Abstract
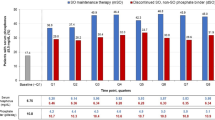
Sucroferric oxyhydroxide (Velphoro®), an iron-based oral phosphate binder, is available for the control of serum phosphorus levels in patients with chronic kidney disease (CKD) on dialysis. In a pivotal phase III trial, sucroferric oxyhydroxide 1000–3000 mg/day for 24 weeks was noninferior to sevelamer carbonate 4800–14,400 mg/day with regard to lowering serum phosphorus levels. Additionally, sucroferric oxyhydroxide at maintenance dosages was significantly more effective than low dosage sucroferric oxyhydroxide (250 mg/day) with regard to maintaining controlled serum phosphorus levels during weeks 24–27 of treatment. Sucroferric oxyhydroxide had a numerically lower mean daily pill burden and better treatment adherence than sevelamer carbonate. Treatment with sucroferric oxyhydroxide was generally well tolerated over 24 weeks’ treatment, with the most frequently reported treatment-emergent adverse events being mild, transient diarrhoea and discoloured faeces. In a 28-week extension study, the efficacy and tolerability profile of sucroferric oxyhydroxide remained similar to sevelamer carbonate for up to 52 weeks. In conclusion, sucroferric oxyhydroxide is a valuable treatment option for hyperphosphataemia in CKD patients on dialysis, providing an effective and generally well tolerated noncalcium-based phosphate binder therapy with a lower pill burden than sevelamer carbonate and the potential for improved treatment adherence.

Similar content being viewed by others
References
Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int Suppl. 2009;113:S1–130.
Covic A, Rastogi A. Hyperphosphatemia in patients with ESRD: assessing the current evidence linking outcomes with treatment adherence. BMC Nephrol. 2013;14:153.
Ketteler M, Wuthrich RP, Floege J. Management of hyperphosphataemia in chronic kidney disease-challenges and solutions. Clin Kidney J. 2013;6(2):128–36.
Cupisti A, Gallieni M, Rizzo MA, et al. Phosphate control in dialysis. Int J Nephrol Renovasc Dis. 2013;6:193–205.
National Institute for Health and Care Excellence. Hyperphosphataemia in chronic kidney disease: management of hyperphosphataemia in patients with stage 4 or 5 chronic kidney disease (NICE clinical guideline 157). 2013. http://www.nice.org.uk. Accessed 23 Feb 2015.
Moe SM, Chertow GM. The case against calcium-based phosphate binders. Clin J Am Soc Nephrol. 2006;1(4):697–703.
Fresenius Medical Care North America. Velphoro (sucroferric oxyhydroxide) chewable tablet for oral use: US prescribing information. 2014. http://www.velphoro.us. Accessed 23 Feb 2015.
Vifor Fresenius Medical Care Renal Pharma France. Velphoro 500 mg chewable tablets: EU summary of product characteristics. 2015. http://www.ema.europa.eu. Accessed 23 Feb 2015.
Kissei Pharmaceutical Co. Ltd. New drug application submitted for “PA21 (development code)” for treatment of hyperphosphatemia [media release]. 19 Nov 2014. http://kissei.co.jp/e_contents.
Wilhelm M, Gaillard S, Rakov V, et al. The iron-based phosphate binder PA21 has potent phosphate binding capacity and minimal iron release across a physiological pH range in vitro. Clin Nephrol. 2014;81(04):251–8.
Hergesell O, Ritz E. Stabilized polynuclear iron hydroxide is an efficient oral phosphate binder in uraemic patients. Nephrol Dial Transplant. 1999;14(4):863–7.
Lanz M, Baldischweiler J, Kriwet B, et al. Chewability testing in the development of a chewable tablet for hyperphosphatemia. Drug Dev Ind Pharm. 2014;40(12):1623–31.
Geisser P, Philipp E. PA21: a novel phosphate binder for the treatment of hyperphosphatemia in chronic kidney disease. Clin Nephrol. 2010;74(1):4–11.
Floege J, Covic AC, Ketteler M, et al. A phase III study of the efficacy and safety of a novel iron-based phosphate binder in dialysis patients. Kidney Int. 2014;86:638–47.
Floege J, Covic AC, Ketteler M, et al. Long-term effects of iron-based phosphate binder, sucroferric oxyhydroxide, in dialysis patients. Nephrol Dial Transplant. 2015. doi:10.1093/ndt/gfv006.
Sprague SM, Covic A, Floege J, et al. Concomitant intravenous iron use drives changes in iron indices in a phase 3 study of PA21 [abstract plus poster]. In: National Kidney Foundation 2014 Spring Clinical Meeting. 2014.
Chong E, Kalia V, Willsie S, et al. Drug-drug interactions between sucroferric oxyhydroxide and losartan, furosemide, omeprazole, digoxin and warfarin in healthy subjects. J Nephrol. 2014;27(6):659–66.
Levesque V, Chong EMF, Moneuse P. Post-hoc analysis of pharmacodynamic interaction of PA21 with statins in a phase 3 study of PA21 in dialysis patients with hyperphosphatemia [abstract no. SA-PO568]. In: American Society of Nephrology Kidney Week 2013. 2013.
Floege J, Botha J, Chong E, et al. PA21 does not interact with oral vitamin D receptor agonists: a post hoc analysis of a phase 3 study [abstract no. SP257]. Nephrol Dial Transplant. 2014;29(Suppl 3):iii157.
Wuthrich RP, Chonchol M, Covic A, et al. Randomized clinical trial of the iron-based phosphate binder PA21 in hemodialysis patients. Clin J Am Soc Nephrol. 2013;8(2):280–9.
Vifor Inc. A phase 3 study to investigate the safety and efficacy of PA21, a phosphate binder, in dialysis patients [ClinicalTrials.gov identifier NCT01324128]. US National Institutes of Health, ClinicalTrials.gov. 2013. http://www.clinicaltrials.gov/. Accessed 23 Feb 2015.
Covic AC, Floege J, Ketteler M, et al. Efficacy and safety of PA21, a novel iron-based phosphate binder in CKD patients on peritoneal- and hemodialysis [abstract]. In: 15th Congress of the International Society of Peritoneal Dialysis. 2014.
Floege J. Efficacy and safety of the novel iron-based phosphate binder PA21 in peritoneal- and hemodialysis-dependent CKD patients [abstract plus poster]. In: 11th European Peritoneal Dialysis Meeting. 2013.
Covic A, Ketteler M, Rastogi A, et al. Comparison of safety profiles of PA21 and sevelamer carbonate in a post hoc analysis of a phase 3 study [abstract no. SP245]. Nephrol Dial Transplant. 2014;29(Suppl 3):iii153.
Malberti F. Hyperphosphataemia: treatment options. Drugs. 2013;73(7):673–88.
Nastou D, Fernandez-Fernandez B, Elewa U, et al. Next-generation phosphate binders: focus on iron-based binders. Drugs. 2014;74(8):863–77.
Chiu YW, Teitelbaum I, Misra M, et al. Pill burden, adherence, hyperphosphatemia, and quality of life in maintenance dialysis patients. Clin J Am Soc Nephrol. 2009;4(6):1089–96.
Wang S, Alfieri T, Ramakrishnan K, et al. Serum phosphorus levels and pill burden are inversely associated with adherence in patients on hemodialysis. Nephrol Dial Transplant. 2014;29(11):2092–9.
Gutzwiller FS, Braunhofer PG, Szucs TD, et al. Health economic evaluation of non-calcium-based phosphate binders in Scotland [abstract no. SP596]. Nephrol Dial Transplant. 2014;29(Suppl 3):iii271.
Disclosure
The preparation of this review was not supported by any external funding. During the peer review process, the manufacturer of the agent under review was offered an opportunity to comment on this article. Changes resulting from comments received were made by the authors on the basis of scientific and editorial merit. Sarah Greig and Greg Plosker are salaried employees of Adis/Springer.
Author information
Authors and Affiliations
Corresponding author
Additional information
The manuscript was reviewed by: B. S. Spinowitz, Nephrology, New York Hospital, Queens, New York, NY, USA; R. P. Wüthrich, Division of Nephrology, University Hospital, Zurich, Switzerland.
Rights and permissions
About this article
Cite this article
Greig, S.L., Plosker, G.L. Sucroferric Oxyhydroxide: A Review in Hyperphosphataemia in Chronic Kidney Disease Patients Undergoing Dialysis. Drugs 75, 533–542 (2015). https://doi.org/10.1007/s40265-015-0366-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-015-0366-1