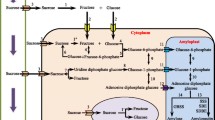
Abstract
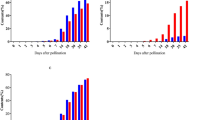
Starch is the predominant compound in bulb scales, and previous studies have shown that bulblet development is closely associated with starch enrichment. However, how starch synthesis affects bulbification at the molecular level is unclear. In this study, we demonstrate that Lilium brownii var. giganteum, a wild lily with a giant bulb in nature, and L. brownii, the native species, have different starch levels and characteristics according to cytological and ultra-structural observations. We cloned the complete sequence of three key gene-encoding enzymes (LbgAGPS, LbgGBSS, and LbgSSIII) during starch synthesis by rapid amplification of 5′ and 3′ complementary DNA (cDNA) ends (RACE) technology. Bioinformatics analysis revealed that the proteins deduced by these genes contain the canonical conserved domains. Constructed phylogenetic trees confirmed the evolutionary relationships with proteins from other species, including monocotyledons and dicotyledons. The transcript levels of various tissues and time course samples obtained during bulblet development uncovered relatively high expression levels in bulblets and gradual increase expression accompanying bulblet growth. Moreover, a set of single nucleotide polymorphisms (SNPs) was discovered in the AGPS genes of four lily genotypes, and a purifying selection fashion was predicted according to the non-synonymous/synonymous (Ka/Ks) values. Taken together, our results suggested that key starch-synthesizing genes might play important roles in bulblet development and lead to distinctive phenotypes in bulblet size.
Abstract
目的
本研究基于鳞茎特异性膨大野生种质巨球百合与其原种野百合在淀粉含量及结构上的差异, 克隆并分析了巨球百合三个淀粉合成关键基因, 旨在为进一步深入研究淀粉代谢在鳞茎形成中的作用机制提供理论参考。
创新点
本研究首次从重要野生百合资源中系统克隆淀粉合成关键酶基因, 并分析其时空表达特性, 获得了AGPS基因候选单核苷酸多态性 (SNP) 位点。
方法
采用蒽酮法测定非结构性碳水化合物含量并用高碘酸-希夫反应染色法及投射电镜观察淀粉的显微及超微结构; 采用cDNA末端快速扩增 (RACE) 技术克隆获得巨球百合淀粉合成相关基因, 并应用BLAST, ExPASy, TMHMM, SWISS-MODE, Cell-PLoc, SMART, MEGA及KaKs_Calculator等生物信息学软件对其基因序列进行分析; 采用实时荧光定量PCR分析基因的时空表达差异。
结论
本研究证实巨球百合及原生种野百合在淀粉含量及特性方面存在差异, 并用RACE技术克隆获得了LbgAGPS, LbgGBSS及LbgSSIII基因。三种基因均在鳞茎中表达较高, 且随着鳞茎发育整体呈现上升趋势, 与鳞茎的形成存在较强的相关性。AGPS基因上存在丰富的SNP位点, 且主要为纯化选择。
Similar content being viewed by others
References
Abt MR, Pfister B, Sharma M, et al., 2020. STARCH SYNTHASE 5, a noncanonical starch synthase-like protein, promotes starch granule initiation in Arabidopsis. Plant Cell, 32(8):2543–2565. https://doi.org/10.1105/tpc.19.00946
Arnold K, Bordoli L, Kopp P, et al., 2006. The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics, 22(2): 195–201. https://doi.org/10.1093/bioinformatics/bti770
Askari N, Visser RGF, de Klerk GJ, 2018. Growth of lily bulblets in vitro, a review. Int J Hortic Sci Technol, 5(2): 133–143. https://doi.org/10.22059/ijhst.2018.268870.263
Bahaji A, Li J, Sánchez-López ÁM, et al., 2014. Starch biosynthesis, its regulation and biotechnological approaches to improve crop yields. Biotechnol Adv, 32(1):87–106. https://doi.org/10.1016/j.biotechadv.2013.06.006
Bakhshaie M, Khosravi S, Azadi P, et al., 2016. Biotechnological advances in Lilium. Plant Cell Rep, 35(9):1799–1826. https://doi.org/10.1007/s00299-016-2017-8
Ballicora MA, Laughlin MJ, Fu Y, et al., 1995. Adenosine 5′-diphosphate-glucose pyrophosphorylase from potato tuber (significance of the N terminus of the small subunit for catalytic properties and heat stability). Plant Physiol, 109(1):245–251. https://doi.org/10.1104/pp.109.1.245
Beckles DM, Craig J, Smith AM, 2001. ADP-glucose pyrophosphorylase is located in the plastid in developing tomato fruit. Plant Physiol, 126(1):261–266. https://doi.org/10.1104/pp.126.1.261
Bejar CM, Ballicora MA, Iglesias AA, et al., 2006. ADPglucose pyrophosphorylase’s N-terminus: structural role in allosteric regulation. Biochem Biophys Res Commun, 343(1): 216–221. https://doi.org/10.1016/j.bbrc.2006.02.123
Cheng N, Zeng XF, Zheng XF, et al., 2015. Cloning and characterization of the genes encoding the small and large subunit of the ADP-glucose pyrophosphorylase in lotus (Nelumbo nucifera Gaertn). Acta Physiolo Plant, 37(1): 1734. https://doi.org/10.1007/s11738-014-1734-2
Chou KC, Shen HB, 2010. Cell-PLoc 2.0: an improved package of web-servers for predicting subcellular localization of proteins in various organisms. Nat Sci, 2(10):1090–1103. https://doi.org/10.4236/ns.2010.210136
de Klerk GJ, 2012. Micropropagation of bulbous crops: technology and present state. Floricult Ornam Biotechnol, 6: 1–8.
Deléage G, 2017. ALIGNSEC: viewing protein secondary structure predictions within large multiple sequence alignments. Bioinformatics, 33(24):3991–3992. https://doi.org/10.1093/bioinformatics/btx521
Doležel J, Greilhuber J, Suda J, 2007. Estimation of nuclear DNA content in plants using flow cytometry. Nat Protoc, 2(9):2233–2244. https://doi.org/10.1038/nprot.2007.310
Du F, Jiang J, Jia HM, et al., 2015. Selection of generally applicable SSR markers for evaluation of genetic diversity and identity in Lilium. Biochem Syst Ecol, 61:278–285. https://doi.org/10.1016/j.bse.2015.05.002
Du HH, Yang T, Ma CY, et al., 2012. Effects of RNAi silencing of SSIII gene on phosphorus content and characteristics of starch in potato tubers. Integr Agric, 11(12): 1985–1992. https://doi.org/10.1016/S2095-3119(12)60455-9
Du YP, Bi Y, Zhang MF, et al., 2017. Genome size diversity in Lilium (Liliaceae) is correlated with karyotype and environmental traits. Front Plant Sci, 8:1303. https://doi.org/10.3389/fpls.2017.01303
Ganal MW, Altmann T, Röder MS, 2009. SNP identification in crop plants. Curr Opin Plant Bio, 12(2):211–217. https://doi.org/10.1016/j.pbi.2008.12.009
Gao MP, Zhang SW, Luo C, et al., 2018. Transcriptome analysis of starch and sucrose metabolism across bulb development in Sagittaria sagittifolia. Gene, 649:99–112. https://doi.org/10.1016/j.gene.2018.01.075
Goren A, Ashlock D, Tetlow IJ, 2018. Starch formation inside plastids of higher plants. Protoplasma, 255(6):1855–1876. https://doi.org/10.1007/s00709-018-1259-4
Guan R, Zhao YP, Zhang H, et al., 2016. Draft genome of the living fossil Ginkgo biloba. Gigascience, 5(1):49. https://doi.org/10.1186/s13742-016-0154-1
Huang JH, Zhou RR, He D, et al., 2020. Rapid identification of Lilium species and polysaccharide contents based on near infrared spectroscopy and weighted partial least square method. Int J Biol Macromol, 154:182–187. https://doi.org/10.1016/j.ijbiomac.2020.03.109
Huang YF, Yang MX, Zhang H, et al., 2009. Genetic diversity and genetic structure analysis of the natural populations of Lilium brownii from Guangdong, China. Biochem Genet, 47(7–8):503–510. https://doi.org/10.1007/s10528-009-9258-y
Kamenetsky R, Okubo H, 2012. Ornamental Geophytes: from Basic Science to Sustainable Production. CRC Press, Boca Raton, Florida, USA, p.1–16.
Krogh A, Larsson B, von Heijne G, et al., 2001. Predicting transmembrane protein topology with a hidden markov model: application to complete genomes. J Mol Biol, 305(3): 567–580. https://doi.org/10.1006/jmbi.2000.4315
Kumar S, Stecher G, Tamura K, 2016. MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol, 33(7):1870–1874. https://doi.org/10.1093/molbev/msw054
Lee GJ, Subburaj S, Kumari S, et al., 2016. Identification of single nucleotide polymorphism (SNP) markers associated with botrytis resistance in lily (Lilium spp.). Hortscience, 51:S320–S321.
Letunic I, Bork P, 2018. 20 years of the SMART protein domain annotation resource. Nucleic Acids Res, 46(D1): D493–D496. https://doi.org/10.1093/nar/gkx922
Letunic I, Bork P, 2019. Interactive Tree Of Life (iTOL) v4: recent updates and new developments. Nucleic Acids Res, 47(W1):W256–W259. https://doi.org/10.1093/nar/gkz239
Li GY, Chen ZH, Yan FB, 2007. Lilium brownin var. giganteum: a new variety of Lilium from Wenling, Zhejiang. J Zhejiang Forestry Coll, 24(6):767–768 (in Chinese). https://doi.org/10.3969/j.issn.2095-0756.2007.06.023
Li XY, Wang CX, Cheng JY, et al., 2014. Transcriptome analysis of carbohydrate metabolism during bulblet formation and development in Lilium davidii var. unicolor. BMC Plant Biol, 14:358. https://doi.org/10.1186/s12870-014-0358-4
Linebarger CRL, Boehlein SK, Sewell AK, et al., 2005. Heat stability of maize endosperm ADP-glucose pyrophosphorylase is enhanced by insertion of a cysteine in the N terminus of the small subunit. Plant Physiol, 139(4): 1625–1634. https://doi.org/10.1104/pp.105.067637
Livak KJ, Schmittgen TD, 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods, 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Lloyd JR, Kossmann J, 2019. Starch trek: the search for yield. Front Plant Sci, 9:1930. https://doi.org/10.3389/fpls.2018.01930
Lombard V, Ramulu HG, Drula E, et al., 2014. The carbohydrate-active enzymes database (CAZy) in 2013. Nucleic Acids Res, 42(D1):D490–D495. https://doi.org/10.1093/nar/gkt1178
Ma PP, Yuan Y, Shen QC, et al., 2019. Evolution and expression analysis of starch synthase gene families in Saccharum spontaneum. Trop Plant Biol, 12(3):158–173. https://doi.org/10.1007/s12042-019-09225-3
Mammadov J, Aggarwal R, Buyyarapu R, et al., 2012. SNP markers and their impact on plant breeding. Int J Plant Genomics, 2012:728398. https://doi.org/10.1155/2012/728398
Matsuo T, Mizuno T, 1974. Changes in the amounts of two kinds of reserve glucose-containing polysaccharides during germination of the Easter lity bulb. Plant Cell Physiol, 15(3):555–558. https://doi.org/10.1093/oxfordjournals.pcp.a075036
Miao HX, Sun PG, Liu WX, et al., 2014. Identification of genes encoding granule-bound starch synthase involved in amylose metabolism in banana fruit. PLoS ONE, 9(2): e88077. https://doi.org/10.1371/journal.pone.0088077
Mishra BP, Kumar R, Mohan A, et al., 2017. Conservation and divergence of Starch Synthase III genes of monocots and dicots. PLoS ONE, 12(12):e0189303. https://doi.org/10.1371/journal.pone.0189303
Moreno-Pachón N, 2017. Mechanisms of Vegetative Propagation in Bulbs: A Molecular Approach. PhD Thesis, Wageningen University, Wageningen, the Netherlands.
Murashige T, Skoog F, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant, 15(3):473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Oteo-García G, Oteo JA, 2019. Evolutionary distances corrected for purifying selection and ancestral polymorphisms. J Theor Biol, 483:110004. https://doi.org/10.1016/j.jtbi.2019.110004
Podwyszyńska M, 2012. The mechanisms of in vitro storage organ formation in ornamental geophytes. Floricult Ornam Biotechnol, 6(1):9–23.
Qu JZ, Xu ST, Zhang ZQ, et al., 2018. Evolutionary, structural and expression analysis of core genes involved in starch synthesis. Sci Rep, 8:12736. https://doi.org/10.1038/s41598-018-30411-y
Seo SG, Bea SH, Jun BK, et al., 2015. Overexpression of ADP-glucose pyrophosphorylase (IbAGPaseS) affects expression of carbohydrate regulated genes in sweet potato [Ipomoea batatas (L.) Lam. cv. Yulmi]. Genes Genom, 37(7): 595–605. https://doi.org/10.1007/s13258-015-0289-y
Shahin A, van Kaauwen M, Esselink D, et al., 2012. Generation and analysis of expressed sequence tags in the extreme large genomes Lilium and Tulipa. BMC Genomics, 13:640. https://doi.org/10.1186/1471-2164-13-640
Smith AM, Zeeman SC, 2020. Starch: a flexible, adaptable carbon store coupled to plant growth. Annu Rev Plant Biol, 71:217–245. https://doi.org/10.1146/annurev-arplant-050718-100241
Thakur R, Sood A, Nagar PK, et al., 2006. Regulation of growth of Lilium plantlets in liquid medium by application of paclobutrazol or ancymidol, for its amenability in a bioreactor system: growth parameters. Plant Cell Rep, 25(5):382–391. https://doi.org/10.1007/s00299-005-0094-1
Thompson JD, Gibson TJ, Plewniak F, et al., 1997. The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res, 25(24):4876–4882. https://doi.org/10.1093/nar/25.24.4876
van Harsselaar JK, Lorenz J, Senning M, et al., 2017. Genome-wide analysis of starch metabolism genes in potato (Solanum tuberosum L.). BMC Genomics, 18:37. https://doi.org/10.1186/s12864-016-3381-z
van Tuyl JM, Arens P, Ramanna MS, et al., 2011. Lilium. In: Kole C (Ed.), Wild Crop Relatives: Genomic and Breeding Resources. Springer, Berlin Heidelberg, Germany, p.161–183. https://doi.org/10.1007/978-3-642-21201-7_9
van Tuyl JM, Arens P, Shahin A, et al., 2018. Lilium. In: van Huylenbroeck J (Ed.), Ornamental Crops. Springer, Cham, Switzerland, p.481–512. https://doi.org/10.1007/978-3-319-90698-0_20
Wang DP, Zhang YB, Zhang Z, et al., 2010. KaKs_Calculator 2.0: a toolkit incorporating gamma-series methods and sliding window strategies. Genom Proteom Bioinf, 8(1): 77–80. https://doi.org/10.1016/s1672-0229(10)60008-3
Wang X, Feng B, Xu ZB, et al., 2014. Identification and characterization of granule bound starch synthase I (GBSSI) gene of tartary buckwheat (Fagopyrum tataricum Gaertn.). Gene, 534(2):229–235. https://doi.org/10.1016/j.gene.2013.10.053
Wu Y, Xia YP, Zhang JP, et al., 2016. Low humic acids promote in vitro lily bulblet enlargement by enhancing roots growth and carbohydrate metabolism. J Zhejiang UnivSci B (Biomed&Biotechnol), 17(11):892–904. https://doi.org/10.1631/jzus.B1600231
Wu Y, Li Y, Ma YD, et al., 2017. Hormone and antioxidant responses of Lilium Oriental hybrids ‘Sorbonne’ bulblets to humic acid treatments in vitro. J Hortic Sci Biotech, 92(2):155–167. https://doi.org/10.1080/14620316.2016.1240019
Wu Y, Sun MY, Zhang JP, et al., 2019a. Differential effects of paclobutrazol on the bulblet growth of oriental lily cultured in vitro: growth behavior, carbohydrate metabolism, and antioxidant capacity. J Plant Growth Regul, 38(2):359–372. https://doi.org/10.1007/s00344-018-9844-5
Wu Y, Ma YD, Li Y, et al., 2019b. Plantlet regeneration from primary callus cultures of Lilium brownii F.E.Br. ex Miellez var. giganteum G. Y. Li & Z. H. Chen, a rare bulbous germplasm. In Vitro Cell Dev-Biol-Plant, 55(1):44–59. https://doi.org/10.1007/s11627-018-09955-1
Xu H, Yang PP, Cao YW, et al., 2020. Cloning and functional characterization of a flavonoid transport-related MATE gene in asiatic hybrid lilies (Lilium spp.). Genes, 11(4):418. https://doi.org/10.3390/genes11040418
Yang M, Zhu LP, Pan C, et al., 2015. Transcriptomic analysis of the regulation of rhizome formation in temperate and tropical lotus (Nelumbo nucifera). Sci Rep, 5:13059. https://doi.org/10.1038/srep13059
Yang PP, Xu LF, Xu H, et al., 2017. Histological and transcriptomic analysis during bulbil formation in Lilium lancifolium. Front Plant Sci, 8:1508. https://doi.org/10.3389/fpls.2017.01508
Zhou YX, Chen YX, Tao X, et al., 2016. Isolation and characterization of cDNAs and genomic DNAs encoding ADP-glucose pyrophosphorylase large and small subunits from sweet potato. Mol Genet Genomics, 291(2):609–620. https://doi.org/10.1007/s00438-015-1134-3
Zhu FL, Cheng N, Sun H, et al., 2020. Molecular cloning and characterization of a gene encoding soluble starch synthase III (SSSIII) in Lotus (Nelumbo nucifera). Biologia, 75(2):279–288. https://doi.org/10.2478/s11756-019-00341-9
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 31772337), the Lishui Research Fund (No. 2020zdhz03), the China Postdoctoral Science Foundation (No. 2017M612005), and the National Key Research and Development Program of China (No. 2018YFD10 00401). We gratefully thanks Tianyu HAN (Nanjing Forestry University, Nanjing, China) for his technical support.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Author contributions
Yiping XIA, Yun WU, and Ziming REN designed the experiments, wrote and edited the manuscript. Minyi SUN, Shiqi LI, Ruihan MIN, Cong GAO, and Qundan LYU performed the study and data analysis. All authors have read and approved the final version of the manuscript and, therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data.
Compliance with ethics guidelines
Yun WU, Minyi SUN, Shiqi LI, Ruihan MIN, Cong GAO, Qundan LYU, Ziming REN, and Yiping XIA declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Wu, Y., Sun, M., Li, S. et al. Molecular cloning, characterization and expression analysis of three key starch synthesis-related genes from the bulb of a rare lily germplasm, Lilium brownii var. giganteum. J. Zhejiang Univ. Sci. B 22, 476–491 (2021). https://doi.org/10.1631/jzus.B2000545
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2000545