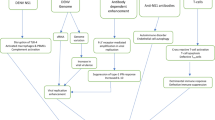
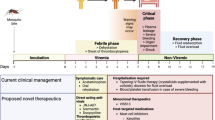
Abstract
Chikungunya virus (CHIKV), an alphavirus, has been responsible for large epidemic outbreaks with serious economic and social impact during the last 6 years. Transmitted by Aedes mosquitoes, it causes Chikungunya fever, an acute illness in patients with a stooped posture often associated with chronic and incapacitating arthralgia. The unprecedented re-emergence has stimulated renewed interest in CHIKV. This review discusses the advantages and disadvantages of different animal models for CHIKV infections and their importance to study the role of the immune system in different pathologies caused by CHIKV. We also reveal how such studies still present a difficult challenge, but are indispensible for mechanistic studies to further understand the pathophysiology of CHIKV infections.

Similar content being viewed by others
References
Her Z, Kam YW, Lin RTP, Ng LFP. Chikungunya: a bending reality. Microbes Infect. 2009;11:1165–76.
Watanaveeradej V, Endy TP, Simasathien S, Kerdpanich A, Polprasert N, Aree C, Vaughn DW, Nisalak A. The study transplacental Chikungunya virus antibody kinetics, Thailand. Emerg Infect Dis. 2006;12:1770–2.
Seneviratne SL, Perera J. Fever epidemic moves into Sri Lanka. BMJ. 2006;333:1220–1.
Kumarasamy V, Prathapa S, Zuridah H, Chem YK, Norizah I, Chua KB. Re-emergence of Chikungunya virus in Malaysia. Med J Malaysia. 2006;61:221–5.
Laras K, Sukri NC, Larasati RP, Bangs MJ, Kosim R, Djauzi S, Wandra T, Master J, Kosasih H, Hartati S, Beckett C, Sedyaningsih ER, Beecham HJ, Corwin AL. Tracking the re-emergence of epidemic Chikungunya virus in Indonesia. Trans R Soc Trop Med Hyg. 2005;99:128–41.
Ravi V. Re-emergence of Chikungunya virus in India. Indian J Med Microbiol. 2006;24:83–4.
Tsetsarkin KA, Vanlandingham DL, McGee CE, Higgs S. A single mutation in Chikungunya virus affects vector specificity and epidemic potential. PLoS Pathog. 2007;3:201.
Salvador B, Zhou Y, Michault A, Muench MO, Simmons G. Characterization of Chikungunya pseudotyped viruses: identification of refractory cell lines and demonstration of cellular tropism differences mediated by mutations in E1 glycoprotein. Virology. 2009;393:33–41.
Higgs S, Ziegler SA. A nonhuman primate model of Chikungunya disease. J Clin Invest. 2010;120:657–60.
Diseases of Environmental and Zoonotic Origin Team European Centre for Disease Prevention and Control. Chikungunya in Italy: actions in and implications for the European Union. Euro Surveill. 2007;12:E070906.2.
Sudeep AB, Parashar D. Chikungunya: an overview. J Biosci. 2008;33:443–9.
Robinson MC. An epidemic of virus disease in Southern Province, Tanganyika Territory, in 1952–53. I. Clinical features. Trans R Soc Trop Med Hyg. 1955;49:28–32.
Borgherini G, Poubeau P, Staikowsky F, Lory M, Le Moullec N, Becquart JP, Wengling C, Michault A, Paganin F. Outbreak of Chikungunya on Reunion Island: early clinical and laboratory features in 157 adult patients. Clin Infect Dis. 2007;44:1401–7.
Lakshmi V, Neeraja M, Subbalaxmi MVS, Parida MM, Dash PK, Santhosh SR, Rao PVL. Clinical features and molecular diagnosis of Chikungunya fever from South India. Clin Infect Dis. 2008;46:1436–42.
Carmona RJ, Shaikh S, Khalidi NA. Chikungunya viral polyarthritis. J Rheumatol. 2008;35:935–6.
Jaffar-Bandjee MC, Das T, Hoarau JJ, Krejbich Trotot P, Denizot M, Ribera A, Roques P, Gasque P. Chikungunya virus takes centre stage in virally induced arthritis: possible cellular and molecular mechanisms to pathogenesis. Microbes Infect. 2009;11:1206–18.
Mizuno Y, Kato Y, Takeshita N, Ujiie M, Kobayashi T, Kanagawa S, Kudo K, Lim CK, Takasaki T. Clinical and radiological features of imported chikungunya fever in Japan: a study of six cases at the National Center for Global Health and Medicine. J Infect Chemother. 2010;17(3):419–23.
Ozden S, et al. Human muscle satellite cells as targets of Chikungunya virus infection. PLoS ONE. 2007;2:e527.
Lokireddy S, Vemula S, Vadde R. Connective tissue metabolism in Chikungunya patients. Virol J. 2008;5:31.
Mohan A. Chikungunya fever: clinical manifestations & management. Indian J Med Res. 2006;124:471–4.
Brighton SW, Prozesky OW, de la Harpe AL. Chikungunya virus infection. A retrospective study of 107 cases. S Afr Med J. 1983;63:313–5.
McGill PE. Viral infections: alpha-viral arthropathy. Baillieres Clin Rheumatol. 1995;9:145–50.
De Andrade DC, Jean S, Clavelou P, Dallel R, Bouhassira D. Chronic pain associated with the Chikungunya fever: long lasting burden of an acute illness. BMC Infect Dis. 2010;10:31.
Borgherini G, Poubeau P, Jossaume A, Gouix A, Cotte L, Michault A, Arvin-Berod C, Paganin F. Persistent arthralgia associated with Chikungunya virus: a study of 88 adult patients on Reunion Island. Clin Infect Dis. 2008;47:469–75.
Solanki BS, Arya SC, Maheshwari P. Chikungunya disease with nephritic presentation. Int J Clin Pract. 2007;61:1941.
Mirabel M, Vignaux O, Lebon P, Legmann P, Weber S, Meune C. Acute myocarditis due to Chikungunya virus assessed by contrast-enhanced MRI. Int J Cardiol. 2007;121:e7–8.
Simon F, Paule P, Oliver M. Chikungunya virus-induced myopericarditis: toward an increase of dilated cardiomyopathy in countries with epidemics? Am J Trop Med Hyg. 2008;78:212–3.
Mittal A, Mittal S, Bharati MJ, Ramakrishnan R, Saravanan S, Sathe PS. Optic neuritis associated with chikungunya virus infection in South India. Arch Ophthalmol. 2007;125:1381–6.
Robin S, Ramful D, Le Seach’ F, Jaffar-Bandjee MC, Rigou G, Alessandri JL. Neurologic manifestations of pediatric Chikungunya infection. J Child Neurol. 2008;23:1028–35.
Chandak NH, Kashyap RS, Kabra D, Karandikar P, Saha SS, Morey SH, Purohit HJ, Taori GM, Daginawala HF. Neurological complications of Chikungunya virus infection. Neurol India. 2009;57:177–80.
Ganesan K, Diwan A, Shankar SK, Desai SB, Sainani GS, Katrak SM. Chikungunya encephalomyeloradiculitis: report of 2 cases with neuroimaging and 1 case with autopsy findings. AJNR Am J Neuroradiol. 2008;29:1636–7.
Lebrun G, Chadda K, Reboux AH, Martinet O, Gaüzère BA. Guillain-Barré syndrome after Chikungunya infection. Emerg Infect Dis. 2009;15:495–6.
Tournebize P, Charlin C, Lagrange M. Neurological manifestations in Chikungunya: about 23 cases collected in Reunion Island. Rev Neurol (Paris). 2009;165:48–51.
Arpino C, Curatolo P, Rezza G. Chikungunya and the nervous system: what we do and do not know. Rev Med Virol. 2009;19:121–9.
Das T, et al. Chikungunya fever: CNS infection and pathologies of a re-emerging arbovirus. Prog Neurobiol. 2010;91:121–9.
Ziegler SA, Lu L, da Rosa APAT, Xiao SY, Tesh RB. An animal model for studying the pathogenesis of Chikungunya virus infection. Am J Trop Med Hyg. 2008;79:133–9.
Couderc T, et al. A mouse model for Chikungunya: young age and inefficient type-I interferon signaling are risk factors for severe disease. PLoS Pathog. 2008;4:e29.
Suckling AJ, Jagelman S, Webb HE. A comparison of brain lysosomal enzyme activities in four experimental Togavirus encephalitides. J Neurol Sci. 1978;35:355–64.
Gardner J, Anraku I, Le TT, Larcher T, Major L, Roques P, Schroder WA, Higgs S, Suhrbier A. Chikungunya virus arthritis in adult wild-type mice. J Virol. 2010;84:8021–32.
Morrison TE, Oko L, Montgomery SA, Whitmore AC, Lotstein AR, Gunn BM, Elmore SA, Heise MT. A mouse model of Chikungunya virus-induced musculoskeletal inflammatory disease: evidence of arthritis, tenosynovitis, myositis, and persistence. Am J Pathol. 2011;178:32–40.
Thangamani S, Higgs S, Ziegler S, Vanlandingham D, Tesh R, Wikel S. Host immune response to mosquito-transmitted Chikungunya virus differs from that elicited by needle inoculated virus. PLoS ONE. 2010;5:e12137.
Schneider BS, Soong L, Girard YA, Campbell G, Mason P, Higgs S. Potentiation of West Nile encephalitis by mosquito feeding. Viral Immunol. 2006;19:74–82.
Edwards JF, Higgs S, Beaty BJ. Mosquito feeding-induced enhancement of Cache Valley Virus (Bunyaviridae) infection in mice. J Med Entomol. 1998;35:261–5.
Ross RW. The Newala epidemic. III. The virus: isolation, pathogenic properties and relationship to the epidemic. J Hyg (Lond). 1956;54:177–91.
Wang E, Volkova E, Adams AP, Forrester N, Xiao SY, Frolov I, Weaver SC. Chimeric alphavirus vaccine candidates for Chikungunya. Vaccine. 2008;26:5030–9.
Powers AM, Logue CH. Changing patterns of Chikungunya virus: re-emergence of a zoonotic arbovirus. J Gen Virol. 2007;88:2363–77.
Grivard P, et al. Molecular and serological diagnosis of Chikungunya virus infection. Pathol Biol (Paris). 2007;55:490–4.
Ziegler SA, Nuckols J, McGee CE, Huang YJS, Vanlandingham DL, Tesh RB, Higgs S. In vivo imaging of Chikungunya virus in mice and Aedes Mosquitoes using a Renilla Luciferase clone. Vector Borne Zoonotic Dis. 2011;11(11):1471–7.
Morrison TE, Whitmore AC, Shabman RS, Lidbury BA, Mahalingam S, Heise MT. Characterization of Ross River virus tropism and virus-induced inflammation in a mouse model of viral arthritis and myositis. J Virol. 2006;80:737–49.
Suckling AJ, Pathak S, Jagelman S, Webb HE. Virus-associated demyelination. A model using avirulent Semliki Forest virus infection of mice. J Neurol Sci. 1978;39:147–54.
Moss HE, Tansey EM, Milner P, Lincoln J, Burnstock G. Neuropeptide immunoreactivity and choline acetyltransferase activity in the mouse urinary bladder following inoculation with Semliki Forest Virus. J Auton Nerv Syst. 1990;31:47–56.
Coppenhaver DH, Singh IP, Sarzotti M, Levy HB, Baron S. Treatment of intracranial alphavirus infections in mice by a combination of specific antibodies and an interferon inducer. Am J Trop Med Hyg. 1995;52:34–40.
Safavi F, Feliberti JP, Raine CS, Mokhtarian F. Role of γδ T cells in antibody production and recovery from SFV demyelinating disease. J Neuroimmunol. 2011;235:18–26.
Kuehne RW, Pannier WL, Stephen EL. Indirect mouse model for the evaluation of potential antiviral compounds: results with Venezuelan Equine Encephalomyelitis virus. Antimicrob Agents Chemother. 1977;11:683–7.
Grieder FB, Davis BK, Zhou XD, Chen SJ, Finkelman FD, Gause WC. Kinetics of cytokine expression and regulation of host protection following infection with molecularly cloned Venezuelan Equine Encephalitis virus. Virology. 1997;233:302–12.
Aronson JF, Grieder FB, Davis NL, Charles PC, Knott T, Brown K, Johnston RE. A single-site mutant and revertants arising in vivo define early steps in the pathogenesis of Venezuelan Equine Encephalitis virus. Virology. 2000;270:111–23.
Cook SH, Griffin DE. Luciferase imaging of a neurotropic viral infection in intact animals. J Virol. 2003;77:5333–8.
Logue CH, et al. Virulence variation among isolates of Western Equine Encephalitis virus in an outbred mouse model. J Gen Virol. 2009;90:1848–58.
Nagata LP, Hu WG, Masri SA, Rayner GA, Schmaltz FL, Das D, Wu J, Long MC, Chan C, Proll D, Jager S, Jebailey L, Suresh MR, Wong JP. Efficacy of DNA vaccination against Western Equine Encephalitis virus infection. Vaccine. 2005;23:2280–3.
Schilte C, et al. Type I IFN controls Chikungunya virus via its action on nonhematopoietic cells. J Exp Med. 2010;207:429–42.
Dhanwani R, Khan M, Alam SI, Rao PVL, Parida M. Differential proteome analysis of Chikungunya virus-infected new-born mice tissues reveal implication of stress, inflammatory and apoptotic pathways in disease pathogenesis. Proteomics. 2011;11:1936–51.
Morrison TE, Fraser RJ, Smith PN, Mahalingam S, Heise MT. Complement contributes to inflammatory tissue destruction in a mouse model of Ross River virus-induced disease. J Virol. 2007;81:5132–43.
Morrison TE, Simmons JD, Heise MT. Complement receptor 3 promotes severe Ross River virus-induced disease. J Virol. 2008;82:11263–72.
Lidbury BA, Rulli NE, Suhrbier A, Smith PN, McColl SR, Cunningham AL, Tarkowski A, van Rooijen N, Fraser RJ, Mahalingam S. Macrophage-derived proinflammatory factors contribute to the development of arthritis and myositis after infection with an arthrogenic alphavirus. J Infect Dis. 2008;197:1585–93.
Herrero LJ, Nelson M, Srikiatkhachorn A, Gu R, Anantapreecha S, Fingerle-Rowson G, Bucala R, Morand E, Santos LL, Mahalingam S. Critical role for macrophage migration inhibitory factor (MIF) in Ross River virus-induced arthritis and myositis. Proc Natl Acad Sci U S A. 2011;108:12048–53.
Bradish CJ, Titmuss D, Fitzgeorge R. The sensitivity to gamma-irradiation of the phases of the virus-host interaction: studies with strains of Semliki Forest virus in mice. J Gen Virol. 1980;48:39–51.
Mokhtarian F, Huan CM, Roman C, Raine CS. Semliki Forest virus-induced demyelination and remyelination–involvement of B cells and anti-myelin antibodies. J Neuroimmunol. 2003;137:19–31.
Keogh B, Atkins GJ, Mills KHG, Sheahan BJ. Role of interferon-gamma and nitric oxide in the neuropathogenesis of avirulent Semliki Forest virus infection. Neuropathol Appl Neurobiol. 2003;29:553–62.
Fragkoudis R, Breakwell L, McKimmie C, Boyd A, Barry G, Kohl A, Merits A, Fazakerley JK. The type I interferon system protects mice from Semliki Forest virus by preventing widespread virus dissemination in extraneural tissues, but does not mediate the restricted replication of avirulent virus in central nervous system neurons. J Gen Virol. 2007;88:3373–84.
Charles PC, Walters E, Margolis F, Johnston RE. Mechanism of neuroinvasion of Venezuelan Equine Encephalitis virus in the mouse. Virology. 1995;208:662–71.
Jackson AC, Rossiter JP. Apoptotic cell death is an important cause of neuronal injury in experimental Venezuelan Equine Encephalitis virus infection of mice. Acta Neuropathol. 1997;93:349–53.
Keogh B, Sheahan BJ, Atkins GJ, Mills KHG. Inhibition of matrix metalloproteinases ameliorates blood-brain barrier disruption and neuropathological lesions caused by avirulent Semliki Forest virus infection. Vet Immunol Immunopathol. 2003;94:185–90.
Paessler S, Yun NE, Judy BM, Dziuba N, Zacks MA, Grund AH, Frolov I, Campbell GA, Weaver SC, Estes DM. Alpha-beta T cells provide protection against lethal encephalitis in the murine model of VEEV infection. Virology. 2007;367:307–23.
Levine B, Hardwick JM, Trapp BD, Crawford TO, Bollinger RC, Griffin DE. Antibody-mediated clearance of alphavirus infection from neurons. Science. 1991;254:856–60.
Tyor WR, Griffin DE. Virus specificity and isotype expression of intraparenchymal antibody-secreting cells during Sindbis virus encephalitis in mice. J Neuroimmunol. 1993;48:37–44.
Kam YW, Ong EKS, Rénia L, Tong JC, Ng LFP. Immuno-biology of Chikungunya and implications for disease intervention. Microbes Infect. 2009;11:1186–96.
Chow A, Her ZS, Ong KS, Chen J, Dimatatac F, Kwek DJC, Barkham T, Yang H, Renia L, Leo YS, Ng LFP. Persistent arthralgia induced by Chikungunya virus infection is associated with interleukin-6 and granulocyte macrophage colony-stimulating factor. JID. 2011;203:149–57.
Ng LFP, Chow A, Sun YJ, Kwek DJC, Lim PL, Dimatatac F, Ng LC, Ooi EE, Choo KH, Her Z, Kourilsky P, Leo YS. IL-1beta, IL-6, and RANTES as biomarkers of Chikungunya severity. PLoS ONE. 2009;4:e4261.
Hoarau JJ, et al. Persistent chronic inflammation and infection by Chikungunya arthritogenic alphavirus in spite of a robust host immune response. J Immunol. 2010;184:5914–27.
Chirathaworn C, Rianthavorn P, Wuttirattanakowit N, Poovorawan Y. Serum IL-18 and IL-18BP levels in patients with Chikungunya virus infection. Viral Immunol. 2010;23:113–7.
Chaaitanya IK, Muruganandam N, Sundaram SG, Kawalekar O, Sugunan AP, Manimunda SP, Ghosal SR, Muthumani K, Vijayachari P. Role of proinflammatory cytokines and chemokines in chronic arthropathy in CHIKV infection. Viral Immunol. 2011;24:265–71.
Kelvin AA, et al. Inflammatory cytokine expression is associated with Chikungunya virus resolution and symptom severity. PLoS Negl Trop Dis. 2011;5:e1279.
Sourisseau M, et al. Characterization of reemerging Chikungunya virus. PLoS Pathog. 2007;3:e89.
Labadie K, et al. Chikungunya disease in nonhuman primates involves long-term viral persistence in macrophages. J Clin Invest. 2010;120:894–906.
Her Z, Malleret B, Chan M, Ong EKS, Wong SC, Kwek DJC, Tolou H, Lin RTP, Tambyah PA, Rénia L, Ng LFP. Active infection of human blood monocytes by Chikungunya virus triggers an innate immune response. J Immunol. 2010;184:5903–13.
Randall RE, Goodbourn S. Interferons and viruses: an interplay between induction, signalling, antiviral responses and virus countermeasures. J Gen Virol. 2008;89:1–47.
Werneke SW, Schilte C, Rohatgi A, Monte KJ, Michault A, Arenzana-Seisdedos F, Vanlandingham DL, Higgs S, Fontanet A, Albert ML, Lenschow DJ. ISG15 is critical in the control of Chikungunya virus infection independent of UbE1L mediated conjugation. PLoS Pathog. 2011;7:e1002322.
Fros JJ, Liu WJ, Prow NA, Geertsema C, Ligtenberg M, Vanlandingham DL, Schnettler E, Vlak JM, Suhrbier A, Khromykh AA, Pijlman GP. Chikungunya virus nonstructural protein 2 inhibits type I/II interferon-stimulated JAK-STAT signaling. J Virol. 2010;84:10877–87.
Manimunda SP, Vijayachari P, Uppoor R, Sugunan AP, Singh SS, Rai SK, Sudeep AB, Muruganandam N, Chaitanya IK, Guruprasad DR. Clinical progression of Chikungunya fever during acute and chronic arthritic stages and the changes in joint morphology as revealed by imaging. Trans R Soc Trop Med Hyg. 2010;104:392–9.
Kulmatycki KM, Jamali F. Drug disease interactions: Role of inflammatory mediators in pain and variability in analgesic drug response. J Pharm Pharm Sci. 2007;10:554–66.
Asquith DL, McInnes IB. Emerging cytokine targets in rheumatoid arthritis. Curr Opin Rheumatol. 2007;19:246–51.
Bryceson YT, Long EO. Line of attack: NK cell specificity and integration of signals. Curr Opin Immunol. 2008;20:344–52.
Lanier LL. Evolutionary struggles between NK cells and viruses. Nat Rev Immunol. 2008;8:259–68.
Petitdemange C, Becquart P, Wauquier N, Béziat V, Debré P, Leroy EM, Vieillard V. Unconventional repertoire profile is imprinted during acute Chikungunya infection for natural killer cells polarization toward cytotoxicity. PLoS Pathog. 2011;7:e1002268.
Rulli NE, Rolph MS, Srikiatkhachorn A, Anantapreecha S, Guglielmotti A, Mahalingam S. Protection from arthritis and myositis in a mouse model of acute Chikungunya virus disease by bindarit, an inhibitor of monocyte chemotactic protein-1 synthesis. J Infect Dis. 2011;204:1026–30.
Wauquier N, Becquart P, Nkoghe D, Padilla C, Ndjoyi-Mbiguino A, Leroy EM. The acute phase of Chikungunya virus infection in humans is associated with strong innate immunity and T CD8 cell activation. J Infect Dis. 2011;204:115–23.
Linn ML, Mateo L, Gardner J, Suhrbier A. Alphavirus-specific cytotoxic T lymphocytes recognize a cross-reactive epitope from the capsid protein and can eliminate virus from persistently infected macrophages. J Virol. 1998;72:5146–53.
Soden M, Vasudevan H, Roberts B, Coelen R, Hamlin G, Vasudevan S, La Brooy J. Detection of viral ribonucleic acid and histologic analysis of inflamed synovium in Ross River virus infection. Arthritis Rheum. 2000;43:365–9.
Yun NE, Peng BH, Bertke AS, Borisevich V, Smith JK, Smith JN, Poussard AL, Salazar M, Judy BM, Zacks MA, Estes DM, Paessler S. CD4+ T cells provide protection against acute lethal encephalitis caused by Venezuelan Equine Encephalitis virus. Vaccine. 2009;27:4064–73.
Latif Z, Gates D, Wust CJ, Brown A. Cross protection among Togaviruses in nude mice and littermates. J Gen Virol. 1979;45:89–98.
Gershon RK, Kondo K. Cell interactions in the induction of tolerance: the role of thymic lymphocytes. Immunology. 1970;18:723–37.
Gershon RK, Cohen P, Hencin R, Liebhaber SA. Suppressor T Cells. J Immunol. 1972;108(3):586–90.
Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol. 1995;155:1151–64.
Olivares-Villagómez D, Wang YJ, Lafaille JJ. Regulatory CD4+ T cells expressing endogenous T cell receptor chains protect myelin basic protein–specific transgenic mice from spontaneous autoimmune encephalomyelitis. J Exp Med. 1998;188:1883–94.
Annacker O, Pimenta-Araujo R, Burlen-Defranoux O, Barbosa TC, Cumano A, Bandeira A. CD25+ CD4+ T cells regulate the expansion of peripheral CD4 T cells through the production of IL-10. J Immunol. 2001;166(5):3008–18.
Lanteri MC, et al. Tregs control the development of symptomatic West Nile virus infection in humans and mice. J Clin Invest. 2009;119:3266–77.
Belkaid Y. Regulatory T cells and infection: a dangerous necessity. Nat Rev Immunol. 2007;7(11):875–88.
Lund JM, Hsing L, Pham TT, Rudensky AY. Coordination of early protective immunity to viral infection by regulatory T cells. Science. 2008;320:1220–4.
Lühn K, et al. Increased frequencies of CD4+ CD25 (high) regulatory T cells in acute Dengue infection. J Exp Med. 2007;204:979–85.
Schwartz O, Albert ML. Biology and pathogenesis of Chikungunya virus. Nat Rev Microbiol. 2010;8:491–500.
Chia PY. Chikungunya fever: a review of a re-emerging mosquito-borne infectious disease and the current status. Curr Res Technol Educ Topics Appl Microbiol Microb Biotechnol. 2010;1:597.
Roediger B, Ng LG, Smith AL, Fazekas de St Groth B, Weninger W. Visualizing dendritic cell migration within the skin. Histochem Cell Biol. 2008;130:1131–46.
Ishii T, Ishii M. Intravital two-photon imaging: a versatile tool for dissecting the immune system. Ann Rheum Dis. 2011;70(Suppl 1):i113–5.
Akahata W, Yang ZY, Andersen H, Sun S, Holdaway HA, Kong WP, Lewis MG, Higgs S, Rossmann MG, Rao S, Nabel GJ. A virus-like particle vaccine for epidemic Chikungunya virus protects nonhuman primates against infection. Nat Med. 2010;16:334–8.
Mallilankaraman K, et al. A DNA vaccine against Chikungunya virus is protective in mice and induces neutralizing antibodies in mice and nonhuman primates. PLoS Negl Trop Dis. 2011;5:e928.
Wang D, Suhrbier A, Penn-Nicholson A, Woraratanadharm J, Gardner J, Luo M, Le TT, Anraku I, Sakalian M, Einfeld D, Dong JY. A complex adenovirus vaccine against Chikungunya virus provides complete protection against viraemia and arthritis. Vaccine. 2011;29:2803–9.
Plante K, et al. Novel Chikungunya vaccine candidate with an IRES-based attenuation and host range alteration mechanism. PLoS Pathog. 2011;7:e1002142.
Mirolo M, Fabbri M, Sironi M, Vecchi A, Guglielmotti A, Mangano G, Biondi G, Locati M, Mantovani A. Impact of the anti-inflammatory agent bindarit on the chemokinome: selective inhibition of the monocyte chemotactic proteins. Eur Cytokine Netw. 2008;19:119–22.
Acknowledgments
We wish to thank Nicholas K.W. Yeo for helpful discussions and suggestions. We are also grateful to Laurent Renia for critical reading of this manuscript. This work is supported in part by the intramural research funds by the Biomedical Research Council (BMRC), Agency for Science, Technology and Research (A*STAR) and also A*STAR’s Joint Council Office (JCO) Research Grant (grant number: CCOGA02_008_2008). T. Teo is funded by a post-graduate scholarship by the A*STAR Graduate Academy (AGA), and F. Lum is funded by a post-graduate research scholarship by the National University of Singapore (NUS).
Author information
Authors and Affiliations
Corresponding author
Additional information
Teck-Hui Teo and Fok-Moon Lum contributed equally to this work.
Rights and permissions
About this article
Cite this article
Teo, TH., Lum, FM., Lee, W.W.L. et al. Mouse models for Chikungunya virus: deciphering immune mechanisms responsible for disease and pathology. Immunol Res 53, 136–147 (2012). https://doi.org/10.1007/s12026-012-8266-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-012-8266-x