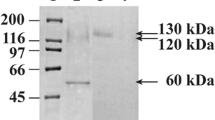
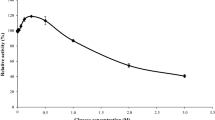
Abstract
For efficient production of isoflavone aglycones from soybean isoflavones, we isolated three novel types of β-glucosidase (BGL1, BGL3, and BGL5) from the filamentous fungi Aspergillus oryzae. Three enzymes were independently displayed on the cell surface of a yeast Saccharomyces cerevisiae as a fusion protein with α-agglutinin. Three β-glucosidase-displaying yeast strains hydrolyzed isoflavone glycosides efficiently but exhibited different substrate specificities. Among these β-glucosidases, BGL1 exhibited the highest activity and also broad substrate specificity to isoflavone glycosides. Although glucose released from isoflavone glycosides are generally known to inhibit β-glucosidase, the residual ratio of isoflavone glycosides in the reaction mixture with BGL1-displaying yeast strain (Sc-BGL1) reached approximately 6.2%, and the glucose concentration in the reaction mixture was maintained at lower level. This result indicated that Sc-BGL1 assimilated the glucose before they inhibited the hydrolysis reaction, and efficient production of isoflavone aglycones was achieved by engineered yeast cells displaying β-glucosidase.





Similar content being viewed by others
References
Abe K, Gomi K, Hasegawa F, Machida M (2006) Impact of Aspergillus oryzae genomics on industrial production of metabolites. Mycopathologia 162:143–153
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Arai Y, Watanabe S, Kimira M, Shimoi K, Mochizuki R, Kinae N (2000) Dietary intakes of flavonols, flavones and isoflavones by Japanese women and the inverse correlation between quercetin intake and plasma LDL cholesterol concentration. J Nutr 130:2243–2250
Bhatia Y, Mishra S, Bisaria VS (2005) Purification and characterization of recombinant Escherichia coli-expressed Pichia etchellsii β-glucosidase II with high hydrolytic activity on sophorose. Appl Microbiol Biotechnol 66:527–535
Chiou RYY, Cheng SL (2001) Isoflavone Transformation during soybean koji preparation and subsequent miso fermentation supplemented with ethanol and NaCl. J Agric Food Chem 49:3656–3660
Decker CH, Visser J, Schreier P (2001) Beta-glucosidase multiplicity from Aspergillus tubingensis CBS 643.92: purification and characterization of four beta-glucosidases and their differentiation with respect to substrate specificity, glucose inhibition and acid tolerance. Appl Microbiol Biotechnol 55:157–163
Fujita Y, Ito J, Ueda M, Fukuda H, Kondo A (2004) Synergistic saccharification, and direct fermentation to ethanol, of amorphous cellulose by use of an engineered yeast strain codisplaying three types of cellulolytic enzyme. Appl Environ Microbiol 70:1207–1212
Hoh YK, Yeoh HH, Tan TK (1992) Properties of β-glucosidase purified from Aspergillus niger mutants USDB 0827 and USDB 0828. Appl Microbiol Biotechnol 37:590–593
Izumi T, Piskula MK, Osawa S (2000) Soy isoflavone aglycones are absorbed faster and in higher amounts than their glucosides in humans. J Nutr 130:1695–1699
Kitamoto N, Go M, Shibayama T, Kimura T, Kito Y, Ohmiya K, Tsukagoshi N (1996) Molecular cloning, purification and characterization of two endo-1,4-beta-glucanases from Aspergillus oryzae KBN616. Appl Microbiol Biotechnol 46:538–544
Kondo A, Ueda M (2004) Yeast cell-surface display—applications of molecular display. Appl Microbiol Biotechnol 64:28–40
Langston J, Sheehy N, Xu F (2006) Substrate specificity of Aspergillus oryzae family 3 beta-glucosidase. Biochim Biophys Acta 1764:972–978
Machida M, Asai K, Sano M, Tanaka T, Kumagai T, Terai G, Kusumoto K, Arima T, Akita O, Kashiwagi Y, Abe K, Gomi K, Horiuchi H, Kitamoto K, Kobayashi T, Takeuchi M, Denning DW, Galagan JE, Nierman WC, Yu J, Archer DB, Bennett JW, Bhatnagar D, Cleveland TE, Fedorova ND, Gotoh O, Horikawa H, Hosoyama A, Ichinomiya M, Igarashi R, Iwashita K, Juvvadi PR, Kato M, Kato Y, Kin T, Kokubun A, Maeda H, Maeyama N, Maruyama J, Nagasaki H, Nakajima T, Oda K, Okada K, Paulsen I, Sakamoto K, Sawano T, Takahashi M, Takase K, Terabayashi Y, Wortman JR, Yamada O, Yamagata Y, Anazawa H, Hata Y, Koide Y, Komori T, Koyama Y, Minetoki T, Suharnan S, Tanaka A, Isono K, Kuhara S, Ogasawara N, Kikuchi H (2005) Genome sequencing and analysis of Aspergillus oryzae. Nature 438:1157–1161
Matsumura K, Obata H, Hata Y, Kawato A, Abe Y, Akita O (2004) Isolation and characterization of a novel gene encoding alpha-L-arabinofuranosidase from Aspergillus oryzae. J Biosci Bioeng 98:77–84
Messina M, Messina V (1991) Increasing use of soyfoods and their potential role in cancer prevention. Perspec Prac 91:836–840
Murai T, Ueda M, Kawaguchi T, Arai M, Tanaka A (1998) Assimilation of cellooligosaccharides by a cell-surface-engineered yeast expressing beta-glucosidase and carboxymethylcellulase from aspergillus aculeatus. Appl Environ Microbiol 64:4857–4861
Odoux E, Chauwin A, Brillouet JM (2003) Purification and characterization of Vanilla Bean (Vanilla planifolia Andrews) b-D-glucosidase. J Agric Food Chem 57:3168–3173
Riou C, Salmon JM, Vallier MJ, Gunata Z, Barre P (1998) Purification, characterization, and substrate specificity of a novel highly glucose-tolerant beta-glucosidase from Aspergillus oryzae. Appl Environ Microbiol 64:3607–3614
Sakamoto R, Arai M, Murao S (1985) Enzymatic properties of three b-glucosidases from Aspergillus aculeatus No. F-50. Agric Biol Chem 49:1283–1290
Schreuder MP, Mooren AT, Toschka HY, Verrips CT, Klis FM (1996) Immobilizing proteins on the surface of yeast cells. Trends Biotechnol 14:115–120
Setchell KDR, Borriello SP, Hulme P, Kirk DN (1984) Nonsteroidal estrogens of dietary origin: possible roles in hormone-dependent disease. Am J Clin Nutr 40:569–578
Song T, Barua K, Buseman G, Murphy PA (1998) Soy isoflavone analysis: quality control and a new internal standard. Am J Clin Nutr 68:1474–1479
Tanino T, Noguchi E, Kimura S, Sahara H, Hata Y, Fukuda H, Kondo A (2007) Effect of cultivation conditions on cell-surface display of Flo1 fusion protein using sake yeast. Biochem Eng J 33:232–237
Ueda M, Tanaka A (2000) Genetic immobilization of proteins on the yeast cell surface. Biotechnol Adv 18:121–140
Yang L, Ning ZS, Shi CZ, Chang ZY, Huan LY (2004) Purification and characterization of an isoflavone-conjugates-hydrolyzing β-glucosidase from endophytic bacterium. J Agric Food Chem 52:1940–1944
Yun SI, Jeong CS, Chung DK, Choi HS (2001) Purification and some properties of a beta-glucosidase from Trichoderma harzianum type C-4. Biosci Biotechnol Biochem 65:2028–2032
Acknowledgement
This work was financed by Research and Development Program for New Bio-industry Initiatives.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaya, M., Ito, J., Kotaka, A. et al. Isoflavone aglycones production from isoflavone glycosides by display of β-glucosidase from Aspergillus oryzae on yeast cell surface. Appl Microbiol Biotechnol 79, 51–60 (2008). https://doi.org/10.1007/s00253-008-1393-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-008-1393-6