Abstract
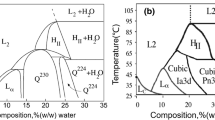
The purpose of this study was to design and investigate the transdermal controlled release cubic phase gels containing capsaicin using glycerol monooleate (MO), propylene glycol (1,2-propanediol, PG), and water. Three types of cubic phase gels were designed based on the ternary phase diagram of the MO–PG–water system, and their internal structures were confirmed by polarizing light microscopy (PLM) and small-angle X-ray scattering (SAXS). Release results showed the cubic phase gels could provide a sustained system for capsaicin, while the initial water content in the gels was the major factor affecting the release rate. Release kinetics was determined to fit Higuchi’s square-root equation indicating that the release was under diffusion control. The calculated diffusion exponent showed the release from cubic phase gels was anomalous transport. The unique structure of the cubic phases, capsaicin distributed in the lipid bilayers, and cubic phase gel swelling contributed to the release mechanism. The cubic phase gel may be an interesting application for transdermal delivery system of capsaicin in alleviating the post-incision pain.






Similar content being viewed by others
REFERENCES
Magnusson BM, Koskinen LOD. In vitro percutaneous penetration of topically applied capsaicin in relation to in vivo sensation responses. Int J Pharm. 2000;195(1–2):55–62.
Fusco B, Giacovazzo M. Peppers and pain: the promise of capsaicin. Drugs (Basel). 1997;53(6):909–14.
Hayman M, Kam PCA. Capsaicin: a review of its pharmacology and clinical applications. Curr Anaesth Crit Care. 2008;19(5–6):338–43.
Hamalainen MM, Subieta A, Arpey C, Brennan TJ. Differential effect of capsaicin treatment on pain-related behaviors after plantar incision. J Pain. 2009;10(6):637–45.
Kang S, Wu C, Banik RK, Brennan TJ. Effect of capsaicin treatment on nociceptors in rat glabrous skin one day after plantar incision. Pain. 2010;148(1):128–40.
Pospisilova E, Palecek J. Post-operative pain behavior in rats is reduced after single high-concentration capsaicin application. Pain. 2006;125(3):233–43. doi:10.1016/j.pain.2006.05.021.
Donnerer J, Amann R, Schuligoi R, Lembeck F. Absorption and metabolism of capsaicinoids following intragastric administration in rats. Naunyn-Schmiedebergs Arch Pharmakol. 1990;342(3):357–61.
Kawada T, Watanabe T, Katsura K, Takami H, Iwai K. Formation and metabolism of pungent principle of Capsicum fruits. XV. Microdetermination of capsaicin by high-performance liquid chromatography with electrochemical detection. J Chromatogr. 1985;329(1):99–105.
Fang JY, Wu PC, Huang YB, Tsai YH. In vivo percutaneous absorption of capsaicin, nonivamide and sodium nonivamide acetate from ointment bases: pharmacokinetic analysis in rabbits. Int J Pharm. 1996;128(1–2):169–77.
Geraghty P, Attwood D, Collett J, Dandiker Y. The in vitro release of some antimuscarinic drugs from monoolein/water lyotropic liquid crystalline gels. Pharm Res. 1996;13(8):1265–71.
Bender J, Ericson M, Merclin N, Iani V, Rosén A, Engström S, et al. Lipid cubic phases for improved topical drug delivery in photodynamic therapy. J Control Release. 2005;106(3):350–60.
Lopes LB, Lopes JLC, Oliveira DCR, Thomazini JA, Garcia MTJ, Fantini MCA, et al. Liquid crystalline phases of monoolein and water for topical delivery of cyclosporin A: characterization and study of in vitro and in vivo delivery. Eur J Pharm Biopharm. 2006;63(2):146–55. doi:10.1016/j.ejpb.2006.02.003.
Clogston J, Craciun G, Hart D, Caffrey M. Controlling release from the lipidic cubic phase by selective alkylation. J Control Release. 2005;102(2):441–61.
Fa N, Babak VG, Stebe MJ. The release of caffeine from hydrogenated and fluorinated gel emulsions and cubic phases. Colloids Surf A Physicochem Eng Asp. 2004;243(1–3):117–25. doi:10.1016/j.colsurfa.2004.05.014.
Rizwan SB, Hanley T, Boyd BJ, Rades T, Hook S. Liquid crystalline systems of phytantriol and glyceryl monooleate containing a hydrophilic protein: characterisation, swelling and release kinetics. J Pharm Sci-Us. 2009;98(11):4191–204. doi:10.1002/jps.21724.
Larsson K. Cubic lipid-water phases: structures and biomembrane aspects. J Phys Chem. 1989;93(21):7304–14.
Shah JC, Sadhale Y, Chilukuri DM. Cubic phase gels as drug delivery systems. Adv Drug Deliv Rev. 2001;47(2–3):229–50.
Chang C, Bodmeier R. Low viscosity monoglyceride-based drug delivery systems transforming into a highly viscous cubic phase. Int J Pharm. 1998;173(1–2):51–60.
Wadsten-Hindrichsen P, Bender J, Unga J, Engström S. Aqueous self-assembly of phytantriol in ternary systems: effect of monoolein, distearoylphosphatidylglycerol and three water-miscible solvents. J Colloid Interface Sci. 2007;315(2):701–13.
Caffrey M. A lipid’s eye view of membrane protein crystallization in mesophases. Curr Opin Struct Biol. 2000;10(4):486–97.
Hyde S, Andersson S, Ericsson B, Larsson K. A cubic structure consisting of a lipid bilayer forming an infinite periodic minimum surface of the gyroid type in the glycerolmonooleat-water system. Z Kristallogr. 1984;168(1–4):213–9.
Park E, Maniar M, Shah J. Biodegradable polyanhydride devices of cefazolin sodium, bupivacaine, and taxol for local drug delivery: preparation, and kinetics and mechanism of in vitro release. J Control Release. 1998;52(1–2):179–89.
Lee J, Kellaway I. In vitro peptide release from liquid crystalline buccal delivery systems. Int J Pharm. 2000;195(1–2):29–33.
Ritger P, Peppas N. A simple equation for description of solute release. II. Fickian and anomalous release from swellable devices. J Control Release. 1987;5(1):37–42.
Li S, Shen Y, Li W, Hao X. A common profile for polymer-based controlled releases and its logical interpretation to general release process. J Pharm Pharm Sci. 2006;9(2):238–44.
Lara M, Bentley M, Collett J. In vitro drug release mechanism and drug loading studies of cubic phase gels. Int J Pharm. 2005;293(1–2):241–50.
Ravi PR, Kotreka UK, Saha RN. Controlled release matrix tablets of zidovudine: effect of formulation variables on the in vitro drug release kinetics. AAPS PharmSciTech. 2008;9(1):302–13. doi:10.1208/s12249-007-9030-8.
Anumolu S, Singh Y, Gao D, Stein S, Sinko P. Design and evaluation of novel fast forming pilocarpine-loaded ocular hydrogels for sustained pharmacological response. J Control Release. 2009;137(2):152–9.
Martinez-Ruvalcaba A, Sanchez-Diaz JC, Becerra F, Cruz-Barba LE, Gonzalez-Alvarez A. Swelling characterization and drug delivery kinetics of polyacrylamide-co-itaconic acid/chitosan hydrogels. Express Polym Lett. 2009;3(1):25–32. doi:10.3144/expresspolymlett.2009.5.
Rizwan S, Dong Y, Boyd B, Rades T, Hook S. Characterisation of bicontinuous cubic liquid crystalline systems of phytantriol and water using cryo field emission scanning electron microscopy (cryo FESEM). Micron. 2007;38(5):478–85.
Alexandridis P, Olsson U, Lindman B. A record nine different phases (four cubic, two hexagonal, and one lamellar lyotropic liquid crystalline and two micellar solutions) in a ternary isothermal system of an amphiphilic block copolymer and selective solvents (water and oil). Langmuir. 1998;14(10):2627–38.
Polyzos A, Alderton M, Dawson R, Hartley P. Biofunctionalized surfactant mesophases as polyvalent inhibitors of cholera toxin. Bioconjugate Chem. 2007;18(5):1442–9.
Wang Z, Zheng L, Inoue T. Effect of sucrose on the structure of a cubic phase formed from a monoolein/water mixture. J Colloid Interface Sci. 2005;288(2):638–41.
Tanaka N, Imai K, Okimoto K, Ueda S, Tokunaga Y, Ohike A, et al. Development of novel sustained-release system, disintegration-controlled matrix tablet (DCMT) with solid dispersion granules of nilvadipine. J Control Release. 2005;108(2–3):386–95. doi:10.1016/j.jcornel.2005.08.024.
ACKNOWLEDGEMENTS
The authors are grateful to Danisco Company, Denmark, for the generous gift samples of MO. We thank the National Natural Science Foundation of China for the financial support (No 81001643/H2806). This work was also supported in part by the Ministry of Science and Technology of Dongguan (NO 2008108101064).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peng, X., Wen, X., Pan, X. et al. Design and In Vitro Evaluation of Capsaicin Transdermal Controlled Release Cubic Phase Gels. AAPS PharmSciTech 11, 1405–1410 (2010). https://doi.org/10.1208/s12249-010-9481-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-010-9481-1