Abstract
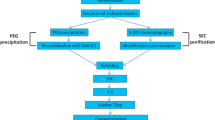
A large-scale approach to the purification of plasmid DNA has been developed that overcomes many of the limitations of current chromatography-based processes. The process consists of a scaleable lysis using recombinant lysozyme and a rapid heating and cooling step followed by a selective precipitation with cetyltrimethylammonium bromide (CTAB). Calcium silicate batch adsorption is then utilized to remove residual genomic DNA, linear plasmid, open circular plasmid, endotoxin, detergents, and proteins. Finally, a concentration and diafiltration step utilizing ultrafiltration and a terminal sterile filtration complete the process. The final product exceeds the requirements for clinical-grade plasmid DNA, and the process has been scaled up to yield an average of 18 ± 4 g (over five lots) of pharmaceutically pure plasmid DNA per 140 L of lysate (from approx 1.3 kg Escherichia coli dry cell weight).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Abdelnoor, A. M. (2001) Plasmid DNA vaccines. Curr. Drug Targets. Immune. Endocr. Metabol. Disord. 1, 79–92.
Robinson, H. L. (2002) New hope for an AIDS vaccine. Nat. Rev. Immunol. 2, 239–250.
Schmidt-Wolf, G. D. and Schmidt-Wolf, I. G. (2003) Non-viral and hybrid vectors in human gene therapy: an update. Trends Mol. Med. 9, 67–72.
Smith, H. A. (2000) Regulation and review of DNA vaccine products. Dev. Biol. (Basel). 104, 57–62.
Levy, M. S., O’Kennedy, R. D., Ayazi-Shamlou, P., and Dunnill, P. (2000) Biochemical engineering approaches to the challenges of producing pure plasmid DNA. Trends Biotechnol. 18, 296–305.
Sagar, S. L., Watson, M. P., and Lee, A. L. (2003) Chromatography-based purification of plasmid DNA. In: Scale-Up and Optimization in Preparative Chromatography: Principles and Biopharmaceutical Applications (Rathore, A. S. and Vella, G., eds.), Marcel Dekker: New York, Vol. 88, pp. 251–272.
Papadopoulos, S., Jurgens, K. D., and Gros, G. (2000) Protein diffusion in living skeletal muscle fibers: dependence on protein size, fiber type, and contraction. Biophys. J. 79, 2084–2094.
Lukacs, G. L., Haggie, P., Seksek, O., Lechardeur, D., Freedman, N., and Verkman, A. S. (2000) Size-dependent DNA mobility in cytoplasm and nucleus. J. Biol. Chem. 275, 1625–1629.
Prather, K. J., Sagar, S. L., Murphy, J. C., and Chartrain, M. (2003) Industrial scale production of plasmid DNA for vaccine and gene therapy: plasmid design, production, and purification. Enzy. Microb. Technol. 33, 865–883.
Lander, R. J., Winters, M. A., Meacle, F. J., Buckland, B. C., and Lee, A. L. (2002) Fractional precipitation of plasmid DNA from lysate by CTAB. Biotechnol. Bioeng. 79, 776–784.
Winters, M. A., Richter, J. D., Sagar, S. L., Lee, A. L., and Lander, R. J. (2003) Plasmid DNA purification by selective calcium silicate adsorption of closely related impurities. Biotechnol. Prog. 19, 440–447.
Lander, R. L., Winters, M. A. and Meacle, F. J. (2004) Process for the scaleable purification of plasmid DNA. Merck and Co., Inc., US Patent No. 6797476, 9-28-2004.
Birnboim, H. C. and Doly, J. (1979) A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 7, 1513–1523.
Chamsart, S., Patel, H., Hanak, J. A., Hitchcock, A. G., and Nienow, A. W. (2001) The impact of fluid-dynamic-generated stresses on chDNA and pDNA stability during alkaline cell lysis for gene therapy products. Biotechnol. Bioeng. 75, 387–392.
Varley, D. L., Hitchcock, A. G., Weiss, A. M., et al. (1999) Production of plasmid DNA for human gene therapy using modified alkaline cell lysis and expanded bed anion exchange chromatography. Bioseparation. 8, 209–217.
Holmes, D. S. and Quigley, M. (1981) A rapid boiling method for the preparation of bacterial plasmids. Anal. Biochem. 114, 193–197.
Sagar, S. L. and Lee, A. L. (2001) Production of a crude lysate from which plasmid DNA can be isolated, comprises passing cells from a large-scale microbial culture through a heat exchanger. Merck, US Patent No. 6197553, 6-3-2001.
Theodossiou, I., Collins, I. J., Ward, J. M., Thomas, O. R. T., and Dunnill, P. (1997) The processing of a plasmid-based gene from E-coli. Primary recovery by filtration. Bioprocess Engineering. 16, 175–183.
Theodossiou, I., Thomas, O. R. T., and Dunnill, P. (1999) Methods of enhancing the recovery of plasmid genes from neutralised cell lysate. Bioprocess Engineering. 20, 147–156.
Ishaq, M., Wolf, B., and Ritter, C. (1990) Large-scale isolation of plasmid DNA using cetyltrimethylammonium bromide. Biotechniques. 9, 19–20, 22, 24.
Dahlstrom, D. and Oldshue, J. Y. (1997) Phase contacting and liquid solid processing. In: Perry’s Chemical Engineers’ Handbook, 7th ed. (Perry, R. P., Green, D. W., and Maloney, J. O., eds.), McGraw-Hill: New York, vol. pp. 18?1-18?18.
Lengsfeld, C. S. and Anchordoquy, T. J. (2002) Shear-induced degradation of plasmid DNA. J. Pharm. Sci. 91, 1581–1589.
Levy, M. S., Collins, I. J., Yim, S. S., et al. (1999) Effect of shear on plasmid DNA in solution. Bioprocess Engineering. 20, 7–13.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2006 Humana Press Inc.
About this protocol
Cite this protocol
Murphy, J.C., Winters, M.A., Sagar, S.L. (2006). Large-Scale, Nonchromatographic Purification of Plasmid DNA. In: Saltzman, W.M., Shen, H., Brandsma, J.L. (eds) DNA Vaccines. Methods in Molecular Medicine™, vol 127. Humana Press. https://doi.org/10.1385/1-59745-168-1:351
Download citation
DOI: https://doi.org/10.1385/1-59745-168-1:351
Publisher Name: Humana Press
Print ISBN: 978-1-58829-484-5
Online ISBN: 978-1-59745-168-0
eBook Packages: Springer Protocols