveness of intestinal epithelial cell turnover to tgf-alpha after bowel resection in a rat is correlated with egf receptor expression along the villus-crypt axis. Pediatr Surg Int. 2008;24:21–28.
Fell JM, Paintin M, Donnet-Hughes A, Arnaud-Battandier F, MacDonald TT, Walker-Smith JA. Remission induced by a new specific oral polymeric diet in children with crohn’s disease. Nestle nutr workshop ser clin perform programme. 1999;2:187–196 (discussion 196–188).
Beck PL, Rosenberg IM, Xavier RJ, Koh T, Wong JF, Podolsky DK. Transforming growth factor-beta mediates intestinal healing and susceptibility to injury in vitro and in vivo through epithelial cells. Am J Pathol. 2003;162:597–608.
Faure M, Moennoz D, Montigon F, Mettraux C, Breuille D, Ballevre O. Dietary threonine restriction specifically reduces intestinal mucin synthesis in rats. J Nutr. 2005;135:486–491.
Coeffier M, Claeyssens S, Lecleire S, et al. Combined enteral infusion of glutamine, carbohydrates, and antioxidants modulates gut protein metabolism in humans. Am J Clin Nutr. 2008;88:1284–1290.
Leblond J, Hubert-Buron A, Bole-Feysot C, Ducrotte P, Dechelotte P, Coeffier M. Regulation of proteolysis by cytokines in the human intestinal epithelial cell line hct-8: role of ifngamma. Biochimie. 2006;88:759–765.
Tran CD, Howarth GS, Coyle P, Philcox JC, Rofe AM, Butler RN. Dietary supplementation with zinc and a growth factor extract derived from bovine cheese whey improves methotrexate-damaged rat intestine. Am J Clin Nutr. 2003;77:1296–1303.
Keefe DM, Brealey J, Goland GJ, Cummins AG. Chemotherapy for cancer causes apoptosis that precedes hypoplasia in crypts of the small intestine in humans. Gut. 2000;47:632–637.
Logan RM, Gibson RJ, Bowen JM, Stringer AM, Sonis ST, Keefe DM. Characterisation of mucosal changes in the alimentary tract following administration of irinotecan: implications for the pathobiology of mucositis. Cancer Chemother Pharmacol. 2008;62:33–41.
Carneiro-Filho BA, Lima IP, Araujo DH, et al. Intestinal barrier function and secretion in methotrexate-induced rat intestinal mucositis. Dig Dis Sci. 2004;49:65–72.
Stringer AM, Gibson RJ, Logan RM, et al. Chemotherapy-induced diarrhea is associated with changes in the luminal environment in the da rat. Exp Biol Med (Maywood). 2007;232:96–106.
Tran CD, Sundar S, Howarth GS. Dietary zinc supplementation and methotrexate-induced small intestinal mucositis in metallothionein-knockout and wild-type mice. Cancer Biol Ther 2009;8.
Whitford EJ, Cummins AG, Butler RN, Prisciandaro LD, Fauser JK, Yazbeck R, Lawrence A, Cheah KY, Wright TH, Lymn KA, Howarth GS. Effects of streptococcus thermophilus th-4 on intestinal mucositis induced by the chemotherapeutic agent, 5-fluorouracil (5-fu). Cancer Biol Ther. 2009;8.
Wright TH, Yazbeck R, Lymn KA, et al. The herbal extract, iberogast, improves jejunal integrity in rats with 5-fluorouracil (5-fu)-induced mucositis. Cancer Biol Ther. 2009;8:923–929.
Menzel K, Hausmann M, Obermeier F, et al. Cathepsins b, l and d in inflammatory bowel disease macrophages and potential therapeutic effects of cathepsin inhibition in vivo. Clin Exp Immunol. 2006;146:169–180.
Bell-McGuinn KM, Garfall AL, Bogyo M, Hanahan D, Joyce JA. Inhibition of cysteine cathepsin protease activity enhances chemotherapy regimens by decreasing tumour growth and invasiveness in a mouse model of multistage cancer. Cancer Res. 2007;67:7378–7385.
Glickman MH, Ciechanover A. The ubiquitin-proteasome proteolytic pathway: destruction for the sake of construction. Physiol Rev. 2002;82:373–428.
Belmonte L, Coeffier M, Le Pessot F, et al. Effects of glutamine supplementation on gut barrier, glutathione content and acute phase response in malnourished rats during inflammatory shock. World J Gastroenterol. 2007;13:2833–2840.
Savarese DM, Savy G, Vahdat L, Wischmeyer PE, Corey B. Prevention of chemotherapy and radiation toxicity with glutamine. Cancer Treat Rev. 2003;29:501–513.
Cetiner M, Sener G, Sehirli AO, et al. Taurine protects against methotrexate-induced toxicity and inhibits leukocyte death. Toxicol Appl Pharmacol. 2005;209:39–50.
Dangin M, Boirie Y, Guillet C, Beaufrere B. Influence of the protein digestion rate on protein turnover in young and elderly subjects. J Nutr. 2002;132:3228S–3233S.
Schaafsma G. Health issues of whey proteins. Protection of lean body mass. Curr Topics Nutraceutical Res. 2006;4:113–122.
Iyer S, Wang ZG, Akhtari M, Zhao W, Seth P. Targeting tgf beta signaling for cancer therapy. Cancer Biol Ther. 2005;4:261–266.
Acknowledgments
The skillful technical assistance of Brigitte Maurer (Laboratory of Medical Biochemistry, Rouen University Hospital) is gratefully acknowledged. We also thank Dr Richard Medeiros (Medical Editor, Rouen University Hospital) for his advice in editing the manuscript.
Conflict of Interest
PD received honoraries for lectures by Nestlé Nutrition. MF, JV and DB are employees of Nestlé. None of the other authors had a conflict of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
The study was supported by Nestlé Research Center, Nutrition and Health Department, Lausanne, Switzerland.
Rights and permissions
About this article
Cite this article
Boukhettala, N., Ibrahim, A., Claeyssens, S. et al. A Diet Containing Whey Protein, Glutamine, and TGFβ Modulates Gut Protein Metabolism During Chemotherapy-Induced Mucositis in Rats. Dig Dis Sci 55, 2172–2181 (2010). https://doi.org/10.1007/s10620-009-1039-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-009-1039-2