Abstract
Chikungunya virus (CHIKV) has been involved in epidemics in African and Asian subcontinents and, of late, has transcended to affect the Americas. Aedes aegypti and Aedes albopictus are the major vectors for CHIKV infection, which results in dissemination of virus to various vital organs. Entry of virus into these tissues causes infiltration of innate immune cells, monocytes, macrophages, neutrophils, natural killer cells, and adaptive immune cells. Macrophages bearing the replicating virus, in turn, secrete pro-inflammatory cytokines IL-1β, TNF-α, and IL-17. Together, this pro-inflammatory milieu induces osteoclastogenesis, bone loss, and erosion. CHIKV is characterized by fever, headache, myalgia, rash, and symmetric polyarthritis, which is generally self-limiting. In a subset of cases, however, musculoskeletal symptoms may persist for up to 3–5 years. Viral culture and isolation from blood cells of infected patients are the gold standards for diagnosis of CHIKV. In routine practice, however, assays for anti-CHIKV IgM antibodies are used for diagnosis, as elevated levels in blood of infected patients are noted from 10 days following infection for up to 3–6 months. Early diagnosis of CHIKV is possible by nucleic acid detection techniques. Treatment of acute CHIKV is mainly symptomatic, with analgesics, non-steroidal anti-inflammatory agents (NSAIDs), and low-dose steroids. No vaccines or anti-viral medicines have been approved for clinical therapy in CHIKV as yet. Hydroxychloroquine and methotrexate have been used in chronic CHIKV infection with variable success.
Similar content being viewed by others
Introduction
Chikungunya virus, belonging to the family Togaviridae and genus Alphavirus, is an arthritogenic virus, first isolated in the southern province of Tanzania in 1952–1953 [1]. Initial infectious outbreaks were locally confined and were of low intensity. Ever since its re-emergence in 2000 after a hiatus of 39 years, the virus has evolved into a major public health concern globally. It has transcended international geographic boundaries with frequent epidemics, affecting millions of people mainly in countries within African and Asian subcontinents and Reunion islands in the Indian Ocean [2]. The first local cases of Chikungunya infection (CHIKV) in the Americas were reported in 2013 [3•]. Since then, it has spread swiftly across the continent, affecting about one million people in Latin America, and cases are being reported from 36 states in the USA [4]. CHIKV is characterized by fever, headache, myalgia, rash, and polyarthritis. Generally, these episodes are self-limiting and resolve within a few weeks. However, in 10–60% of cases, musculoskeletal symptoms may persist for up to 3–5 years [5••, 6]. This wide variation may be because of geographical diversity, lack of uniform methodology in detection, and incomplete and heterogeneous follow-up period in published reports. A recent meta-analysis of 18 studies, involving 5702 patients around the world, has described the pooled prevalence of CHIKV which related all chronic inflammatory rheumatism and chronic arthritis to be 40.22 and 14%, respectively [7].
Over the past decade, there has been profound interest in understanding the etio-pathogenesis and management strategies for chronic CHIKV across the globe. This review aims at emphasizing the epidemiology, pathogenesis, clinical features, and management aspects of CHIKV, focusing on rheumatic symptoms.
Epidemiology
Global Scene
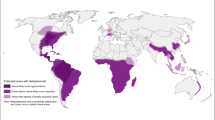
CHIKV is currently geographically distributed in over 60 countries, remaining endemic in Africa, India, and several Southeast Asian countries as well as Latin America (Fig. 1). Four lineages, distinguished by genotypic and antigenic characteristics, have been identified: The West African lineage, the Asian lineage, the East, Central, and South African (ECSA) lineage, and the Indian Ocean (IO) lineage, a monophyletic descendant from the ECSA [8, 9]. Sporadic and intermittent outbreaks of CHIKV are being reported from Asia, Africa, and Reunion islands in the Indian Ocean following the initial isolation 40 years ago. Global attention to this pathogen came later in 2005 following a large epidemic in the coast of Kenya. Massive sequential outbreaks by the IO CHIKV lineage ravaged the islands of the southwest Indian Ocean and the Indian subcontinent in 2005–2006, affecting millions [10•, 11]. The precise reason for resurgence after a long hiatus is still an enigma. Mutated CHIKV genome, adaptations by the vector, reduced herd immunity, and frequent travel are the speculated explanations. Cases attributed to travel were reported for the first time in various parts of Europe, Americas, Canada, and Australia [12]. Indigenous cases were also reported from Italy (2007), France (2010), and the Caribbean islands (2013) [13,14,15].
Vectors
The highly anthropophilic Aedes aegypti, a vector closely associated with humans and widely prevalent in the tropical and subtropical nations, has been the primary vector in CHIKV transmission [16]. After the 2006 outbreak, the Asian tiger mosquito “Aedes albopictus” surfaced as a major vector, responsible for perpetuating the epidemic in regions lacking the typical vector [17]. This species, widely distributed in the western hemisphere, can adapt to different climatic conditions and colonize new geographical locations, making it a competent vector for efficient viral transmission [18]. During the Indian Ocean epidemic, a mutation found in the E1 protein of CHIKV (A226V) permitted enhanced vector specificity, virus uptake, replication, and transmission by Ae. albopictus [19]. Few other Aedes species, Culex, and Anopheles stephensi have also been reported to transmit CHIKV [20, 21].
Reservoirs and Modes of Transmission
During the inter-epidemic period, monkeys, rodents, and birds serve as reservoir hosts for the virus. Humans replace them as the hosts during epidemics. Periodicity has been noted with occurrence of disease in the community, which probably may be due to the oscillations in monkey herd immunity [22]. The established modes of CHIKV transmission include enzootic, urban, nosocomial, and vertical transmissions. Transmission by needle-stick injury, blood donations, and corneal grafts has been reported [23,24,25]. Vertical transmissions in an estimated 48.7% of population were described during the epidemic in Reunion islands [26].
Pathogenesis
Evidence From Animal Studies
Mice and primate models have been used for studying CHIKV. Symptoms in primates following CHIKV resemble human infection. Genetic background, age, route, and site of CHIKV transmission significantly affect clinical presentation, development of musculoskeletal involvement, and immune profile in animal models of CHIKV [27]. Clinical presentations in animals depend on the initial viral load [28]. CHIKV infection results in dissemination of virus to various vital organs like liver, spleen, lymph nodes, skin, muscles, bones, and central nervous system [29, 30]. Entry of virus into these tissues causes infiltration of innate immune cells-monocytes, macrophages, neutrophils, natural killer cells, and adaptive immune cells of CD4 as well as CD8 T cell lineage [28, 30]. The virus survives and replicates for longer periods in these targeted organs and in macrophages of infected animals, even when the viral load is low in blood. Infiltration of immune cells results in pathological musculoskeletal features such as inflammatory arthritis, chronic active tenosynovitis, and myositis during acute CHIKV infection [31•]. In a recent study, Goupil et al. used histological and micro-computed tomography techniques to view the clinical progression of musculoskeletal changes in C57BL/6J mice infected with CHIK virus. Necrosis of skeletal muscles, extensive peri-osteitis, lympho-plasmacytic infiltration of synovium, and cartilage necrosis in joints were observed 21 days following infection [32].
Role of Type I Interferon
Cells infected with CHIKV induce secretion of interferon (IFN) α/β. IFN α/β produced in local infected sites by non-hematopoietic cells controls dissemination of virus to other organs [33]. Macrophages act as hosts for CHIK virus survival and also mediate inflammation in joints [27].
Mediators of Inflammation—Chemokines and Cytokines (Table 1)
Increased production of pro-inflammatory cytokines by infiltrating myeloid cells has been shown in CHIKV. Tumor necrosis factor (TNF), IFN-α, IL-6, IFN-γ, and monocyte chemoattractant protein-1 (MCP-1) were major cytokines and chemokines secreted by macrophages [29, 34•, 35]. A cytokine secretion pattern observed a trend towards Th1-type response in mouse models [36].
Role of Immune Cells in Pathogenesis
Neutrophils which are recruited at the site of infection exceed any other immune cells in the joints [36]. Natural killer (NK) cells play an important role in the acute phase of infection through its cytotoxic activity by secreting IFNγ and granzyme B [27, 36]. The role of T cells in pathogenesis of CHIKV infection is evident in RAG2−/−, CD4−/−, and CD8−/− mouse models. CHIKV-infected RAG2−/− mice harbor persistent viral load in blood, and no inflammation was observed in joints. A similar observation was noted in CD4−/− but not in CD8−/− mice [37]. B cells have been shown to enhance natural antibody response to CHIKV and control viral load in circulation [38]. The anti-CHIKV IgM antibody was detected in plasma before IgG and peaked between 4 and 6 weeks post infection. After 6 weeks, IgM levels decreased, whereas IgG (exclusively of IgG3 isotype) levels increased gradually. Kam et al demonstrated delayed mounting of IgG3 response in some patients with low viral load during the active phase. These patients complained of persistent arthralgia in the chronic phase of disease [39].
Innate and Adaptive Immunity in CHIKV
Innate immune cells, including plasmacytoid dendritic cells and NK cells, exhibit robust activation from day 1 of infection, and adaptive cells subsequently contribute to immune responses in circulation for elimination of virus [40••]. Terminally differentiated NK cells expressing high levels of NKG2C and CD57 have been described in circulation of CHIKV-affected individuals. This increased level of NKG2C+ CD57+ NK cells was transient and mostly confined to the early phase of infection. However, in some convalescent patients, the NK cells were increased more than 30 days after infection; eventually on follow-up, these patients developed chronic arthralgia [41].
CHIKV Arthritis Resembles Rheumatoid Arthritis
CHIKV-infected human osteoblasts show increased expression of IL-6 and RANKL and decreased expression of osteoprotegerin (OPG), a decoy receptor for RANKL [42]. Altered RANKL/OPG ratio levels in synovium lead to enhanced osteoclastogenesis [43•]. These changes are similar to the pathogenesis of rheumatoid arthritis. CHIK viruses persist in synovial fibroblasts and osteoblasts of infected individuals. Upon induction, fibroblasts produce IL-6 and MCP-1. Monocytes or macrophages are recruited to the site of infection. Virus replicates in macrophages, which in turn secrete pro-inflammatory cytokines IL-1β, TNF-α, and IL-17. These pro-inflammatory cytokines and RANKL induce osteoclastogenesis, bone loss, and erosions in chronically infected individuals [44].
Clinical Features
CHIKV can affect any age group of either sex. The incubation period varies between 3 to 7 days (range 1 to 12 days) [45, 46]. Acute chikungunya fever presents most commonly as fever (92%), arthralgia (87%), backache (67%), and headache (62%). Other less common clinical manifestations include skin manifestations like rash, oral ulcer, hyperpigmentation, and exfoliative dermatitis. Rarely, photophobia, retro-orbital pain, vomiting, diarrhea, and meningo-encephalitic features are seen, especially in the pediatric age group [4]. Fever, lasting for 3–5 days is low to high grade and can be associated with chills and rigors. Polyarthralgia begins 2 to 5 days after the onset of fever and can involve multiple joints. Arthralgia is symmetrical in most of the cases, predominantly involving distal joints, which include hands (50 to 76%), wrists (29 to 81%), and ankles (41 to 68%) [7, 47]. Skin manifestations can be seen among 40–75% of patients. Most often, maculo-papular rash is seen, predominantly affecting the limbs and the trunk, but can involve face as well [48]. Physical examination can reveal peri-articular edema or swelling, which is seen in 32–95% of cases. Apart from this, lymphadenopathy (9–41%) and conjunctivitis have also been reported [49•, 50].
Data on the effect of CHIKV on pregnancy outcomes and its effect on infants born to infected mothers is scarce. Torres et al. reviewed pregnancy outcomes during the 2014–15 epidemic in Latin America. Vertical transmission was observed in 27.7 to 48.29% of patients. Case fatality rate was 5.3%. Commonest symptoms included fever, irritability, meningoencephalitis, rash and bullous dermatitis. Serious complications including myocarditis, seizures and acute respiratory failure were documented [51•].
In general, CHIKV has a good prognosis and resolves without any sequelae. However, in a certain group of patients it causes chronic rheumatologic manifestations. Some of the risk factors for developing chronic infection, based on retrospective and small prospective studies, include female gender, older age, co-morbid conditions, and severity of symptoms at onset of infection [5••, 52]. Gerardin et al. have noted the presence of two or more co-morbidities like hypertension, diabetes, osteoarthritis and dyslipidemia to be associated with higher chances of developing chronic infection [53]. Various rheumatic manifestations including polyarthralgia or arthritis, multiple tendonitis, tenosynovitis, enthesopathies, carpal tunnel syndrome, classical rheumatoid arthritis like course, cryoglobulinemia, and psoriatic arthritis have all been observed as post-infectious sequelae [5,55,••, 6, 54–56]. Polyarthralgias or arthritis frequently involves fingers, wrists, knees, ankles, and toes. Rarely, elbows, shoulders, neck, sacroiliacs, and hip joints are also affected [5••, 52, 53]. Studies have indicated the close association of CHIKV with autoimmunity in susceptible individuals [57, 58]. The clinical manifestations of CHIKV may resemble those of other rheumatologic diseases like RA, seronegative inflammatory arthropathies, and systemic lupus erythematosus [59, 60]. In a study from India, 36% of patients with persistent arthralgia 10 months following CHIKV infection met the American College of Rheumatology (ACR) classification for rheumatoid arthritis [58]. CHIKV has been documented to be a precipitating event for development of RA in many studies [6, 55, 59, 61]. These studies have also documented presence of rheumatoid factor (RF) and anti-citrullinated protein (CCP) antibodies, joint space narrowing, effusions and bone erosions.
The frequency of chronic rheumatic symptoms during the post-CHIKV infection period varied in different studies. In a 10-month prospective study from India on 203 CHIKV-infected patients, it was found that 46% experienced persistent joint pain after acute infection [59]. Schilte et al. described 69% patients in their cohort developing persistent arthralgia, 90% reporting symmetric joint involvement, 63% complaining of associated local joint swelling, and 39% suffering from chronic myalgia [5••].
Diagnosis
Currently available tests for diagnosis of CHIKV include viral culture, detection of viral antigen, viral nucleic acid and anti-CHIKV IgM and IgG antibodies. Viral culture and isolation from blood cells of infected patients is the gold standard technique. However, this technique is cumbersome and usually done only in advanced centers, with biosafety level (BSL) 3. Anti-CHIKV IgMs are elevated in the blood of infected patients after 10 days of infection and remain elevated for 3–6 months [62]. Anti-CHIKV IgG is detectable only after 2 weeks of infection and remains elevated for 6 months. Capture ELISA and immune-chromatographic tests are two common techniques for estimation of CHIKV titers in blood. Early diagnosis of CHIKV is possible by nucleic acid detection techniques such as reverse transcription polymerase chain reaction (RT-PCR) and real-time loop-mediated isothermal amplification (RT-LAMP) methods [63]. In low resource centers the difficulties in using RT-PCR can be overcome by RT-LAMP, which does not require a PCR instrument as the assay is carried out in a water bath [64].
Role of Biomarkers
Few studies have reported the role of cytokines and chemokines like IL-1β, IL-6, IL18, MIF, RANTES, CXCL9, CCL2, and CXCL10 [65, 66]. Because of the heterogeneity of these biomarkers in different populations and lack of validation, these tests have not been used in regular clinical practice.
Management of CHIKV
Treatment of acute CHIKV is mainly symptomatic, with analgesics, antipyretics and non-steroidal anti-inflammatory agents (NSAIDs). So far, no vaccines or anti-viral medicines have been approved for clinical therapy in CHIKV.
Chloroquine phosphate (CQ) has long been reported to be effective in the treatment of chronic CHIKV arthritis [67]. In a community intervention trial of chloroquine in 509 CHIKV-infected patients during the 2006 CHIK epidemic in India, 70 patients with early persistent musculoskeletal pain and arthritis were randomized into a 24-week, two-arm, parallel efficacy trial of CQ (250 mg/day) (n = 38) and meloxicam (7.5 mg/day) (n = 32). There was no significant efficacy difference between the meloxicam and CQ groups. Though the patients reported good improvement in symptoms, it did not reach statistical significance [68]. In a very recent randomized controlled trial on 72 patients with persistent post-CHIKV arthritis (>1 year after infection), the efficacy of a combination of disease-modifying anti-rheumatic drugs (DMARD), namely, methotrexate, sulfasalazine, and hydroxychloroquine (HCQ), was compared with HCQ monotherapy. Both groups received oral steroids for 6 weeks. Combination DMARDs was found to be superior to HCQ monotherapy [69•]. Ganu et al. studied 16 patients with persistent arthritis in spite of treatment with NSAIDs and HCQ for 3 months and observed good response in 71.4 and 12.5% among those treated with sulfasalazine with and without methotrexate, respectively [70].
Ravichandran et al. reported the efficacy of ribavirin 200 mg twice daily for 1 week in 10 patients with chronic CHIKV, as compared to placebo. All patients in both the groups reported reduction of pain. Reduction in swelling was reported by 8 and 6 out of 10 in the antiviral and placebo groups, respectively [71]. Rulli et al. have reported the efficacy of bindarit, an inhibitor of MCP-1 in the treatment of chronic CHIKV [34•]. Inhibitors targeting the mannose-binding lectin (MBL) pathway of the complement system may also be useful in the treatment of alphavirus-induced arthritis/myositis [72]. Experiments reported an autophagy-inducing peptide, beclin, to be effective against CHIKV in cell culture and in mice [73].
The French Infectious Disease Society has come up with recommendations to aid clinicians in diagnosis, assessment, and management of acute, post-acute, and chronic patients. Analgesics, NSAIDs, and low-dose steroids have been mentioned as initial therapeutic options for post-acute and chronic CHIKV infections. Methotrexate can be considered for chronic patients (3 months post-infection) not responding to the initial therapy [74•].
In the authors’ experience, a short course of low-dose steroid (<0.5 mg/kg per day of deflazacort) with rapid weekly tapering and withdrawal by 4 to 6 weeks is effective and safe for acute CHIKV infection (unpublished data). Steroids need to be started at least a week after NSAID intake once the acute viremic period is over. Hydroxychloroquine probably does not have much effect in the acute phase, as it takes few weeks to act; by then, the arthritis tends to settle in majority. However, all DMARDs have COX-2-inhibiting actions and hence, may act like NSAIDs initially, thus giving relief before the arthritis self-limits.
Experimental Drugs
Wang et al. looked at the potential of anti-CHIKV drugs in vitro through a high-throughput screening system based on the CHIKV 26S-mediated insect cell fusion inhibition assay. Four compounds were identified, of which niclosamide and nitazoxanide were found to inhibit CHIK virus entry and transmission. This may prove to be a future target for therapy [75]. Suramin, a competitive inhibitor of glycosaminoglycans and heparin, has been shown to have anti-CHIKV action [76, 77]. Kuo et al. demonstrated the efficacy of suramin in reducing viral loads substantially, mitigating acute foot lesions and restoring cartilage integrity in a C57BL/6 mouse model infected with CHIK virus [78]. These results need to be replicated in clinical trials. Intranasal use of a single dose of mDEF201 (adenovirus-vectored IFN-α) prophylactically administered 21 days to 24 h prior to infection significantly reduced synovitis in an experimental mouse model for CHIKV arthritis [79].
Control of Infection
Emphasis should be on early detection of suspected human cases and improved environmental control measures against the vectors. The primary target would be to curtail the urban viral transmission by avoiding mosquito bites. Infection control can be emphasized at two levels—individual and community.
Individual Level
The adult female mosquito bites during daytime. Physical protection measures including full-sleeved clothing, application of mosquito repellent over the exposed body parts, use of permethrin-treated clothing, insecticide-treated bednets, and wire mesh/nets on doors and windows should be encouraged. The Center for Disease Control and Prevention (CDC) and the World Health Organization (WHO) suggest use of 30–50% N,N-diethyl-m-toluamide (DEET), picaridin, IR3535, and oil of lemon eucalyptus or para-menthane-diol as repellents providing long-lasting protection [80, 81]
Community Level
The foremost step would be to target the mosquito breeding grounds. Extensive regional awareness and effective national control programs should be employed. Aedes mosquito breeds in stored/stagnant fresh water in domestic settings such as flower vases and water storage containers, and in peri-domestic areas such as construction sites, coconut shells, unused abandoned tires, plastic, and metal cans. Such breeding sites must be destroyed and treated with insecticide [80]. Vector control can be done through anti-adult and anti-larval control of mosquitoes. The WHO recommends applying appropriate products in intervals that are shorter than the incubation period of pathogen in the vector and in accordance with the generation cycle of vector. Adult mosquitoes can be controlled by the use of chemical insecticides (pyrethrum extract, malathion) for indoor spray, ultra low volume spray, and thermal fogging [82]. However, larval control has been found to be more economical and provides sustainable control by eliminating the source of newly emergent adult mosquitoes [83].
Challenges for Future
The rapid spread of CHIKV involving virgin territories over the last few years has earned renewed interest in the pathogenesis, preventive, and surveillance strategies for this arthritogenic virus. Gaps in the understanding of viral replication and processes contributing to chronic arthritis still exist, which hinder optimal drug discovery. Vaccines are still in the infancy stage and need to be tested in clinical trials to prove their efficacy. Patient surveillance and vector control methods need significant improvement. Consensus on uniform case definitions and evidence-based clinical management guidelines continue to be unmet needs in this area. A global virus network task force on CHIKV was constituted in 2014, outlining clear future plans [84].
Conclusion
CHIKV continues to be a global public health, clinical, and economic challenge. The rheumatologic injury following CHKV seems more often to be immune mediated than direct viral injury. A short course of steroid following the acute viremic phase should suffice to treat the acute complications. Though DMARDs have been tried for this mostly self-limiting disease, use of these agents should be restricted to cases with a more chronic course. Better insight into the factors attributing to chronic complications is warranted for attaining optimal solution to this menace.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Robinson MC. An epidemic of virus disease in Southern Province, Tanganyika Territory in 1952–53—1: clinical features. Trans R Soc Trop Med Hyg. 1955;49:28.
Thiberville SD, Moyen N, Dupuis-Maguiraga L, et al. Chikungunya fever: epidemiology, clinical syndrome, pathogenesis and therapy. Antivir Res. 2013;99:345–70.
• Pineda C, Munoz-Louis R, Caballero-Uribe CV, Viasus D. Chikungunya in the region of the Americas. A challenge for rheumatologists and health care systems. Clin Rheumatol. 2016;35:2381–5. A comprehensive review on the clinical features of CHIKV in the American continent, and also emphasizing on the challenges to the healthcare system following the epidemic.
Centers for Disease Control and Prevention: Chikungunya virus http://www.cdc.gov/chikungunya/geo/united-states.html (Accessed Oct 12, 2016)
•• Schilte C, Staikowsky F, Couderc T, et al. Chikungunya virus-associated long-term arthralgia: a 36 month prospective longitudinal study. PLoS Negl Trop Dis. 2013;7, e2137. doi:10.1371/journal.pntd.0002137. A 3-year post-infection study of patients affected by CHIKV during the 2006 La Reunion island epidemic in France, describing 60% of the patients complaining of arthralgia during the follow up period, emphasizing the relatively high frequency of chronic complications following acute infection.
Mathew AJ, Goyal V, George E, Thekkemuriyil DV, Jayakumar B, Chopra A. Rheumatic-musculoskeletal pain and disorders in a naïve group of individuals 15 months following a Chikungunya viral epidemic in south India: a population based observational study. Int J Clin Pract. 2011;65:1306–12.
Rodriguez-Morales AJ, Cardona-Ospina JA, Urbano-Garzon SF, Hurtado-Zapata JS. Prevalence of post-chikungunya infection chronic inflammatory arthritis: a systemic review and meta-analysis. Arthritis Care Res. 2016;68:1849–58.
Cavrini F, Gaibani P, Pierro AM, Rossini G, Landini MP, Sambri V. Chikungunya: an emerging and spreading arthropod-borne viral disease. J Infect Dev Ctries. 2009;3:744–52.
Vega-Rúa A, Zouache K, Girod R, Failloux A-B, Lourenço-de-Oliveira R. High level of vector competence of Aedes aegypti and Aedes albopictus from ten American countries as a crucial factor in the spread of chikungunya virus. J Virol. 2014;88:6294–306.
• Ng LC, Hapuarachchi HC. Tracing the path of Chikungunya virus-evolution and adaptation. Infect Genet Evol. 2010;10:876–85. In this explicit review, the authors explain the biologically important E1-A226V and E2-I211T mutations undergone by CHIK virus, which resulted in the change in epidemiology of infection as a result of rapid adaptation to the Aedes albopictus vector. The role of environmental factors and preemptive methods for combating the disease are emphasized.
Lahariya C, Pradhan SK. Emergence of chikungunya virus in Indian subcontinent after 32 years: a review. J Vector Borne Dis. 2006;43:151–60.
Savini H, Gautret P, Gaudart J, et al. Travel-associated diseases, Indian Ocean Islands, 1997–2010. Emerg Infect Dis. 2013;19:1297–301.
Rezza G, Nicoletti L, Angelini R, et al. Infection with chikungunya virus in Italy: an outbreak in a temperate region. Lancet Lond Engl. 2007;370:1840–6.
Gould EA, Gallian P, De Lamballerie X, Charrel RN. First cases of autochthonous dengue fever and chikungunya fever in France: from bad dream to reality! Clin Microbiol Infect. 2010;16:1702–4.
Horcada ML, Díaz-Calderón C, Garrido L. Chikungunya fever. Rheumatic manifestations of an emerging disease in Europe. Reumatol Clin. 2015;11:161–4.
Tsetsarkin KA, Chen R, Sherman MB, Weaver SC. Chikungunya virus: evolution and genetic determinants of emergence. Curr Opin Virol. 2011;1:310–7.
Reiter P, Fontenille D, Paupy C. Aedes albopictus as an epidemic vector of chikungunya virus: another emerging problem? Lancet Infect Dis. 2006;6:463–4.
Powers AM, Logue CH. Changing patterns of chikungunya virus: re-emergence of a zoonotic arbovirus. J Gen Virol. 2007;88:2363–77.
Tsetsarkin KA, Vanlandingham DL, McGee CE, Higgs S. A single mutation in chikungunya virus affects vector specificity and epidemic potential. PLoS Pathog. 2007;3, e201.
Yadav P, Gokhale MD, Barde PV, Singh DK, Mishra AC, Mourya DT. Experimental transmission of Chikungunya virus by Anopheles stephensi mosquitoes. Acta Virol. 2003;47:45–7.
Lo Presti A, Lai A, Cella E, Zehender G, Ciccozzi M. Chikungunya virus, epidemiology, clinics and phylogenesis: a review. Asian Pac J Trop Med. 2014;7:925–32.
Diallo M, Thonnon J, Traore-Lamizana M, Fontenille D. Vectors of Chikungunya virus in Senegal: current data and transmission cycles. Am J Trop Med Hyg. 1999;60:281–6.
Parola P, de Lamballerie X, Jourdan J, et al. Novel chikungunya virus variant in travelers returning from Indian Ocean islands. Emerg Infect Dis. 2006;12:1493–9.
Simmons G, Brès V, Lu K, et al. High incidence of chikungunya virus and frequency of viremic blood donations during epidemic, Puerto Rico, USA, 2014. Emerg Infect Dis. 2016;22:1221–8.
Couderc T, Gangneux N, Chrétien F, et al. Chikungunya virus infection of corneal grafts. J Infect Dis. 2012;206:851–9.
Gérardin P, Barau G, Michault A, et al. Multidisciplinary prospective study of mother-to-child chikungunya virus infections on the island of La Réunion. PLoS Med. 2008;5, e60.
Teo TH, Lum FM, Lee WW, Ng LF. Mouse models for Chikungunya virus: deciphering immune mechanisms responsible for disease and pathology. Immunol Res. 2012;53:136–47.
Labadie K, Larcher T, Joubert C, et al. Chikungunya disease in nonhuman primates involves long-term viral persistence in macrophages. J Clin Invest. 2010;120:894–906.
Gardner J, Anraku I, Le TT, et al. Chikungunya virus arthritis in adult wild-type mice. J Virol. 2010;84:8021–32.
Morrison TE, Oko L, Montogomery SA, et al. A mouse model of Chikungunya virus-induced musculoskeletal inflammatory disease: evidence of arthritis, tenosynovitis, myositis and persistence. Am J Pathol. 2011;178:32–40.
• Couderc T, Chretien F, Schilte C, et al. A mouse model for Chikungunya: young age and inefficient type-I interferon signaling are risk factors for severe disease. PLoS Pathog. 2008;4, e29. One of the initial bench side animal model research in CHIK virus, highlighting the role of type-I interferon signaling in severe disease.
Goupil BA, McNulty MA, Martin MJ, McCracken MK, Christofferson RC, Mores CN. Novel lessons of bones and joints associated with Chikungunya virus infection in two mouse models of disease: new insights into disease pathogenesis. PLoS One. 2016;16, e0155243.
Schilte C, Couderc T, Chretien F, et al. Type I IFN controls chikungunya virus via its action on nonhematopoietic cells. J Exp Med. 2010;207:429–42.
• Rulli NE, Rolph MS, Srikiatkhachorn A, Anantapreecha S, Guglielmotti A, Mahalingam S. Protection from arthritis and myositis in a mouse model of acute chikungunya virus disease by bindarit, an inhibitor of monocyte chemotactic protein-1 synthesis. J Infect Dis. 2011;204:1026–30. In this study, the authors have demonstrated the presence of cytokines in a mouse model of CHIKV arthritis and myositis. The role of chemokine MCP-1 and its inhibitor bindarit ameliorating the disease in mice is also studied. This has a potential for a future drug target.
Venugopalan A, Ghorpade RP, Chopra A. Cytokines in acute Chikungunya. PLoS One. 2014;24, e111305.
Teo TH, Her Z, Tan JJ, et al. Caribbean and La Reunion Chikungunya virus isolates differ in their capacity to induce proinflammatory Th1 and NK cell responses and acute joint pathology. J Virol. 2015;89:7955–69.
Teo TH, Lum FM, Claser C, et al. A pathogenic role of CD4+ T cells during Chikungunya virus infection in mice. J Immunol. 2013;190:259–69.
Lum FM, Teo TH, Lee WW, Kam YW, Renia L, Ng LF. An essential role of antibodies in the control of Chikungunya virus infection. J Immunol. 2013;190:6295–302.
Kam YW, Lee WW, Simarmata D, et al. Longitudinal analysis of the human antibody response to Chikungunya virus infection: implications for serodiagnosis and vaccine development. J Virol. 2012;86:13005–15.
•• Hoarau JJ, Jaffar Bandjee MC, Krejbich Trotot P, et al. Persistent chronic inflammation and infection by Chikungunya arthritogenic alphavirus in spite of a robust host immune response. J Immunol. 2010;184:5914–27. A well-designed study analyzing the factors responsible for chronic complications in the La Reunion island epidemic in 2006. In a prospective cohort of 49 patients, categorized into two distinct groups—recovered or chronic, 12 months post-infection, the authors describe contrasting cellular and molecular events, focusing on drug targets.
Petitdemange C, Wauquier N, Devilliers H, et al. Longitudinal analysis of natural killer cells in dengue virus-infected patients in comparison to chikungunya and chikungunya/dengue virus-infected patients. PLoS Negl Trop Dis. 2016;10, e0004499.
Noret M, Herrero L, Rulli N, et al. Interleukin 6, RANKL, and osteoprotegerin expression by chikungunya virus-infected human osteoblasts. J Infect Dis. 2012;206:455–7.
• Chen W, Foo SS, Rulli NE, et al. Arthritogenic alphaviral infection pertubs osteoblast function and triggers pathologic bone loss. Proc Natl Acad Sci U S A. 2014;111:6040–5. A study describing the pathogenesis of bone loss caused by alphaviruses. In Ross River virus-infected human osteoblasts, the authors demonstrated higher levels of inflammatory cytokines and disrupted ratio of RANK/OPG.
Ryman KD, Klimstra WB. Closing the gap between viral and noninfectious arthritis. Proc Natl Acad Sci U S A. 2014;111:5767–8.
Simon F, Savini H, Parola P. Chikungunya: a paradigm of emergence and globalization of vector-borne diseases. Med Clin North Am. 2008;92:1323–43.
Pialoux G, Gaüzère B-A, Jauréguiberry S, Strobel M. Chikungunya, an epidemic arbovirosis. Lancet Infect Dis. 2007;7:319–27.
Parola P, de Lamballerie X, Jourdan J, Rovery C, Vaillant V, Minodier P, et al. Novel Chikungunya virus variant in travelers returning from Indian Ocean islands. Emerg Infect Dis. 2006;12:1493–9.
Taubitz W, Cramer JP, Kapaun A, Pfeffer M, Drosten C, Dobler G, et al. Chikungunya fever in travelers: clinical presentation and course. Clin Infect Dis. 2007;45:e1–4.
• Borgherini G, Poubeau P, Staikowsky F, Lory M, Le Moullec N, Becquart JP, et al. Outbreak of chikungunya on Reunion Island: early clinical and laboratory features in 157 adult patients. Clin Infect Dis. 2007;44:1401–7. In one of the early analyses of the 2005–06 La Reunion island epidemics in France, the authors describe the demography, clinical features, and laboratory parameters of 157 affected patients.
Mahendradas P, Avadhani K, Shetty R. Chikungunya and the eye: a review. J Ophthalmic Inflamm Infect. 2013;3:35.
• Torres JR, Falleiros-Arlant LH, Duenas L, Pleitez-Nevarrete J, Salgado DM, Castillo JB. Congenintal and perinatal complications of chikungunya fever: a Latin American experience. Int J Infect Dis. 2016;51:85–8. A prospective study of 169 symptomatic newborns in Latin America during the 2014–2015 CHIKV epidemic. Clinical features, complications, and vertical transmission rates are described. The authors have also analyzed pregnancy outcomes in symptomatic infected mothers.
Win MK, Chow A, Dimatatac F, Go CJ, Leo YS. Chikungunya fever in Singapore: acute clinical and laboratory features, and factors associated with persistent arthralgia. J Clin Virol. 2010;49:111–4.
Gérardin P, Fianu A, Michault A, et al. Predictors of Chikungunya rheumatism: a prognostic survey ancillary to the TELECHIK cohort study. Arthritis Res Ther. 2013;15:R9.
Ribéra A, Degasne I, Jaffar Bandjee MC, Gasque P. Chronic rheumatic manifestations following chikungunya virus infection: clinical description and therapeutic considerations. Med Trop. 2012;72:83–5.
Bouquillard E, Combe B. A report of 21 cases of rheumatoid arthritis following chikungunya fever. A mean follow-up of two years. Joint Bone Spine. 2009;76:654–7.
Mathew AJ, Chopra A, Antony J, Anandan N, Nair GS. Increased incidence of soft tissue rheumatism following Chikungunya viral epidemic: incidental or cause? Ann Rheum Dis. 2011;70(Suppl3):567.
Nelson P, Rylance P, Roden D, Trela M, Tugnet N. Viruses as potential pathogenic agents in systemic lupus erythematosus. Lupus. 2014;23:596–605.
Vaughan JH. Viruses and autoimmune disease. J Rheumatol. 1996;23:1831–3.
Manimunda SP, Vijayachari P, Uppoor R, et al. Clinical progression of chikungunya fever during acute and chronic arthritic stages and the changes in joint morphology as revealed by imaging. Trans R Soc Trop Med Hyg. 2010;104:392–99.
Essackjee K, Goorah S, Ramchurn SK, Cheeneebash J, Walker-Bone K. Prevalence of and risk factors for chronic arthralgia and rheumatoid-like polyarthritis more than 2 years after infection with chikungunya virus. Postgrad Med J. 2013;89:440–7.
Chopra A, Anuradha V, Lagoo-Joshi V, Kunjir V, Salvi S, Saluja M. Chikungunya virus aches and pains: an emerging challenge. Arthritis Rheum. 2008;58:2921–2.
Prince HE, Seaton BL, Matud JL, Batterman HJ. Chikungunya virus RNA and antibody testing at a national reference laboratory since the emergence of Chikungunya virus in the Americas. Clin Vaccine Immunol. 2015;22:291–7.
Patel P, Abd El Wahed A, Faye O, et al. A field-deployable reverse transcription recombinase polymerase amplification assay for rapid detection of the Chikungunya virus. PLoS Negl Trop Dis. 2016;10:e0004953.
Parida MM, Santhosh SR, Dash PK, et al. Rapid and real-time detection of Chikungunya virus by reverse transcription loop-mediated isothermal amplification assay. J Clin Microbiol. 2007;45:351–7.
Ng LFP, Chow A, Sun J, Dwek DJC, et al. IL-1β, IL-6 and RANTES as biomarkers of Chikungunya severity. PLoS One. 2009;4, e4261.
Chirathworn C, Poovorawan Y, Lertmaharit S, Wuttirattanakowit N. Cytokine levels in patients with Chikungunya virus infection. Asian Pac J Trop Med. 2013;6:631–4.
Brighton SW. Chloroquine phosphate treatment of chronic Chikungunya arthritis. An open pilot study. South Afr Med J. 1984;66:217–8.
Chopra A, Saluja M, Venugopalan A. Effectiveness of chloroquine and inflammatory cytokine response in patients with early persistent musculoskeletal pain and arthritis following chikungunya virus infection. Arthritis Rheumatol. 2014;66:319–26.
• Ravindran V, Alias G. Efficacy of combination DMARD therapy vs hydroxychloroquine monotherapy in chronic persistent chikungunya arthritis: a 24-week randomized controlled open label study. Clin Rheumatol 2016. A randomized control trial of combination DMARDs vs HCQ monotherapy in 72 patients with chronic CHIKV rheumatism, showing better efficacy with combination therapy
Ganu MA, Ganu AS. Post-chikungunya chronic arthritis—our experience with DMARDs over two year follow up. J Assoc Physicians India. 2011;59:83–6.
Ravichandran R, Manian M. Ribavirin therapy for Chikungunya arthritis. J Infect Dev Ctries. 2008;2:140–2.
Gunn BM, Morrison TE, Whitmore AC, et al. Mannose binding lectin is required for alphavirus-induced arthritis/myositis. PLoS Pathog. 2012;8, e1002586.
Shoji-Kawata S, Sumpter R, Leveno M, Campbell GR, Zou Z, Kinch L, et al. Identification of a candidate therapeutic autophagy-inducing peptide. Nature. 2013;494:201–6.
• Simon F, Javelle E, Cabie A, et al. French guidelines for management of chikungunya (acute and persistent presentations), November 2014. Med Mal Infect. 2015;45:243–63. First published guidelines for diagnosis, assessment, and management of acute, post-acute, and chronic CHIKV.
Wang YM, Lu JW, Lin CC, et al. Antiviral activities of niclosamide and nitazoxanide against chikungunya virus entry and transmission. Antiviral Res 2016. doi: 10.1016/j.antiviral.2016.10.003
Ho YJ, Wang YM, Lu JW, et al. Suramin inhibits Chikungunya virus entry and transmission. PLoS One. 2015;10, e0133511. doi:10.1371/journal.pone.0133511.
Henβ L, Beck S, Weidner T, et al. Suramin is a potent inhibitor of Chikungunya and Ebola virus cell entry. Virol J. 2016;13:149.
Kuo SC, Wang YM, Ho YJ, et al. Suramin treatment reduces chikungunya pathogenesis in mice. Antiviral Res. 2016;134:89–96.
Dagley A, Ennis J, Turner JD, et al. Protection against Chikungunya virus induced arthralgia following prophylactic treatment with adenovirus vectored interferon (mDEF201). Antiviral Res. 2014;108:1–9.
Prevention | Chikungunya virus | CDC [Internet]. [cited 2016 Jun 23]. Available from: http://www.cdc.gov/chikungunya/prevention/
Protection against Mosquitoes, Ticks, & Other Arthropods - Chapter 2–2016 Yellow Book | Travelers’ Health | CDC [Internet]. [cited 2016 Jun 23]. Available from: http://wwwnc.cdc.gov/travel/yellowbook/2016/the-pre-travel-consultation/protection-against-mosquitoes-ticks-other-arthropods
WHO | Control strategies [Internet]. WHO. [cited 2016 Jun 23]. Available from: http://www.who.int/denguecontrol/control_strategies/en/
Centers for Disease Control and Prevention (U.S.), National Center for Emerging and Zoonotic Infectious Diseases (U.S.), Pan American Health Organization. Preparedness and response for Chikungunya virus introduction in the Americas [Internet]. Washington, DC: Pan American Health Organization : CDC, Center for Disease Control and Prevention; 2011 [cited 2016 Jun 26]. Available from: http://new.paho.org/hq/index.php?option=com_docman&task=doc_download&gid=16984&Itemid
McSweegan E, Weaver SC, Lecuit M, Frieman M, Morrison TE, Hrynkow S. The global virus network: challenging chikungunya. Antiviral Res. 2015;120:147–52.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Drs. Mathew, Ganapati, Jayakanthan, Nair, Gupta, Chebbi, Mandal, and Danda declare no conflicts of interest relevant to this manuscript.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Autoimmunity
Rights and permissions
About this article
Cite this article
Mathew, A.J., Ganapati, A., Kabeerdoss, J. et al. Chikungunya Infection: a Global Public Health Menace. Curr Allergy Asthma Rep 17, 13 (2017). https://doi.org/10.1007/s11882-017-0680-7
Published:
DOI: https://doi.org/10.1007/s11882-017-0680-7