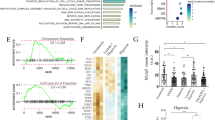
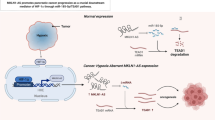
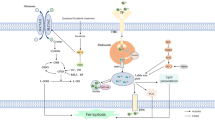
Abstract
Hypoxia, as an important hallmark of the tumor microenvironment, is a major cause of oxidative stress and plays a central role in various malignant tumors, including glioblastoma. Elevated reactive oxygen species (ROS) in a hypoxic microenvironment promote glioblastoma progression; however, the underlying mechanism has not been clarified. Herein, we found that hypoxia promoted ROS production, and the proliferation, migration, and invasion of glioblastoma cells, while this promotion was restrained by ROS scavengers N-acetyl-l-cysteine (NAC) and diphenyleneiodonium chloride (DPI). Hypoxia-induced ROS activated hypoxia-inducible factor-1α (HIF-1α) signaling, which enhanced cell migration and invasion by epithelial-mesenchymal transition (EMT). Furthermore, the induction of serine protease inhibitor family E member 1 (SERPINE1) was ROS-dependent under hypoxia, and HIF-1α mediated SERPINE1 increase induced by ROS via binding to the SERPINE1 promoter region, thereby facilitating glioblastoma migration and invasion. Taken together, our data revealed that hypoxia-induced ROS reinforce the hypoxic adaptation of glioblastoma by driving the HIF-1α-SERPINE1 signaling pathway, and that targeting ROS may be a promising therapeutic strategy for glioblastoma.
概要
缺氧作为肿瘤微环境的重要特征,是氧化应激的主要原因,在肿瘤(包括胶质母细胞瘤)的恶性进展中发挥重要作用。缺氧微环境中高水平活性氧(ROS)促进胶质母细胞瘤进展的潜在机制尚不清楚。本研究发现缺氧促进了胶质母细胞瘤细胞增殖、迁移和侵袭以及ROS生成,而这种促进作用可被ROS清除剂N-乙酰半胱氨酸(NAC)和二苯基氯化碘盐(DPI)抑制。缺氧诱导的ROS可激活缺氧诱导因子-1α(HIF-1α)信号,通过上皮-间充质转化(EMT)增强细胞迁移和侵袭。此外,在缺氧条件下,HIF-1α可与丝氨酸蛋白酶抑制剂家族E成员1(SERPINE1)启动子区结合,ROS经HIF-1α上调SERPINE1表达,进而促进胶质母细胞瘤细胞迁移和侵袭。综上所述,本研究揭示了缺氧诱导的ROS通过驱动HIF-1α-SERPINE1信号促进胶质母细胞瘤的缺氧适应,靶向ROS可能成为胶质母细胞瘤治疗的一种有效策略。
Similar content being viewed by others
References
Andrew AS, Klei LR, Barchowsky A, 2001. Nickel requires hypoxia-inducible factor-1α, not redox signaling, to induce plasminogen activator inhibitor-1. Am J Physiol Lung Cell Mol Physiol, 281(3):L607–L615. https://doi.org/10.1152/ajplung.2001.281.3.L607
Azimi I, Petersen RM, Thompson EW, et al., 2017. Hypoxia-induced reactive oxygen species mediate N-cadherin and SERPINE1 expression, EGFR signalling and motility in MDA-MB-468 breast cancer cells. Sci Rep, 7:15140. https://doi.org/10.1038/s41598-017-15474-7
Carreres L, Mercey-Ressejac M, Kurma K, et al., 2022. Chronic intermittent hypoxia increases cell proliferation in hepatocellular carcinoma. Cells, 11(13):2051. https://doi.org/10.3390/cells11132051
Chandel NS, Maltepe E, Goldwasser E, et al., 1998. Mitochondrial reactive oxygen species trigger hypoxia-induced transcription. Proc Natl Acad Sci USA, 95(20):11715–11720. https://doi.org/10.1073/pnas.95.20.11715
Chandel NS, McClintock DS, Feliciano CE, et al., 2000. Reactive oxygen species generated at mitochondrial complex III stabilize hypoxia-inducible factor-1α during hypoxia: a mechanism of O2 sensing. J Biol Chem, 275(33):25130–25138. https://doi.org/10.1074/jbc.M001914200
Chédeville AL, Lourdusamy A, Monteiro AR, et al., 2020. Investigating glioblastoma response to hypoxia. Biomedicines, 8(9):310. https://doi.org/10.3390/biomedicines8090310
Chen CL, Wang SC, Liu P, 2019. Deferoxamine enhanced mitochondrial iron accumulation and promoted cell migration in triple-negative MDA-MB-231 breast cancer cells via a ROS-dependent mechanism. Int J Mol Sci, 20(19):4852. https://doi.org/10.3390/ijms20194952
Chen D, Wu YX, Qiu YB, et al., 2020. Hyperoside suppresses hypoxia-induced A549 survival and proliferation through ferrous accumulation via AMPK/HO-1 axis. Phytomedicine, 67:153138. https://doi.org/10.1016/j.phymed.2019.153138
Chen XT, Li ZW, Yong HM, et al., 2021. Trim21-mediated HIF-1α degradation attenuates aerobic glycolysis to inhibit renal cancer tumorigenesis and metastasis. Cancer Lett, 508:115–126. https://doi.org/10.1016/j.canlet.2021.03.023
Chiu J, Dawes IW, 2012. Redox control of cell proliferation. Trends Cell Biol, 22(11):592–601. https://doi.org/10.1016/j.tcb.2012.08.002
Chua YL, Dufour E, Dassa EP, et al., 2010. Stabilization of hypoxia-inducible factor-1α protein in hypoxia occurs independently of mitochondrial reactive oxygen species production. J Biol Chem, 285(41):31277–31284. https://doi.org/10.1074/jbc.M110.158485
Dabral S, Muecke C, Valasarajan C, et al., 2019. A RASSF1A-HIF1α loop drives Warburg effect in cancer and pulmonary hypertension. Nat Commun, 10:2130. https://doi.org/10.1038/s41467-019-10044-z
Dao Trong P, Rösch S, Mairbäurl H, et al., 2018. Identification of a prognostic hypoxia-associated gene set in IDH-mutant glioma. Int J Mol Sci, 19(10):2903. https://doi.org/10.3390/ijms19102903
Dröse S, 2013. Differential effects of complex II on mitochondrial ROS production and their relation to cardioprotective pre- and postconditioning. Biochim Biophys Acta Bioenerget, 1827(5):578–587. https://doi.org/10.1016/j.bbabio.2013.01.004
Fandrey J, Gassmann M, 2009. Oxygen sensing and the activation of the hypoxia inducible factor 1 (HIF-1)-Invited Article. In: Gonzalez C, Nurse CA, Peers C (Eds.), Arterial Chemoreceptors. Advances in Experimental Medicine and Biology, Vol. 648. Springer, Dordrecht, p.197–206. https://doi.org/10.1007/978-90-481-2259-2_23
Fink T, Kazlauskas A, Poellinger L, et al., 2002. Identification of a tightly regulated hypoxia-response element in the promoter of human plasminogen activator inhibitor-1. Blood, 99(6):2077–2083. https://doi.org/10.1182/blood.v99.6.2077
Ge WJ, Zhao KM, Wang XW, et al., 2017. iASPP is an antioxidative factor and drives cancer growth and drug resistance by competing with Nrf2 for Keap1 binding. Cancer Cell, 32(5):561–573.6. https://doi.org/10.1016/j.ccell.2017.09.008
Harris AL, 2002. Hypoxia—a key regulatory factor in tumour growth. Nat Rev Cancer, 2(1):38–47. https://doi.org/10.1038/nrc704
Hou P, Zhao Y, Li Z, et al., 2014. LincRNA-ROR induces epithelial-to-mesenchymal transition and contributes to breast cancer tumorigenesis and metastasis. Cell Death Dis, 5(6):e1287. https://doi.org/10.1038/cddis.2014.249
Hu ZZ, Dong N, Lu D, et al., 2017. A positive feedback loop between ROS and Mxi1-0 promotes hypoxia-induced VEGF expression in human hepatocellular carcinoma cells. Cell Signal, 31:79–86. https://doi.org/10.1016/j.cellsig.2017.01.007
Humphries BA, Buschhaus JM, Chen YC, et al., 2019. Plasminogen activator inhibitor 1 (PAI1) promotes actin cytoskeleton reorganization and glycolytic metabolism in triple-negative breast cancer. Mol Cancer Res, 17(5):1142–1154. https://doi.org/10.1158/1541-7786.MCR-18-0836
Hurd TR, DeGennaro M, Lehmann R, 2012. Redox regulation of cell migration and adhesion. Trends Cell Biol, 22(2):107–115. https://doi.org/10.1016/j.tcb.2011.11.002
Ivan M, Kaelin WG, 2017. The EGLN-HIF O2-sensing system: multiple inputs and feedbacks. Mol Cell, 66(6):772–779. https://doi.org/10.1016/j.molcel.2017.06.002
Jin P, Shin SH, Chun YS, et al., 2018. Astrocyte-derived CCL20 reinforces HIF-1-mediated hypoxic responses in glioblastoma by stimulating the CCR6-NF-κB signaling pathway. Oncogene, 37(23):3070–3087. https://doi.org/10.1038/s41388-018-0182-7
Ke QD, Costa M, 2006. Hypoxia-inducible factor-1 (HIF-1). Mol Pharmacol, 70(5):1469–1480. https://doi.org/10.1124/mol.106.027029
Kessler J, Hahnel A, Wichmann H, et al., 2010. HIF-1α inhibition by siRNA or chetomin in human malignant glioma cells: effects on hypoxic radioresistance and monitoring via CA9 expression. BMC Cancer, 10:605. https://doi.org/10.1186/1471-2407-10-605
Klimova T, Chandel NS, 2008. Mitochondrial complex III regulates hypoxic activation of HIF. Cell Death Differ, 15(4):660–666. https://doi.org/10.1038/sj.cdd.4402307
Kunsch C, Medford RM, 1999. Oxidative stress as a regulator of gene expression in the vasculature. Circ Res, 85(8): 753–766. https://doi.org/10.1161/01.res.85.8.753
Lei JJ, Huo XW, Duan WX, et al., 2014. α-Mangostin inhibits hypoxia-driven ROS-induced PSC activation and pancreatic cancer cell invasion. Cancer Lett, 347(1):129–138. https://doi.org/10.1016/j.canlet.2014.02.003
Li SJ, Wei XH, Zhan XM, et al., 2020. Adipocyte-derived leptin promotes PAI-1-mediated breast cancer metastasis in a STAT3/miR-34a dependent manner. Cancers (Basel), 12(12):3864. https://doi.org/10.3390/cancers12123864
Li X, Wu P, Tang YY, et al., 2020. Down-regulation of miR-181c-5p promotes epithelial-to-mesenchymal transition in laryngeal squamous cell carcinoma via targeting SERPINE1. Front Oncol, 10:544476. https://doi.org/10.3389/fonc.2020.544476
Li X, Zuo HW, Zhang L, et al., 2021. Validating HMMR expression and its prognostic significance in lung adenocarcinoma based on data mining and bioinformatics methods. Front Oncol, 11:720302. https://doi.org/10.3389/fonc.2021.720302
Li XD, Dong P, Wei WS, et al., 2019. Overexpression of CEP72 promotes bladder urothelial carcinoma cell aggressiveness via epigenetic CREB-mediated induction of SERPINE1. Am J Pathol, 189(6):1284–1297. https://doi.org/10.1016/j.ajpath.2019.02.014
Li ZW, Hou PF, Fan DM, et al., 2017. The degradation of EZH2 mediated by lncRNA ANCR attenuated the invasion and metastasis of breast cancer. Cell Death Differ, 24(1):59–71. https://doi.org/10.1038/cdd.2016.95
Liu L, Xiao S, Wang Y, et al., 2022. Identification of a novel circular RNA circZNF652/miR-486-5p/SERPINE1 signaling cascade that regulates cancer aggressiveness in glioblastoma (GBM). Bioengineered, 13(1):1411–1423. https://doi.org/10.1080/21655979.2021.2018096
Lu W, Kang YB, 2019. Epithelial-mesenchymal plasticity in cancer progression and metastasis. Dev Cell, 49(3):361–374. https://doi.org/10.1016/j.devcel.2019.04.010
Lukyanova LD, Kirova YI, Germanova EL, 2018. The role of succinate in regulation of immediate HIF-1α expression in hypoxia. Bull Exp Biol Med, 164(3):298–303. https://doi.org/10.1007/s10517-018-3976-2
Mittal M, Gu XQ, Pak O, et al., 2012. Hypoxia induces Kv channel current inhibition by increased NADPH oxidase-derived reactive oxygen species. Free Radic Biol Med, 52(6):1033–1042. https://doi.org/10.1016/j.freeradbiomed.2011.12.004
Miyashita-Ishiwata M, el Sabeh M, Reschke LD, et al., 2022. Hypoxia induces proliferation via NOX4-mediated oxidative stress and TGF-β3 signaling in uterine leiomyoma cells. Free Radic Res, 56(2):163–172. https://doi.org/10.1080/10715762.2022.2061967
Monteiro AR, Hill R, Pilkington GJ, et al., 2017. The role of hypoxia in glioblastoma invasion. Cells, 6(4):45. https://doi.org/10.3390/cells6040045
Niecknig H, Tug S, Reyes BD, et al., 2012. Role of reactive oxygen species in the regulation of HIF-1 by prolyl hydroxylase 2 under mild hypoxia. Free Radic Res, 46(6):705–717. https://doi.org/10.3109/10715762.2012.669041
Olar A, Aldape KD, 2014. Using the molecular classification of glioblastoma to inform personalized treatment. J Pathol, 232(2):165–177. https://doi.org/10.1002/path.4282
Omuro A, DeAngelis LM, 2013. Glioblastoma and other malignant gliomas: a clinical review. JAMA, 310(17):1842–1850. https://doi.org/10.1001/jama.2013.280319
Peng PH, Lai JCY, Hsu KW, et al., 2020. Hypoxia-induced lncRNA RP11-390F4.3 promotes epithelial-mesenchymal transition (EMT) and metastasis through upregulating EMT regulators. Cancer Lett, 483:35–45. https://doi.org/10.1016/j.canlet.2020.04.014
Perillo B, di Donato M, Pezone A, et al., 2020. ROS in cancer therapy: the bright side of the moon. Exp Mol Med, 52(2): 192–203. https://doi.org/10.1038/s12276-020-0384-2
Polyak K, Weinberg RA, 2009. Transitions between epithelial and mesenchymal states: acquisition of malignant and stem cell traits. Nat Rev Cancer, 9(4):265–273. https://doi.org/10.1038/nrc2620
Quinlan CL, Orr AL, Perevoshchikova IV, et al., 2012. Mitochondrial complex II can generate reactive oxygen species at high rates in both the forward and reverse reactions. J Biol Chem, 287(32):27255–27264. https://doi.org/10.1074/jbc.M112.374629
Schieber M, Chandel NS, 2014. ROS function in redox signaling and oxidative stress. Curr Biol, 24(10):R453–R462. https://doi.org/10.1016/j.cub.2014.03.034
Schumacker PT, 2011. SIRT3 controls cancer metabolic reprogramming by regulating ROS and HIF. Cancer Cell, 19(3): 299–300. https://doi.org/10.1016/j.ccr.2011.03.001
Semenza GL, 1998. Hypoxia-inducible factor 1: master regulator of O2 homeostasis. Curr Opin Genet Dev, 8(5):588–594. https://doi.org/10.1016/s0959-437x(98)80016-6
Semenza GL, 2014. Oxygen sensing, hypoxia-inducible factors, and disease pathophysiology. Annu Rev Pathol Mech Dis, 9:47–71. https://doi.org/10.1146/annurev-pathol-012513-104720
Spencer NY, Engelhardt JF, 2014. The basic biology of redoxosomes in cytokine-mediated signal transduction and implications for disease-specific therapies. Biochemistry, 53(10):1551–1564. https://doi.org/10.1021/bi401719r
Srivastava C, Irshad K, Dikshit B, et al., 2018. FAT1 modulates EMT and stemness genes expression in hypoxic glioblastoma. Int J Cancer, 142(4):805–812. https://doi.org/10.1002/ijc.31092
Subramanian A, Tamayo P, Mootha VK, et al., 2005. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA, 102(43):15545–15550. https://doi.org/10.1073/pnas.0506580102
Sun B, Yu L, Xu C, et al., 2021. NAD(P)HX epimerase down-regulation promotes tumor progression through ROS/HIF-1α signaling in hepatocellular carcinoma. Cancer Sci, 112(7):2753–2769. https://doi.org/10.1111/cas.14925
Takayama Y, Hattori N, Hamada H, et al., 2016. Inhibition of PAI-1 limits tumor angiogenesis regardless of angiogenic stimuli in malignant pleural mesothelioma. Cancer Res, 76(11):3285–3294. https://doi.org/10.1158/0008-5472.CAN-15-1796
Tang ZH, Zhang ZH, Lin QQ, et al., 2021. HIF-1α/BNIP3-mediated autophagy contributes to the luteinization of granulosa cells during the formation of corpus luteum. Front Cell Dev Biol, 8:619924. https://doi.org/10.3389/fcell.2020.619924
Teng F, Zhang JX, Chen Y, et al., 2021. LncRNA NKX2-1-AS1 promotes tumor progression and angiogenesis via upregulation of SERPINE1 expression and activation of the VEGFR-2 signaling pathway in gastric cancer. Mol Oncol, 15(4):1234–1255. https://doi.org/10.1002/1878-0261.12911
Thiery JP, Acloque H, Huang RYJ, et al., 2009. Epithelial-mesenchymal transitions in development and disease. Cell, 139(5):871–890. https://doi.org/10.1016/j.cell.2009.11.007
Tsai JH, Yang J, 2013. Epithelial-mesenchymal plasticity in carcinoma metastasis. Genes Dev, 27(20):2192–2206. https://doi.org/10.1101/gad.225334.113
Vordermark D, 2005. Significance of hypoxia in malignant glioma. Re: Evans et al. Hypoxia is important in the biology and aggression of human glial brain tumors. Clin Cancer Res 2004; 10:8177–84. Clin Cancer Res, 11(10): 3966–3968. https://doi.org/10.1158/1078-0432.CCR-05-0097
Wang Q, Lu WJ, Yin T, et al., 2019. Calycosin suppresses TGF-β-induced epithelial-to-mesenchymal transition and migration by upregulating BATF2 to target PAI-1 via the Wnt and PI3K/Akt signaling pathways in colorectal cancer cells. J Exp Clin Cancer Res, 38:240. https://doi.org/10.1186/s13046-019-1243-7
Wang XW, Bustos MA, Zhang XQ, et al., 2020. Downregulation of the ubiquitin-E3 ligase RNF123 promotes upregulation of the NF-κB1 target SerpinE1 in aggressive glioblastoma tumors. Cancers (Basel), 12(5):1081. https://doi.org/10.3390/cancers12051081
Wang ZL, Shi YP, Ying CT, et al., 2021. Hypoxia-induced PLOD1 overexpression contributes to the malignant phenotype of glioblastoma via NF-κB signaling. Oncogene, 40(8):1458–1475. https://doi.org/10.1038/s41388-020-01635-y
Watson J, 2013. Oxidants, antioxidants and the current incurability of metastatic cancers. Open Biol, 3(1):120144. https://doi.org/10.1098/rsob.120144
Weidemann A, Johnson RS, 2008. Biology of HIF-1α. Cell Death Differ, 15(4):621–627. https://doi.org/10.1038/cdd.2008.12
Willson JA, Arienti S, Sadiku P, et al., 2022. Neutrophil HIF-1α stabilization is augmented by mitochondrial ROS produced via the glycerol 3-phosphate shuttle. Blood, 139(2):281–286. https://doi.org/10.1182/blood.2021011010
Wilson WR, Hay MP, 2011. Targeting hypoxia in cancer therapy. Nat Rev Cancer, 11(6):393–410. https://doi.org/10.1038/nrc3064
Wu K, Mao YY, Chen Q, et al., 2021. Hypoxia-induced ROS promotes mitochondrial fission and cisplatin chemosensitivity via HIF-1α/Mff regulation in head and neck squamous cell carcinoma. Cell Oncol (Dordr), 44(5):1167–1181. https://doi.org/10.1007/s13402-021-00629-6
Xia LM, Mo P, Huang WJ, et al., 2012. The TNF-α/ROS/HIF-1-induced upregulation of FoxMI expression promotes HCC proliferation and resistance to apoptosis. Carcinogenesis, 33(11):2250–2259. https://doi.org/10.1093/carcin/bgs249
Xu BD, Bai ZG, Yin J, et al., 2019. Global transcriptomic analysis identifies SERPINE1 as a prognostic biomarker associated with epithelial-to-mesenchymal transition in gastric cancer. PeerJ, 7:e7091. https://doi.org/10.7717/peerj.7091
Xu YQ, Chen WC, Liang J, et al., 2021. The miR-1185-2-3p-GOLPH3L pathway promotes glucose metabolism in breast cancer by stabilizing p53-induced SERPINE1. J Exp Clin Cancer Res, 40:47. https://doi.org/10.1186/s13046-020-01767-9
Yang J, Antin P, Berx G, et al., 2020. Guidelines and definitions for research on epithelial-mesenchymal transition. Nat Rev Mol Cell Biol, 21(6):341–352. https://doi.org/10.1038/s41580-020-0237-9
Yang JL, Ma Q, Zhang MM, et al., 2021. LncRNA CYTOR drives L-OHP resistance and facilitates the epithelial-mesenchymal transition of colon carcinoma cells via modulating miR-378a-5p/SERPINE1. Cell Cycle, 20(14):1415–1430. https://doi.org/10.1080/15384101.2021.1934626
Yang Y, Zhang GM, Guo FZ, et al., 2020. Mitochondrial UQCC3 modulates hypoxia adaptation by orchestrating OXPHOS and glycolysis in hepatocellular carcinoma. Cell Rep, 33(5):108340. https://doi.org/10.1016/j.celrep.2020.108340
Yu LM, Zhang WH, Han XX, et al., 2019. Hypoxia-induced ROS contribute to myoblast pyroptosis during obstructive sleep apnea via the NF-κB/HIF-1α signaling pathway. Oxid Med Cell Longev, 2019:4596368. https://doi.org/10.1155/2019/4596368
Zhang W, Zhang YW, Zhou WS, et al., 2021. PlGF knockdown attenuates hypoxia-induced stimulation of cell proliferation and glycolysis of lung adenocarcinoma through inhibiting Wnt/β-catenin pathway. Cancer Cell Int, 21:18. https://doi.org/10.1186/s12935-020-01714-w
Zhang YS, Jin GS, Zhang JW, et al., 2018. Overexpression of STAT1 suppresses angiogenesis under hypoxia by regulating VEGF-A in human glioma cells. Biomed Pharmacother, 104:566–575. https://doi.org/10.1016/j.biopha.2018.05.079
Zhao Q, Zhang LW, He QF, et al., 2023. Targeting TRMT5 suppresses hepatocellular carcinoma progression via inhibiting the HIF-1α pathways. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 24(1):50–63. https://doi.org/10.1631/jzus.B2200224
Zheng H, Kang Y, 2014. Multilayer control of the EMT master regulators. Oncogene, 33(14):1755–1763. https://doi.org/10.1038/onc.2013.128
Zong SQ, Tang YF, Li W, et al., 2020. A Chinese herbal formula suppresses colorectal cancer migration and vasculogenic mimicry through ROS/HIF-1α/MMP2 pathway in hypoxic microenvironment. Front Pharmacol, 11:705. https://doi.org/10.3389/fphar.2020.00705
Acknowledgments
We thank the supports from Hasenbio Technology Company (Wuxi, China) and the Public Experimental Research Center of Xuzhou Medical University (Xuzhou, China). This study was supported by the National Natural Science Foundation of China (Nos. 81772688 and 81372698), the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), the Research Foundation for Talented Scholars of Xuzhou Medical University (No. RC20552223), and the Postgraduate Research & Practice Innovation Program of Jiangsu Province (No. KYCX20_2463), China.
Author information
Authors and Affiliations
Contributions
Lin ZHANG and Yuanyuan CAO conceived and designed the study, performed the experiments and data analysis, and wrote and edited the manuscript. Xiaoxiao GUO and Xiaoyu WANG performed the experiments, analyzed the data, and drafted the manuscript. Xiao HAN performed data analysis and figure arrangements. Kouminin KANWORE checked the data and drafted the manuscript. Xiaoliang HONG and Han ZHOU provided technical supports. Dianshuai GAO supervised and conceived this work, and wrote the manuscript. All authors have reviewed and approved the final manuscript, and therefore, have full access to all the data in the work and take responsibility for the integrity and security of the data.
Corresponding author
Additional information
Compliance with ethics guidelines
Lin ZHANG, Yuanyuan CAO, Xiaoxiao GUO, Xiaoyu WANG, Xiao HAN, Kouminin Kanwore, Xiaoliang HONG, Han ZHOU, and Dianshuai GAO declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Supplementary information
Tables S1–S3; Figs. S1–S4
Supplementary Information
Rights and permissions
About this article
Cite this article
Zhang, L., Cao, Y., Guo, X. et al. Hypoxia-induced ROS aggravate tumor progression through HIF-1α-SERPINE1 signaling in glioblastoma. J. Zhejiang Univ. Sci. B 24, 32–49 (2023). https://doi.org/10.1631/jzus.B2200269
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2200269
Keywords
- Glioblastoma
- Hypoxia
- Reactive oxygen species (ROS)
- Hypoxia-inducible factor-1α (HIF-1α)
- Serine protease inhibitor family E member 1 (SERPINE1)