Abstract
Background
In recent years, the list of tumor entities in the sinonasal tract has significantly expanded, requiring advanced diagnostic testing. We report the case of a 32-year-old patient with an unusual NUT carcinoma originating in the maxillary sinus, which showed extensive well-differentiated, papillary squamous morphology, similar to the spectrum of the recently described DEK::AFF2 fusion-associated carcinoma.
Methods
We performed immunohistochemical and molecular studies including EBV- and HPV-testing, as well as DNA/RNA next generation sequencing.
Results
The tumor showed predominantly exophytic papillary growth with mature squamous differentiation. An additional component harbored atypical, less differentiated basaloid tumor cells with infiltration of the adjacent stroma. Conspicuous inflammation was evident. There was no evidence of HPV DNA or EBV RNA. Next-generation sequencing revealed a NUT::NSD3 gene fusion corresponding to (“speckled-type”) immunopositivity of NUT in the tumor cells.
Conclusions
We describe a NUT::NSD3 gene fusion-associated NUT carcinoma of the sinonasal tract with a deceptively well-differentiated papillary growth pattern, thus expanding the morphological spectrum of this typically poorly differentiated neoplasm.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over the past decade, the spectrum of malignant tumors of the head and neck has expanded, with many entities characterized by distinct molecular alterations. For example, carcinomas comprise conventional squamous cell carcinomas, NUT carcinomas, DEK::AFF2 carcinomas, EBV- and HPV-associated carcinomas, undifferentiated as well as SWI/SNF complex deficient sinonasal carcinomas, highlighting the variety of different morphologies and molecular pathogeneses [1]. However, morphological overlap between different entities should be considered during the process of histopathological diagnosis. Here we report the case of a 32-year-old male patient, who presented with a non-healing lesion of the upper alveolar ridge after tooth extraction, leading to an oro-antral fistula. The histological features of the initial biopsy appeared deceptively bland, prompting the differential diagnosis of reactive inflammatory changes. However, an external histopathological consultation accompanied by molecular work-up with the detection of a NSD3::NUTM1 fusion, yielded the unexpected diagnosis of a sinonasal NUT carcinoma originating from the maxillary sinus.
Clinical Presentation
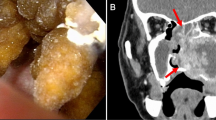
A 32-year-old, otherwise healthy, actively smoking male patient, presented with a 5-month history of pain in the left upper jaw. He was referred to a dentist, and after treatment, including extraction of tooth 25, wound healing delay was accompanied by a persistent oro-antral fistula (Fig. 1). On examination, the fistula was localized at the site of the extracted tooth and the alveolar ridge of the posterior part appeared enlarged. The first biopsy showed chronic-active inflammatory changes. A post-biopsy CT scan of the paranasal sinuses demonstrated a large osseous defect and bone erosion at the site of the extracted tooth with complete opacification of the left maxillary sinus (Fig. 2A). Despite the inflammatory changes noted in the first biopsy, the CT scan was interpreted as suspicious for malignancy. A second, larger biopsy was performed one month later, revealing a non-keratinizing squamous cell carcinoma. For staging purposes and resection planning, a whole body FDG-PET/CT was performed, showing metabolically enhanced osseous destruction in the left maxillary sinus (Fig. 2B). The patient was discussed at our multidisciplinary tumor board with the consensus that disease was staged as cT2 cN0 cM0 (UICC/TNM 8th edition), requiring primary resection. A hemimaxillectomy with wide margins was performed. In addition, the patient underwent a selective neck dissection level I-III on the left side followed by reconstruction of the defect with a superficial circumflex iliac artery-based iliac bone-free flap.
Pathology
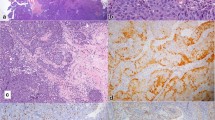
The initial biopsies revealed an exophytic-papillomatous (Fig. 3A), partly inverted tumor with squamous differentiation without unequivocal evidence of invasion. Based on the clinical context of a prior tooth extraction with persistent oro-antral fistula, differential diagnostic considerations encompassed prominent reactive inflammatory changes as well as an exophytic-papillomatous, well-differentiated carcinoma. Mucocytes were not detected in the Alcian-Blue-PAS-stain. Therefore, despite abundantly admixed granulocytes, a sinonasal papilloma (which could have aided the diagnosis as a possible precursor lesion) could not be confirmed. In light of the relatively mature squamous differentiation, minimal cytologic atypia and prominent inflammation, a clear diagnosis was hampered. In the second biopsy, small, discohesive collections of epithelial cells infiltrating the stroma with focal transformation into larger, basaloid aggregates without clear demarcation by a basement membrane, militated against the diagnosis of a reactive process (Fig. 3B–D). Additionally, the time course and clinico-radiological features favored a malignant process. The interpretation as a reactive squamous epithelial proliferation was revised with the descriptive diagnosis of an exophytic-papillomatous and partly endophytic growing carcinoma. In the ensuing external pathologic consultation, a diagnosis of a non-keratinizing squamous cell carcinoma (NKSCC) was rendered, assuming that the lesion originated from the sinonasal tract rather than the mucosa of the oral cavity, based on the latest WHO classification of Head & Neck Tumours 5th edition (beta version) [2]. HPV DNA testing as well as EBV-RNA in situ hybridization and p16 immunohistochemistry were negative. In order to address the differential diagnosis of a DEK::AFF2 fusion-associated carcinoma, molecular profiling was performed using the FoundationOne® Heme test. DEK::AFF2 fusion-associated carcinomas have been described recently as an emerging entity in the sinonasal tract, with the majority showing a strikingly bland histologic appearance and overlap with so-called low-grade papillary Schneiderian carcinomas [3, 4]. Importantly, the detection of a DEK::AFF2 gene fusion would allow for more accurate classification and prognostic assessment. Surprisingly, no DEK::AFF2, but a NUT::NSD3 gene fusion was detected, leading to the diagnosis of a NUT carcinoma. A subsequently performed NUT immunohistochemistry (Fig. 3E) showed a matching “speckled type” positivity in the majority of the carcinoma cells (almost 100% in both, basaloid and more differentiated components), corroborating the diagnosis and visualizing the fusion product. In concordance with the morphology lacking mucocytes, no EGFR mutation was detected, which are very common in inverted sinonasal papilloma and their carcinoma ex papilloma [5]. The macroscopy of the following left-sided hemimaxillectomy showed the main tumor originating in the maxillary sinus and breaking through the bone into the oral cavity. Together with the neck dissection specimen level I-III the final pathologic tumor staging (according to carcinomas of the nasal cavity and paranasal sinuses) was pT2 pN0 (0/57) L0 V0 Pn1, high-grade, R0 (UICC/TNM 8th edition, 2017). Extensive perineural spread was noted.
A–D Histopathology of the tumor: A Microscopically, the tumor shows papillomatous-exophytic well-differentiated squamous differentiation (10x), B–C infiltration of basaloid-appearing, less differentiated cells into the adjacent stroma (20x and 40x), D accompanied by prominent granulocytic infiltration (20x). E Immunohistochemistry for NUT (C52B1, Rabbit mAb, Cell Signaling Technology) (40x, magnified inset)
Clinical Follow-up
Adjuvant local radiotherapy was recommended at our multidisciplinary tumorboard. One year after diagnosis and six months after completion of treatment (at the time of the case report submission), the patient showed no evidence of disease, neither clinically nor on PET/CT.
Discussion
The histological and clinical features of the current case represent a highly unusual constellation. Typical NUT carcinoma is characterized by a more undifferentiated monomorphic morphology with small squamous islets and abrupt keratinization. These features were not present in our case. Nevertheless, the molecular profile, the NUT::NSD3 gene fusion, has been recurrently described in NUT carcinomas and confirms the diagnosis, especially in association with a squamous phenotype. Accordingly, this case can be regarded as part of the spectrum of NUT carcinomas and emphasizes the importance of considering this differential diagnosis in mature and well-differentiated squamous cell carcinoma. Such atypical features as well as the lack of awareness of this entity suggest an under-diagnosis and -reporting of NUT carcinomas [6]. The partly prominent squamous epithelial differentiation and the growth pattern are highly unusual and to the best of our knowledge have not been described in NUT carcinomas. In this regard, NUT carcinomas are characterized by translocation-associated fusion oncoproteins that interfere with cell differentiation and cell growth. The majority of NUT-fusions involves BRD4 (bromodomain containing protein 4), leading to an epigenetically induced block of cell differentiation and promotion of cellular growth. NSD3 encodes a histone lysine methyltransferase that binds the extraterminal domain of BRD. In cases harboring the NUT::NSD3 fusion, this alteration probably leads to similar functional oncogenic consequences. However, as presented in this case, the level of interference with cell differentiation might be different in NUT::NSD3 fusion than in NUT::BRD4 fusion [7]. This could explain why NUT::NSD3 fusion positive carcinomas outside the thorax appear to have a significantly better prognosis than their NUT::BRD4 positive counterparts [8]. An additional diagnostic challenge are the reactive, inflammatory squamous epithelial changes, which can be prominent after an intervention such as a tooth extraction. The relatively young patient age and the unusual morphology led to the consideration of an HPV-associated carcinoma, which could not be substantiated, as immunohistochemistry for p16 and molecular analysis for HPV DNA were negative. A carcinoma with DEK::AFF2 gene fusion was considered as the primary differential diagnosis on morphologic grounds. These carcinomas have recently been described and exhibit similar morphologic features to the current case [3, 4]. Importantly, this case presented significant morphological overlap with other head and neck carcinomas. The NUT::NSD3 gene fusion has recently been described in a subset of thyroid carcinomas without classical features, so that there is a rationale for NUT immunohistochemistry and/or molecular testing in unusual cases. In particular, there is increasing evidence that NUT gene fusions can occur in tumors with different underlying cell types (other than squamoid-like cells), such as thyroid follicle cells. Additional data are needed for accurate classification of these increasingly detected neoplasms [9, 10]. The concept of tumoral-mucosal fusion as a potential pitfall of processes underlying the surface mucosa is recognized in minor salivary gland neoplasia [11]. However, the observation that the majority of bland squamous cells in the mucosa were NUT IHC positive in our case, suggests that maturation may be involved. This case further demonstrates that highly sensitive and specific NUT immunohistochemistry is useful in identifying cases with unusual morphology, thus enabling accurate classification.
Future studies on larger numbers of cases are needed for comparing the biological behavior and other features of “differentiated NUT carcinoma” with the classical type.
Data Availability
Not Applicable.
Code Availability
Not Applicable.
References
Thompson LDR, Bishop JA (2022) Update from the 5th Edition of the World health organization classification of head and neck tumors: nasal cavity paranasal sinuses and skull base. Head Neck Pathol 16:1–18. https://doi.org/10.1007/s12105-021-01406-5
WHO Classification of Tumours Editorial Board. Head and neck tumours [Internet; beta version ahead of print]. Lyon (France): International Agency for Research on Cancer; 2022 [cited 2022 12 19]. (WHO classification of tumours series, 5th ed.; vol. 9). Available from: https://tumourclassification.iarc.who.int/chapters/52.
Kuo YJ, Lewis JS Jr, Zhai C, Chen YA, Chernock RD, Hsieh MS, Lan MY, Lee CK, Weinreb I, Hang JF (2021) DEK-AFF2 fusion-associated papillary squamous cell carcinoma of the sinonasal tract: clinicopathologic characterization of seven cases with deceptively bland morphology. Mod Pathol 34:1820–1830. https://doi.org/10.1038/s41379-021-00846-2
Rooper LM, Agaimy A, Dickson BC, Dueber JC, Eberhart CG, Gagan J, Hartmann A, Khararjian A, London NR, MacMillan CM, Palsgrove DN, Nix JS, Sandison A, Stoehr R, Truong T, Weinreb I, Bishop JA (2021) DEK-AFF2 carcinoma of the sinonasal region and skull base: detailed clinicopathologic characterization of a distinctive entity. Am J Surg Pathol 45:1682–1693. https://doi.org/10.1097/PAS.0000000000001741
Udager AM, Rolland DCM, McHugh JB, Betz BL, Murga-Zamalloa C, Carey TE, Marentette LJ, Hermsen MA, DuRoss KE, Lim MS, Elenitoba-Johnson KSJ, Brown NA (2015) High-Frequency targetable EGFR mutations in sinonasal squamous cell carcinomas arising from inverted sinonasal papilloma. Cancer Res 75:2600–2606. https://doi.org/10.1158/0008-5472.CAN-15-0340
French CA, Cheng ML, Hanna GJ, DuBois SG, Chau NG, Hann CL, Storck S, Salgia R, Trucco M, Tseng J, Stathis A, Piekarz R, Lauer UM, Massard C, Bennett K, Coker S, Tontsch-Grunt U, Sos ML, Liao S, Wu CJ, Polyak K, Piha-Paul SA, Shapiro GI (2022) Report of the first international symposium on NUT carcinoma. Clin Cancer Res 28:2493–2505. https://doi.org/10.1158/1078-0432.CCR-22-0591
Moreno V, Saluja K, Pina-Oviedo S (2022) NUT carcinoma: clinicopathologic features, molecular genetics and epigenetics. Front Oncol 12:860830. https://doi.org/10.3389/fonc.2022.860830
Chau NG, Ma C, Danga K, Al-Sayegh H, Nardi V, Barrette R, Lathan CS, DuBois SG, Haddad RI, Shapiro GI, Sallan SE, Dhar A, Nelson JJ, French CA (2020) An anatomical site and genetic-based prognostic model for patients with nuclear protein in testis (NUT) midline carcinoma: analysis of 124 patients. JNCI Cancer Spectr 4:pkz094. https://doi.org/10.1093/jncics/pkz094
Allison DB, Rueckert J, Cornea V, Lee CY, Dueber J, Bocklage T (2022) Thyroid carcinoma with NSD3::NUTM1 fusion: a Case with thyrocyte differentiation and colloid production. Endocr Pathol 33:315–326. https://doi.org/10.1007/s12022-021-09700-2
Barletta JA, Gilday SD, Afkhami M, Bell D, Bocklage T, Boisselier P, Chau NG, Cipriani NA, Costes-Martineau V, Ghossein RA, Hertzler HJ, Kramer AM, Limaye S, Lopez CA, Ng TL, Weissferdt A, Xu B, Zhang S, French CA (2022) NUTM1-rearranged carcinoma of the thyroid: a distinct subset of NUT carcinoma characterized by frequent NSD3-NUTM1 fusions. Am J Surg Pathol. https://doi.org/10.1097/PAS.0000000000001967
Ihrler S, Agaimy A, Guntinas-Lichius O, Haas CJ, Mollenhauer M, Sandison A, Greber L (2021) Why is the histomorphological diagnosis of tumours of minor salivary glands much more difficult? Histopathology 79:779–790. https://doi.org/10.1111/his.14421
Acknowledgements
We thank the Department of Oral Surgery and Stomatology, ZMK School of Dentistry, University of Bern, Bern, Switzerland for their contribution of the clinical photography of the oral lesion and Matteo Montani, senior pathologist at the Institute of Pathology, University of Bern, who confirmed the carcinoma diagnosis on the second biopsy. Further thanks go to the Molecular Tumor Profiling Laboratory at the Department of Pathology and Molecular Pathology, University Hospital Zurich, for performing the molecular analysis. Further thanks go to Prof. Dr. med. Elisabeth Rushing (Medica Zürich) for editing the manuscript.
Funding
Open access funding provided by University of Bern. This study was not supported by any funding.
Author information
Authors and Affiliations
Contributions
MW/AM: manuscript preparation (including pathologic diagnoses and images), discussion and review; SLH/RG: manuscript preparation (including clinical diagnoses and images), discussion and review; NJR: manuscript preparation (including pathologic diagnoses/external consultation including molecular diagnosis), discussion and review.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. The authors have no financial or non-financial interests that are directly or indirectly related to the work submitted for publication to disclose.
Ethical Approval
Written consent for publication was obtained from the patient. All procedures involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent for Publication
Consent for publication was obtained from every individual included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wartenberg, M., Hool, SL., Marrazzini, A. et al. Differentiated Papillary NUT Carcinoma: An Unexpected, Deceptively Bland Presentation of a Sinonasal Carcinoma. Head and Neck Pathol 17, 803–807 (2023). https://doi.org/10.1007/s12105-023-01554-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-023-01554-w