Abstract
Sirolimus (previously known as rapamycin), a macrocyclic lactone, is a potent immunosuppressive agent. Sirolimus was recently approved by the US Food and Drug Administration, on the basis of 2 large, double-blind, prospective clinical trials, for use in kidney transplant recipients at a fixed dosage of 2 or 5 mg/day in addition to full dosages of cyclosporin and prednisone. However, despite the fixed dosage regimens used in these pivotal trials, pharmacokinetic and clinical data show that sirolimus is a critical-dose drug requiring therapeutic drug monitoring to minimise drug-related toxicities and maximise efficacy.
Assays using high performance liquid chromatography coupled to mass spectrometry, although cumbersome, are the gold standard for evaluating other commonly used assays, such as liquid chromatography with ultraviolet detection, radioreceptor assay and microparticle enzyme immunoassay.
Sirolimus is available in oral solution and tablet form. It has poor oral absorption and distributes widely in tissues, displaying not only a wide inter- and intrapatient variability in drug clearance, but also less than optimal correlations between whole blood concentrations and drug dose, demographic features or patient characteristics. Furthermore, the critical role of the cytochrome P450 3A4 system for sirolimus biotransformation leads to extensive drug-drug interactions, among which are increases in cyclosporin concentrations. Thus, sirolimus is now being used to reduce or eliminate exposure to cyclosporin or corticosteroids. The long elimination half-life of sirolimus necessitates a loading dose but allows once daily administration, which is more convenient and thereby could help to improve patient compliance.
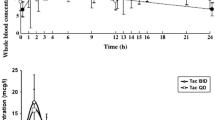
This review emphasises the importance of blood concentration monitoring in optimising the use of sirolimus. The excellent correlation between steady-state trough concentration (at least 4 days after inception of, or change in, therapy) and area under the concentration-time curve makes the former a simple and reliable index for monitoring sirolimus exposure. Target trough concentration ranges depend on the concomitant immunosuppressive regimen, but a range of 5 to 15 μg/L is appropriate if cyclosporin is being used at trough concentrations of 75 to 150 μg/L. Weekly monitoring is recommended for the first month and bi-weekly for the next month; thereafter, concentration measurements are necessary only if warranted clinically.









Similar content being viewed by others
References
Sehgal SN, Baker H, Vezina C. Rapamycin (AY-22,989), a new antifungal antibiotic: II. Fermentation, isolation and characterization. J Antibiot (Tokyo) 1975; 28: 727–32
Calne RY. The rejection of renal homograft inhibition in dogs by 6-mercaptopurine. Lancet 1960; I: 417–8
Kahan BD, Chang JY, Sehgal SN. Preclinical evaluation of a new potent immunosuppressive agent, rapamycin. Transplantation 1991; 52: 185–91
Kahan BD, Gibbons S, Tejpal N, et al. Synergistic interactions of cyclosporine and rapamycin to inhibit immune performances of normal human peripheral blood lymphocytes in vitro. Transplantation 1991; 51: 232–9
Stepkowski SM, Napoli KL, Wang ME, et al. Effects of the pharmacokinetic interaction between orally administered sirolimus and cyclosporine on the synergistic prolongation ofheart allograft survival in rats. Transplantation 1996; 62: 986–94
Dumont FJ, Staruch MJ, Koprak SL, et al. Distinct mechanisms of suppression of murine T cell activation by the related mac-rolides FK-506 and rapamycin. J Immunol 1990; 144: 251–8
Salm P, Tresillian MJ, Taylor PJ, et al. Stability of sirolimus (rapamycin) in whole blood. Ther Drug Monit 2000; 22: 423–6
Granger DK, Cromwell JW, Chen SC, et al. Prolongation of renal allograft survival in a large animal model by oral rapamycin monotherapy. Transplantation 1995; 59: 183–6
Kahan BD. Established immunosuppressive drugs: clinical and toxic effects. In: Kahan BD, Ponticelli C, editors. Principles and practice of renal transplantation. London: Dunitz, 2000: 349–414
McAlister VC, Gao Z, Peltekian K, et al. Sirolimus-tacrolimus combination immunosuppression [letter]. Lancet 2000; 355: 376–7
Christians U, Sattler M, Schiebel HM, et al. Isolation of two immunosuppressive metabolites after in vitro metabolism of rapamycin. Drug Metab Dispos 1992; 20: 186–91
Yatscoff RW, Wang P, Chan K, et al. Rapamycin: distribution, pharmacokinetics, and therapeutic range investigations. Ther Drug Monit 1995; 17: 666–71
Yatscoff R, LeGatt D, Keenan R, et al. Blood distribution of rapamycin. Transplantation 1993; 56: 1202–6
Streit F, Christians U, Schiebel HM, et al. Sensitive and specific quantification of sirolimus (rapamycin) and its metabolites in blood of kidney graft recipients by HPLC/electrospray-mass spectrometry. Clin Chem 1996; 42: 1417–25
Taylor PJ, Johnson AG. Quantitative analysis of sirolimus (Rapamycin) in blood by high-performance liquid chromatography-electrospray tandem mass spectrometry. J Chro- matogr B Biomed Sci Appl 1998; 718: 251–7
Napoli KL, Kahan BD. Sample clean-up and high-performance liquid chromatographic techniques for measurement of whole blood rapamycin concentrations. J Chromatogr B Biomed Appl 1994; 654: 111–20
Davis DL, Murthy JN, Napoli KL, et al. Comparison of steady-state trough sirolimus samples by HPLC and a radioreceptor assay. Clin Biochem 2000; 33: 31–6
Salm P, Taylor PJ, Pillans PI. Analytical performance of microparticle enzyme immunoassay and HPLC-tandem mass spectrometry in the determination of sirolimus in whole blood. Clin Chem 1999; 45: 2278–80
Svensson JO, Brattström C, Säwe J. Determination of rapamycin in whole blood by HPLC. Ther Drug Monit 1997; 19: 112–6
Kahan BD, Napoli KL, Kelly PA, et al. Therapeutic drug monitoring of sirolimus: correlations with efficacy and toxicity. Clin Transplant 2000; 14: 97–109
Holt DW, Lee T, Johnston A. Measurement of sirolimus in whole blood using high-performance liquid chromatography with ultraviolet detection. Clin Ther 2000; 22 Suppl. B: B38–48
Napoli KL, Kahan BD. Routine clinical monitoring of sirolimus (rapamycin) whole-blood concentrations by HPLC with ultraviolet detection. Clin Chem 1996; 42: 1943–8
Goodyear N, Murthy JN, Gallant HL, et al. Comparison of binding characteristics offour rapamycin metabolites to the 14 and 52 kDa immunophilins with their pharmacologic activity measured by the mixed-lymphocyte culture assay. Clin Biochem 1996; 29: 309–13
Davis DL, Murthy JN, Gallant-Haidner H, et al. Minor immunophilin binding of tacrolimus and sirolimus metabolites. Clin Biochem 2000; 33: 1–6
Goodyear N, Napoli KL, Murthy JN, et al. Radioreceptor assay for sirolimus in patients with decreased platelet counts. Clin Biochem 1997; 30: 539–43
Salm P, Taylor PJ, Pillans PI. The quantification ofsirolimus by high-performance liquid chromatography-tandem mass spectrometry and microparticle enzyme immunoassay in renal transplant recipients. Clin Ther 2000; 22 Suppl. B: B71–85
Brattstrom C, Sawe J, Tyden G, et al. Kinetics and dynamics of single oral doses of sirolimus in sixteen renal transplant recipients. Ther Drug Monit 1997; 19: 397–406
Zimmerman JJ, Ferron GM, Lim H-K, et al. The effect of a high-fat meal on the oral bioavailability of the immunosuppressant sirolimus (rapamycin). J Clin Pharmacol 1999; 39: 1155–61
Johnson EM, Zimmerman J, Duderstadt K, et al. A randomized, double-blind, placebo-controlled study of the safety, tolerance, and preliminary pharmacokinetics of ascending single doses of orally administered sirolimus (rapamycin) in stable renal transplant recipients. Transplant Proc 1996; 28: 987
Yatscoff RW. Pharmacokinetics of rapamycin. Transplant Proc 1996; 28: 970–3
Zimmerman J, Kahan BD. Pharmacokinetics of sirolimus in stable renal transplant patients after multiple oral dose administration. J Clin Pharmacol 1997; 37: 405–15
Ferron GM, Mishina EV, Zimmerman JJ, et al. Population pharmacokinetics of sirolimus in kidney transplant patients. Clin Pharmacol Ther 1997; 61: 416–28
Van Buren CT, Rapamune Study Group. Sirolimus oral solution and tablets demonstrate equivalent safety and efficacy in renal allografts [abstract no. 157]. American Society of Transplantation/American Society of Transplant Surgeons Joint Meeting; 2000 May 13-17; Chicago. Transplantation 2000; 69 Suppl.: S153
Kelly P, Napoli KL, Kahan BD. Comparison of the pharmacokinetics of sirolimus (Rapamune) in renal transplant recipients following administration of the liquid or solid table formations [abstract no. 158]. Transplantation 2000; 69: S154
Kelly PA, Napoli KL, Dunne C, et al. Conversion from liquid to solid sirolimus formulations in stable renal allograft transplant recipients. Biopharm Drug Dispos 1999; 20: 249–53
Zimmerman J, Rapamune Study Group. A comparative study of the pharmacokinetic profiles of sirolimus oral solution and tablets in renal allograft patients [abstract no. 159]. Transplantation 2000; 69: S154
Kaplan B, Meier-Kriesche HU, Napoli KL, et al. The effects of relative timing of sirolimus and cyclosporine microemulsion formulation co-administration on the pharmacokinetics of each agent. Clin Pharmacol Ther 1998; 63: 48–53
Yacyshyn BR, Bowen-Yacyshyn MB, Pilarski LM. Inhibition by rapamycin of P-glycoprotein 170-mediated export from normal lymphocytes. Scand J Immunol 1996; 43: 449–55
Kay JE, Sampare-Kwateng E, Geraghty F, et al. Uptake of FK 506 by lymphocytes and erythrocytes. Transplant Proc 1991; 23: 2760–2
Hoogeveen RC, Ballantyne CM, Pownall HJ, et al. Effect of sirolimus on the metabolism of ApoB-100 containing lipoproteins in renal transplant patients. Transplantation. In press
Napoli KL, Wang ME, Stepkowski SM, et al. Distribution of sirolimus in rat tissue. Clin Biochem 1997; 30: 135–42
Lown KS, Mayo RR, Leichtman AB, et al. Role of intestinal P-glycoprotein (mdr1) in interpatient variation in the oral bioavailability of cyclosporine. Clin Pharmacol Ther 1997; 62: 248–60
Lampen A, Zhang Y, Hackbarth I, et al. Metabolism and transport of the macrolide immunosuppressant sirolimus in the small intestine. J Pharmacol Exp Ther 1998; 285: 1104–12
Sattler M, Guengerich FP, Yun CH, et al. Cytochrome P-450 3A enzymes are responsible for biotransformation of FK506 and rapamycin in man and rat. Drug Metab Dispos 1992; 20: 753–61
Crowe A, Lemaire M. In vitro and in situ absorption of SDZ-RAD using a human intestinal cell line (Caco-2) and a single pass perfusion model in rats: comparison with rapamycin. Pharm Res 1998; 15: 1666–72
MacDonald A, Scarola J, Burke JT, et al. Clinical pharmacokinetics and therapeutic drug monitoring of sirolimus. Clin Ther 2000; 22 Suppl. B: B101–21
Wyeth Laboratories. Sirolimus (Rapamune®): prescribing in formation. Philadelphia (PA): Wyeth-Ayerst Pharmaceuticals Inc., 2001
Trepanier DJ, Gallant H, Legatt DF, et al. Rapamycin: distribution, pharmacokinetics and therapeutic range investigations: an update. Clin Biochem 1998; 31: 345–51
Wang CP, Chan KW, Schiksnis RA, et al. High performance liquid chromatographic isolation, spectroscopic characterization and immunosuppressive activities of two rapamycin degradation products. J Liq Chromatogr 1994; 17: 3383–92
Wang CP, Lim HK, Chan KW, et al. High performance liquid chromatographic isolation and spectroscopic characterization of three major metabolites from the plasma of rats receiving rapamycin (sirolimus) orally. J Liq Chromatogr 1995; 18: 2259
Napoli KL, Wang ME, Stepkowski SM, et al. Relative tissue distributions of cyclosporine and sirolimus after concomitant peroral administration to the rat: evidence for pharmacokinetic interactions. Ther Drug Monit 1998; 20: 123–33
Kahan BD, Rapamune U.S. Study Group. Sirolimus (Rapamune, rapamycin) is more effective than azathioprine to reduce the incidence of acute renal allograft rejection episodes when used in combination with cyclosporine and prednisone: a phase III U.S. multicenter trial. Lancet 2000; 356: 194–202
Jusko WJ, Ferron GM, Mis SM, et al. Pharmacokinetics of prednisolone during administration of sirolimus in patients with renal transplants. J Clin Pharmacol 1996; 36: 1100–6
Fryer J, Yatscoff RW, Pascoe EA, et al. The relationship of blood concentrations of rapamycin and cyclosporine to suppression of allograft rejection in a rabbit heterotopic heart transplant model. Transplantation 1993; 55: 340–5
Kahan BD, Podbielski J, Napoli KL, et al. Immunosuppressive effects and safety of a sirolimus/cyclosporine combination regimen for renal transplantation. Transplantation 1998; 66:1040–6
Kahan BD, Julian BA, Pescovitz MD, et al. Sirolimus reduces the incidence of acute rejection episodes despite lower cyclosporine doses in Caucasian recipients of mismatched primary renal allografts: a phase II trial. Transplantation 1999; 68: 1526–32
Groth CG, Backman L, Morales JM, et al., Sirolimus European Renal Transplant Study Group. Sirolimus (rapamycin)-based therapy in human renal transplantation: similar efficacy and different toxicity compared with cyclosporine. Transplantation 1999; 67: 1036–42
Kreis H, Cisterne JM, Land W, et al. Sirolimus in association with mycophenolate mofetil induction for the prevention of acute graft rejection in renal allograft recipients. Transplantation 2000; 69: 1252–60
Hong JC, Kahan BD. Use of anti-CD25 monoclonal antibody in combination with rapamycin to eliminate cyclosporine treatment during the induction phase of immunosuppression [brief communication]. Transplantation 1999; 68: 701–4
Hong JC, Kahan BD. A calcineurin-free strategy for induction immunosuppression for delayed graft function in cadaveric kidney transplantation. Transplant Proc 2001; 33: 1271–2
Acknowledgements
This work was supported by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK 38016-14).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mahalati, K., Kahan, B.D. Clinical Pharmacokinetics of Sirolimus. Clin Pharmacokinet 40, 573–585 (2001). https://doi.org/10.2165/00003088-200140080-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200140080-00002