Abstract
Purpose
Upper airway stimulation (UAS) is an effective treatment for obstructive sleep apnea (OSA) in positive airway pressure (PAP) failure. Most reports have presented short-term data, so long-term safety and efficacy reports are rare. The German post-market study (G-PMS) has followed approximately 60 patients from three implanting centers for several years.
Methods
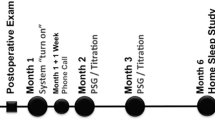
Patients with OSA and PAP failure qualified for the G-PMS by the absence of obesity class 2 an AHI between 15 and 65 events/h and absence of complete concentric collapse at the velum during drug-induced sleep endoscopy. Optional 2- and 3-year follow-ups after implantation were collected during routine clinical practice. We measured respiratory parameters such as apnea–hypopnea index (AHI) and oxygen desaturation index (ODI) and daytime sleepiness using the Epworth sleepiness scale (ESS) in a per protocol analysis. Usage was calculated from device-downloaded reports. Device-related complications were documented.
Results
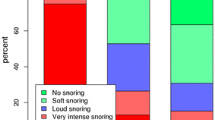
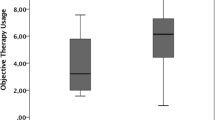
Of the 60 original patients, 41 returned for 2-year follow-up, and 38 for 3 years. About 76% at 2 years and 68% at 3 years met the criterion of therapy success defined as an AHI below 15/h. The median AHI was reduced from 28.6/h (baseline) to 9.0/h (2 years) and 10.0/h (3 years); whereas median ODI decreased from 27.0 to 6.3/h (2 years), and 8.3/h (3 years). Median ESS improved from baseline 13 points to 4 (2 years) and 6 (3 years). Usage was stable at approximately 45 h per week at 2 and 3 years. Serious device-related adverse events were rare, with two-device explantation between 12 to 36 months postoperatively.
Conclusions
The German multi-center long-term outcomes compare favorably with previously published studies. Respiratory and sleepiness efficacy outcomes were sustained over 2 and 3 years, with a favorable safety profile, supporting the safety and efficacy of a chronic implantable therapy.

Similar content being viewed by others
References
Vanderveken OM, Maurer JT, Hohenhorst W et al (2013) Evaluation of drug-induced sleep endoscopy as a patient selection tool for implanted upper airway stimulation for obstructive sleep apnea. J Clin Sleep Med 9:433–438
Hasselbacher K, Bruchhage KL, Abrams N, Steffen A (2018) Sleep endoscopy and complete concentric collapse in CPAP failure. HNO 66:837–842
Heiser C, Thaler E, Boon M, Soose RJ, Woodson BT (2016) Updates of operative techniques for upper airway stimulation. Laryngoscope 126(Suppl 7):S12–S16
Heiser C, Hofauer B, Lozier L, Woodson BT, Stark T (2016) Nerve monitoring-guided selective hypoglossal nerve stimulation in obstructive sleep apnea patients. Laryngoscope 126:2852–2858
Steffen A, Kilic A, König IR, Suurna MV, Hofauer B, Heiser C (2018) Tongue motion variability with changes of upper airway stimulation electrode configuration and effects on treatment outcomes. Laryngoscope 128:1970–1976
Strollo PJ Jr, Soose RJ, Maurer JT, STAR Trial Group et al (2014) Upper-airway stimulation for obstructive sleep apnea. N Engl J Med 370:139–149
Woodson BT, Gillespie MB, Soose RJ, STAR Trial Investigators; STAR Trial Investigators et al (2014) Randomized controlled withdrawal study of upper airway stimulation on OSA: short- and long-term effect. Otolaryngol Head Neck Surg 151:880–887
Soose RJ, Woodson BT, Gillespie MB, STAR Trial Investigators et al (2016) Upper airway stimulation for obstructive sleep apnea: self-reported outcomes at 24 months. J Clin Sleep Med 12:43–48
Gillespie MB, Soose RJ, Woodson BT, Strohl KP, Maurer JT, de Vries N, Steward DL, Baskin JZ, Badr MS, Lin HS, Padhya TA, Mickelson S, Anderson WMD, Vanderveken OM, Strollo PJ Jr, on Behalf of the STAR Trial Investigators STAR Trial Investigators(2017) Upper airway stimulation for obstructive sleep apnea: patient-reported outcomes after 48 months of follow-up. Otolaryngol Head Neck Surg 156:765–771
Woodson BT, Soose RJ, Gillespie MB, STAR Trial Investigators et al (2016) Three-year outcomes of cranial nerve stimulation for obstructive sleep apnea: the STAR trial. Otolaryngol Head Neck Surg 154:181–188
Woodson BT, Strohl KP, Soose RJ, Gillespie MB, Maurer JT, de Vries N, Padhya TA, Badr MS, Lin HS, Vanderveken OM, Mickelson S, Strollo PJ Jr STAR Trial Investigators(2018) Upper airway stimulation for obstructive sleep apnea: 5-year outcomes. Otolaryngol Head Neck Surg 159:194–202
Boon M, Huntley C, Steffen A, ADHERE Registry Investigators et al (2018) Upper airway stimulation for obstructive sleep apnea: results from the ADHERE registry. Otolaryngol Head Neck Surg 159:379–385
Heiser C, Steffen A, Boon M, ADHERE registry investigators et al (2019) Post-approval upper airway stimulation predictors of treatment effectiveness in the ADHERE registry. Eur Respir J 53:1801405. https://doi.org/10.1183/13993003.01405-2018
Heiser C, Maurer JT, Hofauer B, Sommer UJ, Seitz A, Steffen A (2017) Outcomes of upper airway stimulation for obstructive sleep apnea in a multi-center German post-market study. Otolaryngol Head Neck Surg 156:378–384
Steffen A, Sommer JU, Hofauer B, Maurer JT, Hasselbacher K, Heiser C (2018) Outcome after one year of upper airway stimulation for obstructive sleep apnea in a multicenter German post-market study. Laryngoscope 128:509–515
Hasselbacher K, Hofauer B, Maurer JT, Heiser C, Steffen A, Sommer JU (2018) Patient-reported outcome: results of the multicenter German post-market study. Eur Arch Otorhinolaryngol 275:1913–1919
Steffen A, Hartmann JT, König IR, Ravesloot MJL, Hofauer B, Heiser C (2018) Evaluation of body position in upper airway stimulation for obstructive sleep apnea-is continuous voltage sufficient enough? Sleep Breath 22:1207–1212
Pietzsch JB, Richter AK, Randerath W, Steffen A, Liu S, Geisler BP, Wasem J, Biermann-Stallwitz J (2019) Clinical and economic benefits of upper airway stimulation for obstructive sleep apnea in a European setting. Respiration 98:38–47
Steffen A, Abrams N, Suurna MV, Wollenberg B, Hasselbacher K (2019) Upper-airway stimulation before, after, or without Uvulopalatopharyngoplasty: a two-year perspective. Laryngoscope 129:514–518
Maurer JT, Van de Heyning PH, Lin H-S et al (2012) Operative technique of upper airway stimulation: an implantable treatment of obstructive sleep apnea. Oper Tech Otolaryngol Head Neck Surg 23:227–233
Heiser C, Knopf A, Hofauer B (2017) Surgical anatomy of the hypoglossal nerve: a new classification system for selective upper airway stimulation. Head Neck 39:2371–2380
Hofauer B, Steffen A, Knopf A, Hasselbacher K, Heiser C (2018) Adherence to upper-airway stimulation in the treatment of OSA. Chest 153:574–575
McArdle N, Devereux G, Heidarnejad H et al (1999) Long-term use of CPAP therapy for sleep apnea/hypopnea syndrome. Am J Respir Crit Care Med 159:1108–1114
Krieger J, Kurtz D, Petiau C, Sforza E, Trautmann D (1996) Long-term compliance with CPAP therapy in obstructive sleep apnea patients and in snorers. Sleep 19:S136–S143
Benjafield AV, Pépin JL, Valentine K et al (2019) Compliance after switching from CPAP to bilevel for patients with non-compliant OSA: big data analysis. BMJ Open Respir Res 6(1):e000380
Acknowledgments
The authors would like to express their gratitude to the study nurses and colleagues of the local sleep laboratories Katharina Eckbauer, Nicole Behn, Oliver Schmidt, and Sabrina Sihman for their commitment in collecting the necessary data for this study. Underlying data for the 1-year evaluation from German post-market study was furnished by Inspire Medical Systems, Inc.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Armin Steffen, Ulrich J. Sommer, Joachim T. Maurer and Clemens Heiser are study investigators and received honoraria and travel expenses for invited talks on behalf of Inspire Medical, Inc., outside the submitted work. Clemens Heiser is a consultant for Inspire Medical Systems. Benedikt Hofauer received travel expenses from Inspire Medical, Inc. Nils Abrams has nothing to declare.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The German post-market study was approved by the ethics committee at all three institutions and was registered as NCT02293746 on clinicaltrials.gov.
Informed consent
Informed consent was obtained from all individual participants included in the German post-market study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Steffen, A., Sommer, U.J., Maurer, J.T. et al. Long-term follow-up of the German post-market study for upper airway stimulation for obstructive sleep apnea. Sleep Breath 24, 979–984 (2020). https://doi.org/10.1007/s11325-019-01933-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-019-01933-0